CHILDRENS ALLERGY - diphenhydramine hydrochloride solution
C.D.M.A., Inc.
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
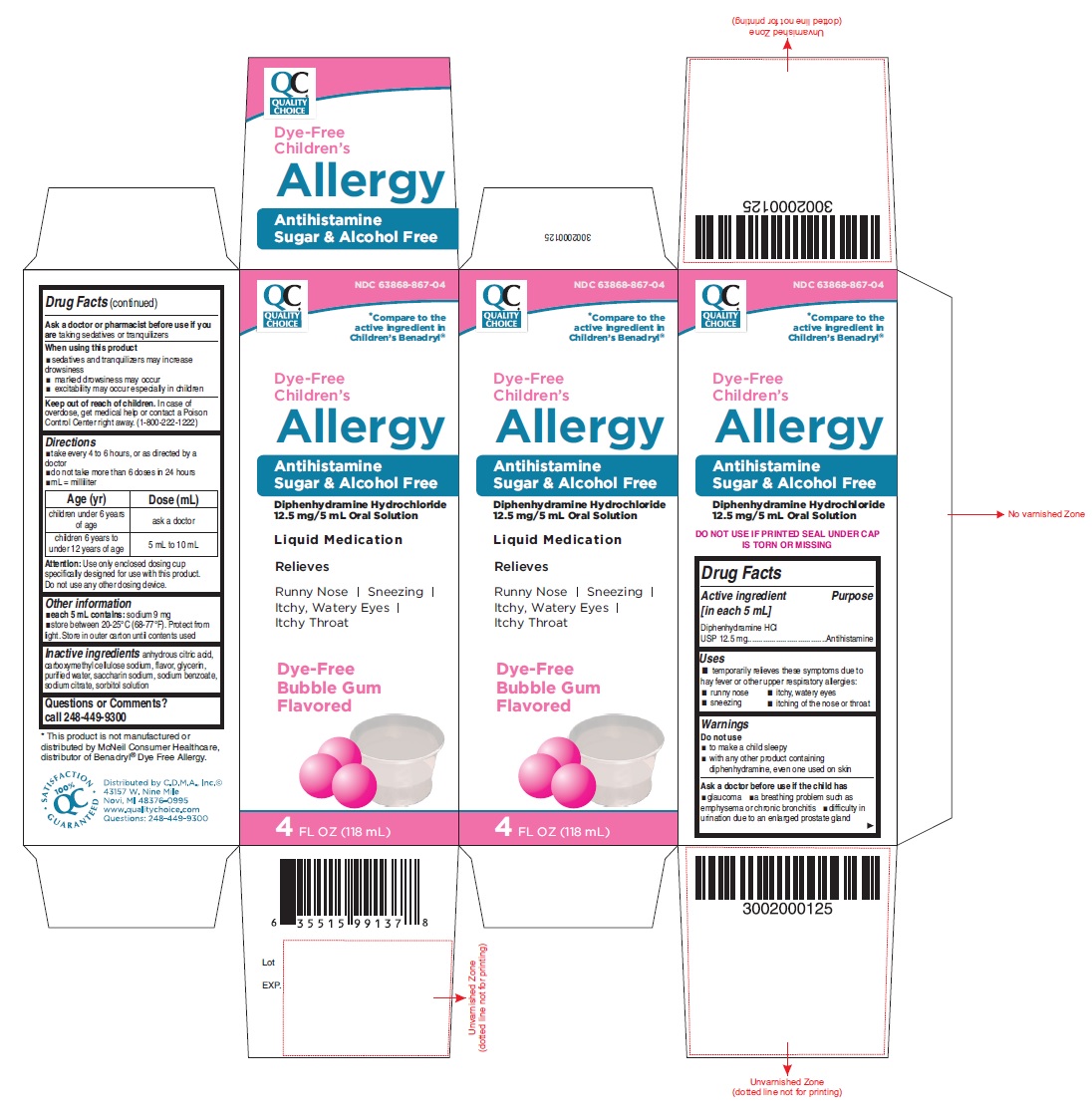
Uses
- temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- runny nose
- sneezing
- itchy, watery eyes
- itching of the nose or throat
Warnings
Do not use
- to make a child sleepy
- with any other product containing diphenhydramine, even one used on skin
Ask a doctor before use if the child has
- glaucoma
- a breathing problem such as emphysema or chronic bronchitis
- difficulty in urination due to an enlarged prostate gland
When using this product
- sedatives and tranquilizers may increase drowsiness
- marked drowsiness may occur
- excitability may occur, especially in children
Keep out of reach of children.
In case of overdose, get medical help or contact a Poison Control Center right away. (1-800-222-1222)
Directions
- take every 4 to 6 hours, or as directed by a doctor
- do not take more than 6 doses in 24 hours
- mL = milliliter
| Age (yr)
| Dose (mL)
|
| children under 6 years of age | ask a doctor |
| children 6 years to under 12 years of age | 5 mL to 10 mL |
Attention: Use only enclosed dosing cup specifically designed for use with this product. Do not use any other dosing device.
Other information
- each 5 mL contains: sodium 9 mg
- store between 20-25°C (68-77°F). Protect from light. Store in outer carton until contents used
Inactive ingredients
anhydrous citric acid, carboxymethyl cellulose sodium, flavor, glycerin, purified water, saccharin sodium, sodium benzoate, sodium citrate, sorbitol solution
Questions or Comments?
call 248-449-9300
* This product is not manufactured or distributed by McNeil Consumer Healthcare, distributor of Benadryl® Dye Free Allergy.
Distributed by:
C.D.M.A, Inc.©
43157 W.Nine Mile
Novi.MI 48376-0995
www.qualitychoice.com
Questions: 248-449-9300
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 4 FL OZ (118 mL Bottle)
NDC 63868-867-04
*Compare to the
active Ingredient in
children’s Benadryl®
Dye-Free
Children’s
Allergy
Antihistamine
Sugar & Alcohol Free
Diphenhydramine Hydrochloride
12.5 mg/5 mL Oral Solution
Liquid Medication
Relieves
Runny Nose | Sneezing
Itchy, Watery Eyes |
Itchy Throat
Dye-Free
Bubble Gum
Flavored
4 FL OZ (118 mL)
| CHILDRENS ALLERGY
diphenhydramine hydrochloride solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - C.D.M.A., Inc. (011920774) |
| Registrant - Aurohealth LLC (078728447) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Aurohealth LLC | 078728447 | MANUFACTURE(63868-867) | |