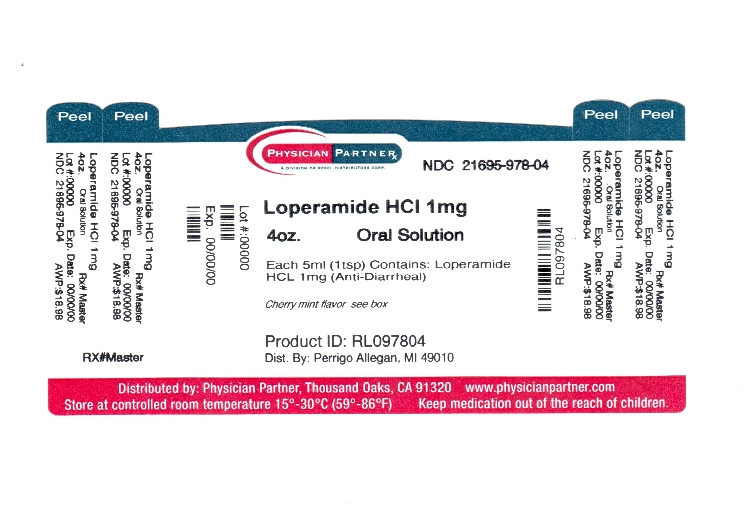
Label: ANTI DIARRHEAL- loperamide hcl solution
-
Contains inactivated NDC Code(s)
NDC Code(s): 21695-978-04 - Packager: Rebel Distributors Corp
- This is a repackaged label.
- Source NDC Code(s): 0113-0377
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated September 13, 2011
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient (in each 5 mL)
- Purpose
- Uses
-
Warnings
Allergy alert: Do not use if you have ever had a rash or other allergic reaction to loperamide HCl
When using this product
- tiredness, drowsiness or dizziness may occur. Be careful when driving or operating machinery.
-
Directions
- drink plenty of clear fluids to help prevent dehydration caused by diarrhea
- find right dose on chart. If possible, use weight to dose; otherwise use age.
- only use attached measuring cup to dose product
adults and children 12 years and over 4 teaspoonfuls (1 dosage cup) after the first loose stool; 2 teaspoonfuls (1/2 dosage cup) after each subsequent loose stool; but no more than 8 teaspoonfuls in 24 hours children 9-11 years (60-95 lbs) 2 teaspoonfuls (1/2 dosage cup) after the first loose stool; 1 teaspoonful (1/4 dosage cup) after each subsequent loose stool; but no more than 6 teaspoonfuls in 24 hours children 6-8 years (48-59 lbs) 2 teaspoonfuls (1/2 dosage cup) after the first loose stool; 1 teaspoonful (1/4 dosage cup) after each subsequent loose stool; but no more than 4 teaspoonfuls in 24 hours children under 6 years (up to 47 lbs) ask a doctor - Other information
- Inactive ingredients
- Questions or comments?
- Principal Display Panel
-
INGREDIENTS AND APPEARANCE
ANTI DIARRHEAL
loperamide hcl solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:21695-978(NDC:0113-0377) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LOPERAMIDE HYDROCHLORIDE (UNII: 77TI35393C) (LOPERAMIDE - UNII:6X9OC3H4II) LOPERAMIDE HYDROCHLORIDE 1 mg in 5 mL Inactive Ingredients Ingredient Name Strength ALCOHOL (UNII: 3K9958V90M) BENZOIC ACID (UNII: 8SKN0B0MIM) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) GLYCERIN (UNII: PDC6A3C0OX) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) SODIUM BENZOATE (UNII: OJ245FE5EU) SORBITOL (UNII: 506T60A25R) SUCROSE (UNII: C151H8M554) Product Characteristics Color ORANGE (amber, clear, pale) Score Shape Size Flavor CHERRY (cherry mint) Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:21695-978-04 1 in 1 CARTON 1 120 mL in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA073243 07/13/1992 Labeler - Rebel Distributors Corp (118802834) Establishment Name Address ID/FEI Business Operations Rebel Distributors Corp 118802834 RELABEL, REPACK