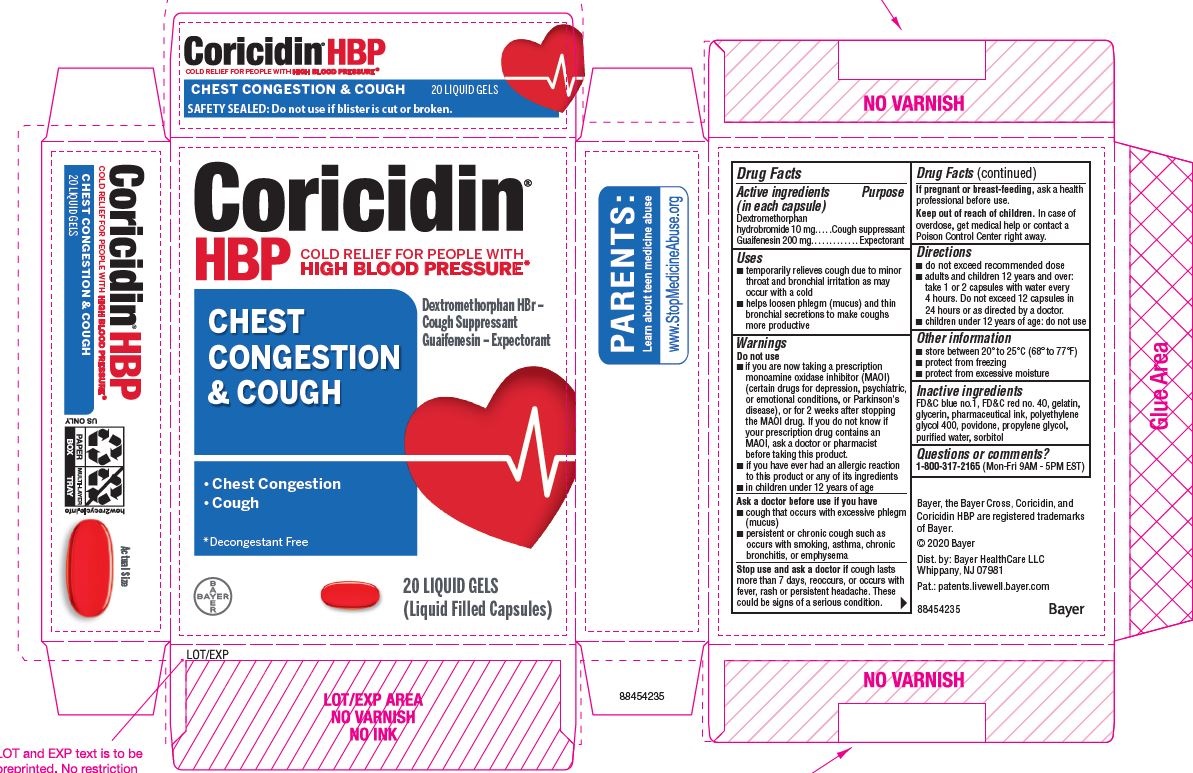
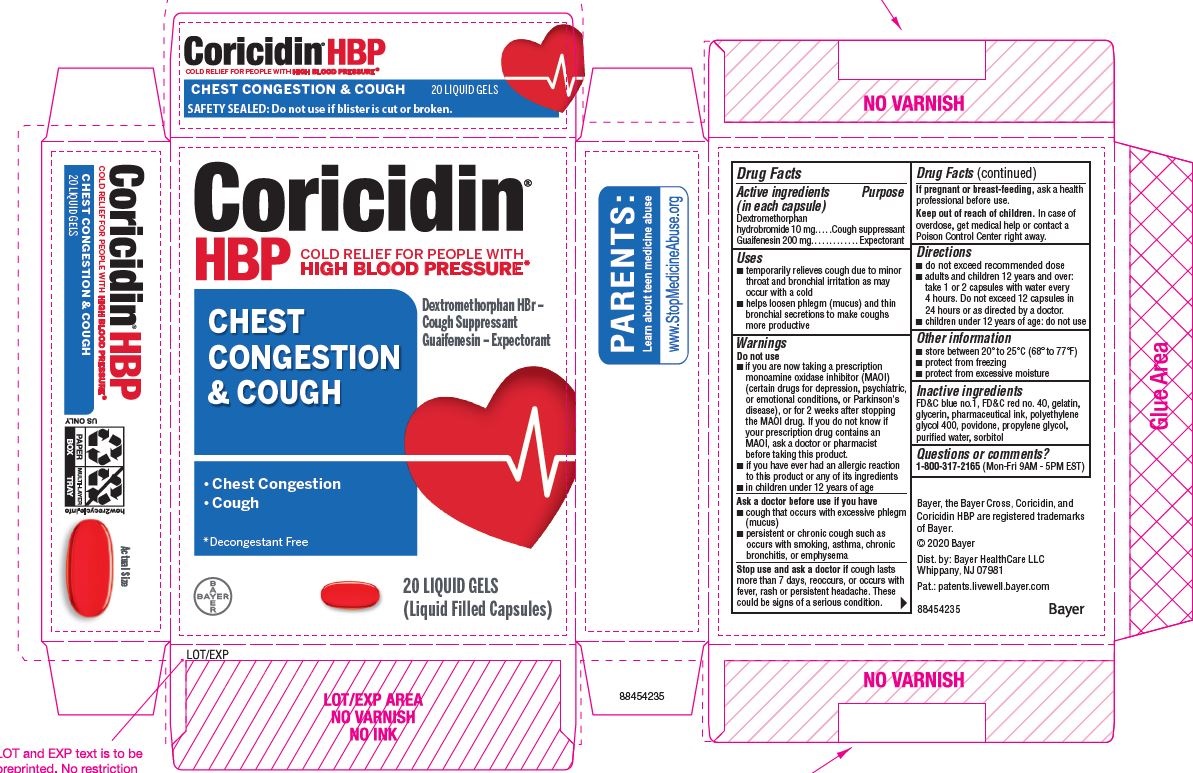
Label: CORICIDIN HBP CHEST CONGESTION AND COUGH- dextromethorphan hydrobromide and guaifenesin capsule, gelatin coated
- NDC Code(s): 11523-7158-2
- Packager: Bayer HealthCare LLC.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated March 28, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
- PURPOSE
- Uses
-
Warnings
Do not use
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
- if you have ever had an allergic reaction to this product or any of its ingredients
Ask a doctor before use if you have
- cough that occurs with excessive phlegm (mucus)
- persistent or chronic cough as occurs with smoking, asthma, chronic bronchitis, or emphysema
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
-
PRINCIPAL DISPLAY PANEL - 2 x 10 Softgel Blister Pack Carton
Coricidin ®
HBP
COLD SYMPTOM RELIEF for people with
HIGH BLOOD PRESSURECHEST
CONGESTION
& COUGHRelieves:
- Chest Congestion
- Cough
Guaifenesin - Expectorant,
Dextromethorphan hydrobromide -
cough suppressent
CONTROLS COUGH
FASTLiqui-Gels ®
DECONGESTANT-FREE
American Heart Association
Health Information Enclosed
20 LIQUID GELS
(Liquid Filled Capsules)
-
INGREDIENTS AND APPEARANCE
CORICIDIN HBP CHEST CONGESTION AND COUGH
dextromethorphan hydrobromide and guaifenesin capsule, gelatin coatedProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:11523-7158 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DEXTROMETHORPHAN HYDROBROMIDE (UNII: 9D2RTI9KYH) (DEXTROMETHORPHAN - UNII:7355X3ROTS) DEXTROMETHORPHAN HYDROBROMIDE 10 mg GUAIFENESIN (UNII: 495W7451VQ) (GUAIFENESIN - UNII:495W7451VQ) GUAIFENESIN 200 mg Inactive Ingredients Ingredient Name Strength SODIUM HYDROXIDE (UNII: 55X04QC32I) SHELLAC (UNII: 46N107B71O) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) SORBITAN (UNII: 6O92ICV9RU) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FD&C RED NO. 40 (UNII: WZB9127XOA) GELATIN (UNII: 2G86QN327L) GLYCERIN (UNII: PDC6A3C0OX) POVIDONE (UNII: FZ989GH94E) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SORBITOL (UNII: 506T60A25R) WATER (UNII: 059QF0KO0R) Product Characteristics Color red Score no score Shape OVAL Size 20mm Flavor Imprint Code C;HBP Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:11523-7158-2 2 in 1 CARTON 08/31/2000 04/30/2022 1 10 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part341 08/31/2000 Labeler - Bayer HealthCare LLC. (112117283)