PULMICORT TURBUHALER- budesonide powder, metered
AstraZeneca Pharmaceuticals LP
----------
PULMICORT TURBUHALER 200 mcg
(budesonide inhalation powder)
For Oral Inhalation Only.
DESCRIPTION
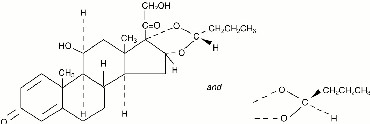
Budesonide, the active component of PULMICORT TURBUHALER 200 mcg, is a corticosteroid designated chemically as (RS)-11β, 16α, 17,21-Tetrahydroxypregna-1,4-diene-3,20-dione cyclic 16,17-acetal with butyraldehyde. Budesonide is provided as a mixture of two epimers (22R and 22S). The empirical formula of budesonide is C25H34O6 and its molecular weight is 430.5. Its structural formula is:
Budesonide is a white to off-white, tasteless, odorless powder that is practically insoluble in water and in heptane, sparingly soluble in ethanol, and freely soluble in chloroform. Its partition coefficient between octanol and water at pH 7.4 is 1.6 x 103.
PULMICORT TURBUHALER is an inhalation-driven multi-dose dry powder inhaler that contains only micronized budesonide. Each actuation of PULMICORT TURBUHALER provides 200 mcg budesonide per metered dose, which delivers approximately 160 mcg budesonide from the mouthpiece (based on in vitro testing at 60 L/min for 2 sec).
In vitro testing has shown that the dose delivery for PULMICORT TURBUHALER is substantially dependent on airflow through the device. Patient factors such as inspiratory flow rates will also affect the dose delivered to the lungs of patients in actual use (see Patient’s Instructions for Use). In adult patients with asthma (mean FEV1 2.9 L [0.8 - 5.1 L]) mean peak inspiratory flow (PIF) through PULMICORT TURBUHALER was 78 (40-111) L/min. Similar results (mean PIF 82 [43-125] L/min) were obtained in asthmatic children (6 to 15 years, mean FEV1 2.1 L [0.9 - 5.4 L]). Patients should be carefully instructed on the use of this drug product to assure optimal dose delivery.
CLINICAL PHARMACOLOGY
Mechanism of Action
Budesonide is an anti-inflammatory corticosteroid that exhibits potent glucocorticoid activity and weak mineralocorticoid activity. In standard in vitro and animal models, budesonide has approximately a 200-fold higher affinity for the glucocorticoid receptor and a 1000-fold higher topical anti-inflammatory potency than cortisol (rat croton oil ear edema assay). As a measure of systemic activity, budesonide is 40 times more potent than cortisol when administered subcutaneously and 25 times more potent when administered orally in the rat thymus involution assay.
The activity of PULMICORT TURBUHALER is due to the parent drug, budesonide. In glucocorticoid receptor affinity studies, the 22R form was two times as active as the 22S epimer. In vitro studies indicated that the two forms of budesonide do not interconvert.
The precise mechanism of corticosteroid actions on inflammation in asthma is not known. Inflammation is an important component in the pathogenesis of asthma. Corticosteroids have been shown to have a wide range of inhibitory activities against multiple cell types (eg, mast cells, eosinophils, neutrophils, macrophages, and lymphocytes) and mediators (eg, histamine, eicosanoids, leukotrienes, and cytokines) involved in allergic and non-allergic-mediated inflammation. These anti-inflammatory actions of corticosteroids may contribute to their efficacy in asthma.
Studies in asthmatic patients have shown a favorable ratio between topical anti-inflammatory activity and systemic corticosteroid effects over a wide range of doses from PULMICORT TURBUHALER. This is explained by a combination of a relatively high local anti-inflammatory effect, extensive first pass hepatic degradation of orally absorbed drug (85-95%), and the low potency of formed metabolites (see below).
Pharmacokinetics
Absorption: After oral administration of budesonide, peak plasma concentration was achieved in about 1 to 2 hours and the absolute systemic availability was 6-13%. In contrast, most of budesonide delivered to the lungs is systemically absorbed. In healthy subjects, 34% of the metered dose was deposited in the lungs (as assessed by plasma concentration method) with an absolute systemic availability of 39% of the metered dose. Pharmacokinetics of budesonide do not differ significantly in healthy volunteers and asthmatic patients. Peak plasma concentrations of budesonide occurred within 30 minutes of inhalation from PULMICORT TURBUHALER.
In asthmatic patients, budesonide showed a linear increase in AUC and Cmax with increasing dose after both a single dose and repeated dosing from PULMICORT TURBUHALER.
Distribution: The volume of distribution of budesonide was approximately 3 L/kg. It was 85-90% bound to plasma proteins. Protein binding was constant over the concentration range (1-100 nmol/L) achieved with, and exceeding, recommended doses of PULMICORT TURBUHALER. Budesonide showed little or no binding to corticosteroid binding globulin. Budesonide rapidly equilibrated with red blood cells in a concentration independent manner with a blood/plasma ratio of about 0.8.
Metabolism:In vitro studies with human liver homogenates have shown that budesonide is rapidly and extensively metabolized. Two major metabolites formed via cytochrome P450 (CYP) isoenzyme 3A4 (CYP3A4) catalyzed biotransformation have been isolated and identified as 16α-hydroxyprednisolone and 6β-hydroxybudesonide. The corticosteroid activity of each of these two metabolites is less than 1% of that of the parent compound. No qualitative differences between the in vitro and in vivo metabolic patterns have been detected. Negligible metabolic inactivation was observed in human lung and serum preparations.
Excretion/Elimination: The 22R form of budesonide was preferentially cleared by the liver with systemic clearance of 1.4 L/min vs. 1.0 L/min for the 22S form. The terminal half-life, 2 to 3 hours, was the same for both epimers and was independent of dose. Budesonide was excreted in urine and feces in the form of metabolites. Approximately 60% of an intravenous radiolabelled dose was recovered in the urine. No unchanged budesonide was detected in the urine.
Special Populations: No pharmacokinetic differences have been identified due to race, gender or advanced age.
Pediatric: Following intravenous dosing in pediatric patients age 10-14 years, plasma half-life was shorter than in adults (1.5 hours vs. 2.0 hours in adults). In the same population following inhalation of budesonide via a pressurized metered-dose inhaler, absolute systemic availability was similar to that in adults.
Hepatic Insufficiency: Reduced liver function may affect the elimination of corticosteroids. The pharmacokinetics of budesonide were affected by compromised liver function as evidenced by a doubled systemic availability after oral ingestion. The intravenous pharmacokinetics of budesonide were, however, similar in cirrhotic patients and in healthy subjects.
Drug-Drug Interactions: Ketoconazole, a potent inhibitor of cytochrome P450 (CYP) isoenzyme 3A4 (CYP3A4), the main metabolic enzyme for corticosteroids, increased plasma levels of orally ingested budesonide. At recommended doses, cimetidine had a slight but clinically insignificant effect on the pharmacokinetics of oral budesonide.
Pharmacodynamics
To confirm that systemic absorption is not a significant factor in the clinical efficacy of inhaled budesonide, a clinical study in patients with asthma was performed comparing 400 mcg budesonide administered via a pressurized metered-dose inhaler with a tube spacer to 1400 mcg of oral budesonide and placebo. The study demonstrated the efficacy of inhaled budesonide but not orally ingested budesonide despite comparable systemic levels. Thus, the therapeutic effect of conventional doses of orally inhaled budesonide are largely explained by its direct action on the respiratory tract.
Generally, PULMICORT TURBUHALER has a relatively rapid onset of action for an inhaled corticosteroid. Improvement in asthma control following inhalation of PULMICORT TURBUHALER can occur within 24 hours of beginning treatment although maximum benefit may not be achieved for 1 to 2 weeks, or longer.
PULMICORT TURBUHALER has been shown to decrease airway reactivity in various challenge models, including histamine, methacholine, sodium metabisulfite, and adenosine monophosphate in patients with hyperreactive airways. The clinical relevance of these models is not certain.
Pretreatment with PULMICORT TURBUHALER 1600 mcg daily (800 mcg twice daily) for 2 weeks reduced the acute (early-phase reaction) and delayed (late-phase reaction) decrease in FEV1 following inhaled allergen challenge.
The effects of PULMICORT TURBUHALER on the hypothalamic-pituitary-adrenal (HPA) axis were studied in 905 adults and 404 pediatric patients with asthma. For most patients, the ability to increase cortisol production in response to stress, as assessed by cosyntropin (ACTH) stimulation test, remained intact with PULMICORT TURBUHALER treatment at recommended doses. For adult patients treated with 100, 200, 400, or 800 mcg twice daily for 12 weeks, 4%, 2%, 6%, and 13% respectively, had an abnormal stimulated cortisol response (peak cortisol <14.5 mcg/dL assessed by liquid chromatography following short-cosyntropin test) as compared with 8% of patients treated with placebo. Similar results were obtained in pediatric patients. In another study in adults, doses of 400, 800 and 1600 mcg budesonide twice daily via PULMICORT TURBUHALER for 6 weeks were examined; 1600 mcg twice daily (twice the maximum recommended dose) resulted in a 27% reduction in stimulated cortisol (6-hour ACTH infusion) while 10 mg prednisone resulted in a 35% reduction. In this study, no patient on PULMICORT TURBUHALER at doses of 400 and 800 mcg twice daily met the criterion for an abnormal stimulated cortisol response (peak cortisol <14.5 mcg/dL assessed by liquid chromatography) following ACTH infusion. An open-label, long-term follow-up of 1133 patients for up to 52 weeks confirmed the minimal effect on the HPA axis (both basal and stimulated plasma cortisol) of PULMICORT TURBUHALER when administered at recommended doses. In patients who had previously been oral steroid-dependent, use of PULMICORT TURBUHALER in recommended doses was associated with higher stimulated cortisol response compared with baseline following 1 year of therapy.
The administration of budesonide via PULMICORT TURBUHALER in doses up to 800 mcg/day (mean daily dose 445 mcg/day) or via a pressurized metered-dose inhaler in doses up to 1200 mcg/day (mean daily dose 620 mcg/day) to 216 pediatric patients (age 3 to 11 years) for 2 to 6 years had no significant effect on statural growth compared with non-corticosteroid therapy in 62 matched control patients. However, the long-term effect of PULMICORT TURBUHALER on growth is not fully known.
CLINICAL TRIALS
The therapeutic efficacy of PULMICORT TURBUHALER has been evaluated in controlled clinical trials involving more than 1300 patients (6 years and older) with asthma of varying disease duration (<1 year to >20 years) and severity.
Double-blind, parallel, placebo-controlled clinical trials of 12 weeks duration and longer have shown that, compared with placebo, PULMICORT TURBUHALER significantly improved lung function (measured by PEF and FEV1), significantly decreased morning and evening symptoms of asthma, and significantly reduced the need for as-needed inhaled β2-agonist use at doses of 400 mcg to 1600 mcg per day (200 mcg to 800 mcg twice daily) in adults and 400 mcg to 800 mcg per day (200 mcg to 400 mcg twice daily) in pediatric patients 6 years of age and older.
Improved lung function (morning PEF) was observed within 24 hours of initiating treatment in both adult and pediatric patients 6 years of age and older, although maximum benefit was not achieved for 1 to 2 weeks, or longer, after starting treatment. Improved lung function was maintained throughout the 12 weeks of the double-blind portion of the trials.
Patients Not Receiving Corticosteroid Therapy
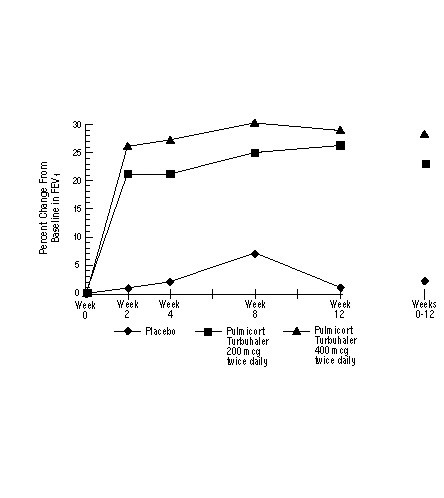
In a 12-week clinical trial in 273 patients with mild to moderate asthma (mean baseline FEV1 2.27 L) who were not well controlled by bronchodilators alone, PULMICORT TURBUHALER was evaluated at doses of 200 mcg twice daily and 400 mcg twice daily versus placebo. The FEV1 results from this trial are shown in the figure below. Pulmonary function improved significantly on both doses of PULMICORT TURBUHALER compared with placebo.
A 12-Week Trial in Patients Not on Corticosteroid Therapy Prior to Study Entry
In a 12-month controlled trial in 75 patients not previously receiving corticosteroids, PULMICORT TURBUHALER at 200 mcg twice daily resulted in improved lung function (measured by PEF) and reduced bronchial hyperreactivity compared with placebo.
Patients Previously Maintained on Inhaled Corticosteroids
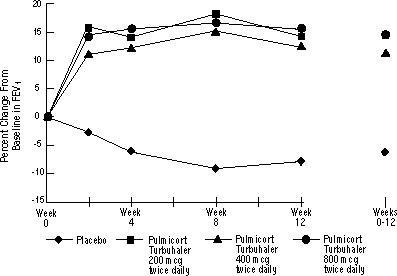
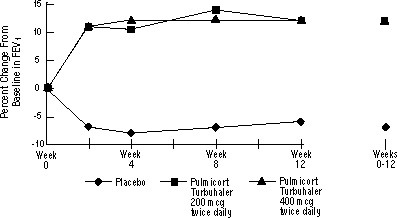
The safety and efficacy of PULMICORT TURBUHALER was also evaluated in adult and pediatric patients (age 6 to 18 years) previously maintained on inhaled corticosteroids (adults: N=473, mean baseline FEV1 2.04 L, baseline doses of beclomethasone dipropionate 126-1008 mcg/day; pediatrics: N=404, mean baseline FEV1 2.09 L, baseline doses of beclomethasone dipropionate 126-672 mcg/day or triamcinolone acetonide 300-1800 mcg/day). The FEV1 results of these two trials, both 12 weeks in duration, are presented in the following figures. Pulmonary function improved significantly with all doses of PULMICORT TURBUHALER compared with placebo in both trials.
Adult Patients Previously Maintained on Inhaled Corticosteroids
Pediatric Patients Age 6 to 18 Years Previously Maintained on Inhaled Corticosteroids
Patients Receiving PULMICORT TURBUHALER Once Daily
The efficacy and safety of once-daily administration of PULMICORT TURBUHALER 200 mcg and 400 mcg and placebo were also evaluated in 309 adult asthmatic patients (mean baseline FEV1 2.7 L) in an 18-week study. Compared with placebo, patients receiving Pulmicort 200 or 400 mcg once daily showed significantly better asthma stability as assessed by PEF and FEV1 over an initial 6-week treatment period, which was maintained with a 200 mcg daily dose over the subsequent 12 weeks. Although the study population included both patients previously treated with inhaled corticosteroids, as well as patients not previously receiving corticosteroid therapy, the results showed that once-daily dosing was most clearly effective for those patients previously maintained on orally inhaled corticosteroids (see DOSAGE AND ADMINISTRATION).
Patients Previously Maintained on Oral Corticosteroids
In a clinical trial in 159 severe asthmatic patients requiring chronic oral prednisone therapy (mean baseline prednisone dose 19.3 mg/day) PULMICORT TURBUHALER at doses of 400 mcg twice daily and 800 mcg twice daily was compared with placebo over a 20-week period. Approximately two-thirds (68% on 400 mcg twice daily and 64% on 800 mcg twice daily) of PULMICORT TURBUHALER-treated patients were able to achieve sustained (at least 2 weeks) oral corticosteroid cessation (compared with 8% of placebo-treated patients) and improved asthma control. The average oral corticosteroid dose was reduced by 83% on 400 mcg twice daily and 79% on 800 mcg twice daily for PULMICORT TURBUHALER-treated patients vs. 27% for placebo. Additionally, 58 out of 64 patients (91%) who completely eliminated oral corticosteroids during the double-blind phase of the trial remained off oral corticosteroids for an additional 12 months while receiving PULMICORT TURBUHALER.
INDICATIONS AND USAGE
PULMICORT TURBUHALER is indicated for the maintenance treatment of asthma as prophylactic therapy in adult and pediatric patients six years of age or older. It is also indicated for patients requiring oral corticosteroid therapy for asthma. Many of those patients may be able to reduce or eliminate their requirement for oral corticosteroids over time.
PULMICORT TURBUHALER is NOT indicated for the relief of acute bronchospasm.
CONTRAINDICATIONS
PULMICORT TURBUHALER is contraindicated in the primary treatment of status asthmaticus or other acute episodes of asthma where intensive measures are required.
Hypersensitivity to budesonide contraindicates the use of PULMICORT TURBUHALER.
WARNINGS
Particular care is needed for patients who are transferred from systemically active corticosteroids to PULMICORT TURBUHALER because deaths due to adrenal insufficiency have occurred in asthmatic patients during and after transfer from systemic corticosteroids to less systemically available inhaled corticosteroids. After withdrawal from systemic corticosteroids, a number of months are required for recovery of hypothalamic-pituitary-adrenal (HPA) function.
Patients who have been previously maintained on 20 mg or more per day of prednisone (or its equivalent) may be most susceptible, particularly when their systemic corticosteroids have been almost completely withdrawn. During this period of HPA suppression, patients may exhibit signs and symptoms of adrenal insufficiency when exposed to trauma, surgery, or infection (particularly gastroenteritis) or other conditions associated with severe electrolyte loss. Although PULMICORT TURBUHALER may provide control of asthma symptoms during these episodes, in recommended doses it supplies less than normal physiological amounts of glucocorticoid systemically and does NOT provide the mineralocorticoid activity that is necessary for coping with these emergencies.
During periods of stress or a severe asthma attack, patients who have been withdrawn from systemic corticosteroids should be instructed to resume oral corticosteroids (in large doses) immediately and to contact their physicians for further instruction. These patients should also be instructed to carry a medical identification card indicating that they may need supplementary systemic corticosteroids during periods of stress or a severe asthma attack.
Patients requiring oral corticosteroids should be weaned slowly from systemic corticosteroid use after transferring to PULMICORT TURBUHALER. Lung function (FEV1 or AM PEF), beta-agonist use, and asthma symptoms should be carefully monitored during withdrawal of oral corticosteroids. In addition to monitoring asthma signs and symptoms, patients should be observed for signs and symptoms of adrenal insufficiency such as fatigue, lassitude, weakness, nausea and vomiting, and hypotension.
Transfer of patients from systemic corticosteroid therapy to PULMICORT TURBUHALER may unmask allergic conditions previously suppressed by the systemic corticosteroid therapy, eg, rhinitis, conjunctivitis, arthritis, eosinophilic conditions and eczema (see DOSAGE AND ADMINISTRATION).
Patients who are on drugs that suppress the immune system are more susceptible to infection than healthy individuals. Chicken pox and measles, for example, can have a more serious or even fatal course in susceptible pediatric patients or adults on immunosuppressant doses of corticosteroids. In pediatric or adult patients who have not had these diseases, particular care should be taken to avoid exposure. How the dose, route, and duration of corticosteroid administration affects the risk of developing a disseminated infection is not known. The contribution of the underlying disease and/or prior corticosteroid treatment to the risk is also not known. If exposed, therapy with varicella zoster immune globulin (VZIG) or pooled intravenous immunoglobulin (IVIG), as appropriate, may be indicated. If exposed to measles, prophylaxis with pooled intramuscular immunoglobulin (IG) may be indicated. (See the respective package inserts for complete VZIG and IG prescribing information.) If chicken pox develops, treatment with antiviral agents may be considered.
PULMICORT TURBUHALER is not a bronchodilator and is not indicated for rapid relief of bronchospasm or other acute episodes of asthma.
As with other inhaled asthma medications, bronchospasm, with an immediate increase in wheezing, may occur after dosing. If bronchospasm occurs following dosing with PULMICORT TURBUHALER, it should be treated immediately with a fast-acting inhaled bronchodilator. Treatment with PULMICORT TURBUHALER should be discontinued and alternate therapy instituted.
Patients should be instructed to contact their physician immediately when episodes of asthma not responsive to their usual doses of bronchodilators occur during treatment with PULMICORT TURBUHALER. During such episodes, patients may require therapy with oral corticosteroids.
PRECAUTIONS
General
During withdrawal from oral corticosteroids, some patients may experience symptoms of systemically active corticosteroid withdrawal, eg, joint and/or muscular pain, lassitude, and depression, despite maintenance or even improvement of respiratory function (see DOSAGE AND ADMINISTRATION).
In responsive patients, PULMICORT TURBUHALER may permit control of asthma symptoms with less suppression of HPA-axis function than therapeutically equivalent oral doses of prednisone. Since budesonide is absorbed into the circulation and can be systemically active, the beneficial effects of PULMICORT TURBUHALER in minimizing HPA dysfunction may be expected only when recommended dosages are not exceeded and individual patients are titrated to the lowest effective dose. Since individual sensitivity to effects on cortisol production exists, physicians should consider this information when prescribing PULMICORT TURBUHALER.
Because of the possibility of systemic absorption of inhaled corticosteroids, patients treated with PULMICORT TURBUHALER should be observed carefully for any evidence of systemic corticosteroid effects. Particular care should be taken in observing patients postoperatively or during periods of stress for evidence of inadequate adrenal response.
It is possible that systemic corticosteroid effects such as hypercorticism, reduced bone mineral density, and adrenal suppression may appear in a small number of patients, particularly at higher doses. If such changes occur, PULMICORT TURBUHALER should be reduced slowly, consistent with accepted procedures for management of asthma symptoms and for tapering of systemic steroids.
Orally inhaled corticosteroids, including budesonide, may cause a reduction in growth velocity when administered to pediatric patients. A reduction in growth velocity may occur as a result of inadequate control of asthma or from use of corticosteroids for treatment. The potential effects of prolonged treatment on growth velocity should be weighed against the clinical benefits obtained and the risks associated with alternative therapies. To minimize the systemic effects of orally inhaled corticosteroids, including PULMICORT TURBUHALER, each patient should be titrated to his/her lowest effective dose (see PRECAUTIONS, Pediatric Use).
Although patients in clinical trials have received PULMICORT TURBUHALER on a continuous basis for periods of 1 to 2 years, the long-term local and systemic effects of PULMICORT TURBUHALER in human subjects are not completely known. In particular, the effects resulting from chronic use of PULMICORT TURBUHALER on developmental or immunological processes in the mouth, pharynx, trachea, and lung are unknown.
In clinical trials with PULMICORT TURBUHALER, localized infections with Candida albicans occurred in the mouth and pharynx in some patients. These infections may require treatment with appropriate antifungal therapy and/or discontinuance of treatment with PULMICORT TURBUHALER.
Inhaled corticosteroids should be used with caution, if at all, in patients with active or quiescent tuberculosis infection of the respiratory tract, untreated systemic fungal, bacterial, viral or parasitic infections, or ocular herpes simplex.
Rare instances of glaucoma, increased intraocular pressure, and cataracts have been reported following the inhaled administration of corticosteroids.
Information for Patients
Patients being treated with PULMICORT TURBUHALER should receive the following information and instructions. This information is intended to aid the patient in the safe and effective use of the medication. It is not a disclosure of all possible adverse or intended effects. For proper use of PULMICORT TURBUHALER and to attain maximum improvement, the patient should read and follow the accompanying Patient’s Instructions for Use carefully.
- Patients should use PULMICORT TURBUHALER at regular intervals as directed since its effectiveness depends on regular use. The patient should not alter the prescribed dosage unless advised to do so by the physician.
- •
- Patients should be advised that PULMICORT TURBUHALER is not a bronchodilator and is not intended to treat acute or life-threatening episodes of asthma.
- •
- Patients should be advised that the effectiveness of PULMICORT TURBUHALER depends on proper use of the device and inhalation-administering technique:
- PULMICORT TURBUHALER must be in the upright position (mouthpiece on top) during loading in order to provide the correct dose.
- PULMICORT TURBUHALER must be primed when the unit is used for the very first time. To prime the unit, it must be held in an upright position and the brown grip turned fully to the right, then turned fully to the left until it clicks. Repeat.
- To load the first dose, the grip must be turned fully to the right and fully to the left until it clicks.
- After the first dose, it is not necessary to prime the unit. However, it must be loaded in the upright position immediately prior to use as described above.
- Patients should be advised not to shake the inhaler.
- Patients should place the mouthpiece between the lips and inhale forcefully and deeply. The powder is then delivered to the lungs.
- •
- Patients should not exhale through PULMICORT TURBUHALER.
- •
- Due to the small volume of powder, the patient may not taste or sense the presence of any medication entering the lungs when inhaling from the TURBUHALER inhaler. This lack of sensation does not indicate that the patient is not receiving benefit from PULMICORT TURBUHALER.
- •
- Patients should be advised that rinsing the mouth with water without swallowing after each dosing may decrease the risk of the development of oral candidiasis.
- •
- Patients should be instructed that they will receive a new PULMICORT TURBUHALER unit each time they refill their prescription. Patients should be advised to discard the whole device after the labelled number of inhalations has been used. When there are 20 doses remaining in PULMICORT TURBUHALER, a red mark will appear in the indicator window.
- •
- PULMICORT TURBUHALER should not be used with a spacer.
- •
- The mouthpiece should not be bitten or chewed.
- •
- The cover should be replaced securely after each opening.
- •
- Patients should keep PULMICORT TURBUHALER clean and dry at all times.
- •
- Patients should be advised that improvement in asthma control following inhalation of PULMICORT TURBUHALER can occur within 24 hours of beginning treatment although maximum benefit may not be achieved for 1 to 2 weeks, or longer. If symptoms do not improve in that time frame, or if the condition worsens, the patient should be instructed not to increase the dosage, but to contact the physician.
- Patients whose systemic corticosteroids have been reduced or withdrawn should be instructed to carry a warning card indicating that they may need supplemental systemic corticosteroids during periods of stress or an asthma attack that does not respond to bronchodilators.
- Patients should be advised not to stop the use of PULMICORT TURBUHALER abruptly.
- •
- Patients should be warned to avoid exposure to chicken pox or measles and if they are exposed, to consult their physicians without delay.
- •
- Long-term use of inhaled corticosteroids, including budesonide, may increase the risk of some eye problems (cataracts or glaucoma). Regular eye examinations should be considered.
- •
- Women considering the use of PULMICORT TURBUHALER should consult with their physician if they are pregnant or intend to become pregnant, or if they are breast-feeding a baby.
- •
- Patients considering use of PULMICORT TURBUHALER should consult with their physician if they are allergic to budesonide or any other orally inhaled corticosteroid.
- •
- Patients should inform their physician of other medications they are taking as PULMICORT TURBUHALER may not be suitable in some circumstances and the physician may wish to use a different medicine.
Drug Interactions
In clinical studies, concurrent administration of budesonide and other drugs commonly used in the treatment of asthma has not resulted in an increased frequency of adverse events. The main route of metabolism of budesonide, as well as other corticosteroids, is via cytochrome P450 (CYP) isoenzyme 3A4 (CYP3A4). After oral administration of ketoconazole, a potent inhibitor of CYP3A4, the mean plasma concentration of orally administered budesonide increased. Concomitant administration of other known inhibitors of CYP3A4 (eg, itraconazole, clarithromycin, erythromycin, etc.) may inhibit the metabolism of, and increase the systemic exposure to, budesonide. Care should be exercised when budesonide is coadministered with long-term ketoconazole and other known CYP3A4 inhibitors.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies were conducted in rats and mice using oral administration to evaluate the carcinogenic potential of budesonide.
In a 104-week oral study in Sprague-Dawley rats, a statistically significant increase in the incidence of gliomas was observed in male rats receiving an oral dose of 50 mcg/kg/day (less than the maximum recommended daily inhalation dose in adults and children on a mcg/m2 basis). No tumorigenicity was seen in male and female rats at respective oral doses up to 25 and 50 mcg/kg (less than the maximum recommended daily inhalation dose in adults and children on a mcg/m2 basis). In two additional two-year studies in male Fischer and Sprague-Dawley rats, budesonide caused no gliomas at an oral dose of 50 mcg/kg (less than the maximum recommended daily inhalation dose in adults and children on a mcg/m2 basis). However, in the male Sprague-Dawley rats, budesonide caused a statistically significant increase in the incidence of hepatocellular tumors at an oral dose of 50 mcg/kg (less than the maximum recommended daily inhalation dose in adults and children on a mcg/m2 basis). The concurrent reference corticosteroids (prednisone and triamcinolone acetonide) in these two studies showed similar findings.
There was no evidence of a carcinogenic effect when budesonide was administered orally for 91 weeks to mice at doses up to 200 mcg/kg/day (less than the maximum recommended daily inhalation dose in adults and children on a mcg/m2 basis).
Budesonide was not mutagenic or clastogenic in six different test systems: Ames Salmonella/microsome plate test, mouse micronucleus test, mouse lymphoma test, chromosome aberration test in human lymphocytes, sex-linked recessive lethal test in Drosophila melanogaster, and DNA repair analysis in rat hepatocyte culture.
In rats, budesonide had no effect on fertility at subcutaneous doses up to 80 mcg/kg (less than the maximum recommended human daily inhalation dose on a mcg/m2 basis).
At 20 mcg/kg/day (less than the maximum recommended human daily inhalation dose on a mcg/m2 basis), decreases in maternal body weight gain, prenatal viability, and viability of the young at birth and during lactation were observed. No such effects were noted at 5 mcg/kg (less than the maximum recommended human daily inhalation dose in adults on a mcg/m2 basis).
Pregnancy: Teratogenic Effects
Pregnancy Category B: As with other glucocorticoids, budesonide produced fetal loss, decreased pup weight, and skeletal abnormalities at subcutaneous doses of 25 mcg/kg/day in rabbits (less than the maximum recommended human daily inhalation dose on a mcg/m2 basis) and 500 mcg/kg/day in rats (approximately 3 times the maximum recommended human daily inhalation dose on a mcg/m2 basis).
No teratogenic or embryocidal effects were observed in rats when budesonide was administered by inhalation at doses up to 250 mcg/kg/day (equivalent to the maximum recommended human daily inhalation dose on a mcg/m2 basis).
Experience with oral corticosteroids since their introduction in pharmacologic as opposed to physiologic doses suggests that rodents are more prone to teratogenic effects from corticosteroids than humans.
Studies of pregnant women, however, have not shown that PULMICORT TURBUHALER increases the risk of abnormalities when administered during pregnancy. The results from a large population-based prospective cohort epidemiological study reviewing data from three Swedish registries covering approximately 99% of the pregnancies from 1995-1997 (i.e., Swedish Medical Birth Registry; Registry of Congenital Malformations; Child Cardiology Registry) indicate no increased risk for congenital malformations from the use of inhaled budesonide during early pregnancy. Congenital malformations were studied in 2,014 infants born to mothers reporting the use of inhaled budesonide for asthma in early pregnancy (usually 10-12 weeks after the last menstrual period), the period when most major organ malformations occur. The rate of recorded congenital malformations was similar compared with the general population rate (3.8% vs. 3.5%, respectively). In addition, after exposure to inhaled budesonide, the number of infants born with orofacial clefts was similar to the expected number in the normal population (4 children vs. 3.3, respectively).
These same data were utilized in a second study bringing the total to 2,534 infants whose mothers were exposed to inhaled budesonide. In this study, the rate of congenital malformations among infants whose mothers were exposed to inhaled budesonide during early pregnancy was not different from the rate for all newborn babies during the same period (3.6%).
Despite the animal findings, it would appear that the possibility of fetal harm is remote if the drug is used during pregnancy. Nevertheless, because the studies in humans cannot rule out the possibility of harm, PULMICORT TURBUHALER should be used during pregnancy only if clearly needed.
Nonteratogenic Effects
Hypoadrenalism may occur in infants born of mothers receiving corticosteroids during pregnancy. Such infants should be carefully observed.
Nursing Mothers
Corticosteroids are secreted in human milk. Because of the potential for adverse reactions in nursing infants from any corticosteroid, a decision should be made whether to discontinue nursing or discontinue the drug, taking into account the importance of the drug to the mother. Actual data for budesonide are lacking.
Pediatric Use
Safety and effectiveness of PULMICORT TURBUHALER in pediatric patients below 6 years of age have not been established.
In pediatric asthma patients the frequency of adverse events observed with PULMICORT TURBUHALER was similar between the 6- to 12-year age group (N=172) compared with the 13- to 17-year age group (N=124).
Controlled clinical studies have shown that orally inhaled corticosteroids may cause a reduction in growth velocity in pediatric patients. This effect has been observed in the absence of laboratory evidence of hypothalamic-pituitary-adrenal (HPA) axis suppression, suggesting that growth velocity is a more sensitive indicator of systemic corticosteroid exposure in pediatric patients than some commonly used tests of HPA-axis function. The long-term effects of this reduction in growth velocity associated with orally inhaled corticosteroids including the impact on final adult height are unknown. The potential for “catch up” growth following discontinuation of treatment with orally inhaled corticosteroids has not been adequately studied.
In a study of asthmatic children 5-12 years of age, those treated with PULMICORT TURBUHALER 200 mcg twice daily (n=311) had a 1.1-centimeter reduction in growth compared with those receiving placebo (n=418) at the end of one year; the difference between these two treatment groups did not increase further over three years of additional treatment. By the end of four years, children treated with PULMICORT TURBUHALER and children treated with placebo had similar growth velocities. Conclusions drawn from this study may be confounded by the unequal use of corticosteroids in the treatment groups and inclusion of data from patients attaining puberty during the course of the study.
The growth of pediatric patients receiving orally inhaled corticosteroids, including PULMICORT TURBUHALER, should be monitored routinely (eg, via stadiometry). The potential growth effects of prolonged treatment should be weighed against clinical benefits obtained and the risks and benefits associated with alternative therapies. To minimize the systemic effects of inhaled corticosteroids, including PULMICORT TURBUHALER, each patient should be titrated to his/her lowest effective dose.
Geriatric Use
One hundred patients 65 years or older were included in the US and non-US controlled clinical trials of PULMICORT TURBUHALER. There were no differences in the safety and efficacy of the drug compared with those seen in younger patients.
In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
ADVERSE REACTIONS
The following adverse reactions were reported in patients treated with PULMICORT TURBUHALER.
The incidence of common adverse events is based upon double-blind, placebo-controlled US clinical trials in which 1116 adult and pediatric patients age 6-70 years (472 females and 644 males) were treated with PULMICORT TURBUHALER (200 to 800 mcg twice daily for 12 to 20 weeks) or placebo.
The following table shows the incidence of adverse events in patients previously receiving bronchodilators and/or inhaled corticosteroids in US controlled clinical trials. This population included 232 male and 62 female pediatric patients (age 6 to 17 years) and 332 male and 331 female adult patients (age 18 years and greater).
|
Adverse Events with ≥ 3% Incidence reported by Patients on PULMICORT TURBUHALER |
||||
|
PULMICORT TURBUHALER |
||||
|
Adverse Event |
Placebo N=284 % |
200 mcg twice daily N=286 % |
400 mcg twice daily N=289 % |
800 mcg twice daily N=98 % |
|
Respiratory System | ||||
|
Respiratory Infection Pharyngitis Sinusitis Voice alteration |
17 9 7 0 |
20 10 11 1 |
24 9 7 2 |
19 5 2 6 |
|
Body As A Whole | ||||
|
Headache Flu syndrome Pain Back pain Fever |
7 6 2 1 2 |
14 6 5 2 2 |
13 6 5 3 4 |
14 14 5 6 0 |
|
Digestive System | ||||
|
Oral candidiasis Dyspepsia Gastroenteritis Nausea |
2 2 1 2 |
2 1 1 2 |
4 2 2 1 |
4 4 3 3 |
|
Average Duration of Exposure (days) |
59 |
79 |
80 |
80 |
The table above includes all events (whether considered drug-related or non-drug-related by the investigators) that occurred at a rate of ≥3% in any one PULMICORT TURBUHALER group and were more common than in the placebo group. In considering these data, the increased average duration of exposure for PULMICORT TURBUHALER patients should be taken into account.
The following other adverse events occurred in these clinical trials using PULMICORT TURBUHALER with an incidence of 1 to 3% and were more common on PULMICORT TURBUHALER than on placebo.
Body As A Whole: neck pain
Cardiovascular: syncope
Digestive: abdominal pain, dry mouth, vomiting
Metabolic and Nutritional: weight gain
Musculoskeletal: fracture, myalgia
Nervous: hypertonia, migraine
Platelet, Bleeding and Clotting: ecchymosis
Psychiatric: insomnia
Resistance Mechanisms: infection
Special Senses: taste perversion
In a 20-week trial in adult asthmatics who previously required oral corticosteroids, the effects of PULMICORT TURBUHALER 400 mcg twice daily (N=53) and 800 mcg twice daily (N=53) were compared with placebo (N=53) on the frequency of reported adverse events. Adverse events, whether considered drug-related or non-drug-related by the investigators, reported in more than five patients in the PULMICORT TURBUHALER group and which occurred more frequently with PULMICORT TURBUHALER than placebo are shown below (% PULMICORT TURBUHALER and % placebo). In considering these data, the increased average duration of exposure for PULMICORT TURBUHALER patients (78 days for PULMICORT TURBUHALER vs. 41 days for placebo) should be taken into account.
Body As A Whole:asthenia (9% and 2%)
headache (12% and 2%)
pain (10% and 2%)
Digestive:dyspepsia (8% and 0%)
nausea (6% and 0%)
oral candidiasis (10% and 0%)
Musculoskeletal:arthralgia (6% and 0%)
Respiratory:cough increased (6% and 2%)
respiratory infection (32% and 13%)
rhinitis (6% and 2%)
sinusitis (16% and 11%)
Patients Receiving PULMICORT TURBUHALER Once Daily
The adverse event profile of once-daily administration of PULMICORT TURBUHALER 200 mcg and 400 mcg, and placebo, was evaluated in 309 adult asthmatic patients in an 18-week study. The study population included both patients previously treated with inhaled corticosteroids, and patients not previously receiving corticosteroid therapy. There was no clinically relevant difference in the pattern of adverse events following once-daily administration of PULMICORT TURBUHALER when compared with twice-daily dosing.
Pediatric Studies
In a 12-week placebo-controlled trial in 404 pediatric patients 6 to 18 years of age previously maintained on inhaled corticosteroids, the frequency of adverse events for each age category (6 to 12 years, 13 to 18 years) was comparable for PULMICORT TURBUHALER (at 100, 200 and 400 mcg twice daily) and placebo. There were no clinically relevant differences in the pattern or severity of adverse events in children compared with those reported in adults.
Adverse Event Reports From Other Sources
Rare adverse events reported in the published literature or from worldwide marketing experience with any formulation of inhaled budesonide include: immediate and delayed hypersensitivity reactions including rash, contact dermatitis, urticaria, angioedema and bronchospasm; symptoms of hypocorticism and hypercorticism; glaucoma, cataracts; psychiatric symptoms including depression, aggressive reactions, irritability, anxiety and psychosis.
OVERDOSAGE
The potential for acute toxic effects following overdose of PULMICORT TURBUHALER is low. If used at excessive doses for prolonged periods, systemic corticosteroid effects such as hypercorticism may occur (see PRECAUTIONS). PULMICORT TURBUHALER at twice the highest recommended dose (3200 mcg daily) administered for 6 weeks caused a significant reduction (27%) in the plasma cortisol response to a 6-hour infusion of ACTH compared with placebo (+1%). The corresponding effect of 10 mg prednisone daily was a 35% reduction in the plasma cortisol response to ACTH.
The minimal inhalation lethal dose in mice was 100 mg/kg (approximately 320 times the maximum recommended daily inhalation dose in adults and approximately 380 times the maximum recommended daily inhalation dose in children on a mcg/m2 basis). There were no deaths following the administration of an inhalation dose of 68 mg/kg in rats (approximately 430 times the maximum recommended daily inhalation dose in adults and approximately 510 times the maximum recommended daily inhalation dose in children on a mcg/m2 basis). The minimal oral lethal dose was 200 mg/kg in mice (approximately 630 times the maximum recommended daily inhalation dose in adults and approximately 750 times the maximum recommended daily inhalation dose in children on a mcg/m2 basis) and less than 100 mg/kg in rats (approximately 630 times the maximum recommended daily inhalation dose in adults and approximately 750 times the maximum recommended daily inhalation dose in children based on a mcg/m2 basis).
Post-marketing experience showed that patients experiencing acute overdose of inhaled budesonide commonly remained asymptomatic. The use of excessive doses (up to 6400 mcg daily) for prolonged periods showed systemic corticosteroid effects such as hypercorticism.
DOSAGE AND ADMINISTRATION
PULMICORT TURBUHALER should be administered by the orally inhaled route in asthmatic patients age 6 years and older. Individual patients will experience a variable onset and degree of symptom relief. Generally, PULMICORT TURBUHALER has a relatively rapid onset of action for an inhaled corticosteroid. Improvement in asthma control following inhaled administration of PULMICORT TURBUHALER can occur within 24 hours of initiation of treatment, although maximum benefit may not be achieved for 1 to 2 weeks, or longer. The safety and efficacy of PULMICORT TURBUHALER when administered in excess of recommended doses have not been established.
The recommended starting dose and the highest recommended dose of PULMICORT TURBUHALER, based on prior asthma therapy, are listed in the following table.
| Previous Therapy | Recommended Starting Dose | Highest Recommended Dose | |
|---|---|---|---|
|
Adults: |
Bronchodilators alone |
200 to 400 mcg twice daily |
400 mcg twice daily |
|
Inhaled Corticosteroids* |
200 to 400 mcg twice daily |
800 mcg twice daily |
|
|
Oral Corticosteroids |
400 to 800 mcg twice daily |
800 mcg twice daily |
|
|
Children: |
Bronchodilators alone |
200 mcg twice daily |
400 mcg twice daily |
|
Inhaled Corticosteroids* |
200 mcg twice daily |
400 mcg twice daily |
|
|
Oral Corticosteroids |
The highest recommended dose in children is 400 mcg twice daily |
||
*In patients with mild to moderate asthma who are well controlled on inhaled corticosteroids, dosing with PULMICORT TURBUHALER 200 mcg or 400 mcg once daily may be considered. PULMICORT TURBUHALER can be administered once daily either in the morning or in the evening.
If the once-daily treatment with PULMICORT TURBUHALER does not provide adequate control of asthma symptoms, the total daily dose should be increased and/or administered as a divided dose.
Patients Maintained on Chronic Oral Corticosteroids
Initially, PULMICORT TURBUHALER should be used concurrently with the patient’s usual maintenance dose of systemic corticosteroid. After approximately one week, gradual withdrawal of the systemic corticosteroid is started by reducing the daily or alternate daily dose. The next reduction is made after an interval of one or two weeks, depending on the response of the patient. Generally, these decrements should not exceed 2.5 mg of prednisone or its equivalent. A slow rate of withdrawal is strongly recommended. During reduction of oral corticosteroids, patients should be carefully monitored for asthma instability, including objective measures of airway function, and for adrenal insufficiency (see WARNINGS). During withdrawal, some patients may experience symptoms of systemic corticosteroid withdrawal, eg, joint and/or muscular pain, lassitude, and depression, despite maintenance or even improvement in pulmonary function. Such patients should be encouraged to continue with PULMICORT TURBUHALER but should be monitored for objective signs of adrenal insufficiency. If evidence of adrenal insufficiency occurs, the systemic corticosteroid doses should be increased temporarily and thereafter withdrawal should continue more slowly. During periods of stress or a severe asthma attack, transfer patients may require supplementary treatment with systemic corticosteroids.
NOTE: In all patients it is desirable to titrate to the lowest effective dose once asthma stability is achieved.
Directions for Use
Illustrated Patient's Instructions for Use accompany each package of PULMICORT TURBUHALER.
Patients should be instructed to prime PULMICORT TURBUHALER prior to its initial use, and instructed to inhale deeply and forcefully each time the unit is used. Rinsing the mouth after inhalation is also recommended.
HOW SUPPLIED
PULMICORT TURBUHALER consists of a number of assembled plastic details, the main parts being the dosing mechanism, the storage unit for drug substance and the mouthpiece. The inhaler is protected by a white outer tubular cover screwed onto the inhaler. The body of the inhaler is white and the turning grip is brown. The following wording is printed on the grip in raised lettering, “Pulmicort™ 200 mcg”. The TURBUHALER inhaler cannot be refilled and should be discarded when empty.
PULMICORT TURBUHALER is available as 200 mcg/dose, 200 doses (NDC 0186-0915-42) and has a target fill weight of 104 mg.
When there are 20 doses remaining in PULMICORT TURBUHALER, a red mark will appear in the indicator window. If the unit is used beyond the point at which the red mark appears at the bottom of the window, the correct amount of medication may not be obtained. The unit should be discarded.
Store with the cover tightened in a dry place at controlled room temperature 20-25°C (68-77°F) [see USP]. Keep out of the reach of children.
All trademarks are the property of the AstraZeneca group of companies.
©AstraZeneca 2001, 2006
Manufactured for: AstraZeneca LP, Wilmington, DE 19850
By: AstraZeneca AB, Södertälje, Sweden
33020–00
Rev. 10/06
| PULMICORT TURBUHALER
budesonide powder, metered |
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
| Labeler - AstraZeneca Pharmaceuticals LP (938368834) |
| Registrant - AstraZeneca PLC (230790719) |