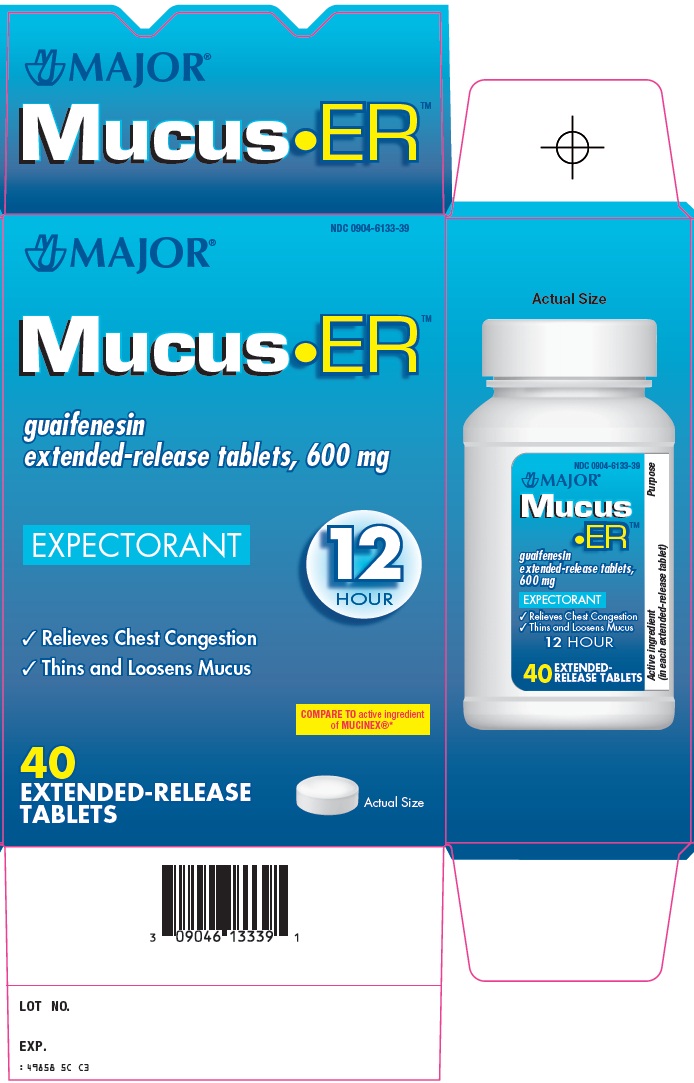
MUCUS ER- guaifenesin tablet, extended release
Major Pharmaceuticals
----------
Major Pharmaceuticals Mucus ER™ Drug Facts
Uses
helps loosen phlegm (mucus) and thin bronchial secretions to rid the bronchial passageways of bothersome mucus and make coughs more productive
Warnings
Ask a doctor before use if you have
- •
- persistent or chronic cough such as occurs with smoking, asthma, chronic bronchitis, or emphysema
- •
- cough accompanied by too much phlegm (mucus)
Directions
- •
- do not crush, chew, or break tablet
- •
- take with a full glass of water
- •
- this product can be administered without regard for the timing of meals
- •
- adults and children 12 years of age and over: one or two tablets every 12 hours. Do not exceed 4 tablets in 24 hours.
- •
- children under 12 years of age: do not use
Other information
- •
- do not use if printed seal under cap is broken or missing
- •
- store between 20-25°C (68-77°F)
| MUCUS ER
guaifenesin tablet, extended release |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Labeler - Major Pharmaceuticals (191427277) |
Revised: 11/2017
Document Id: 79010646-0fb0-43cc-964d-4fd5a6286185
Set id: 36e1d697-daf6-43be-883d-57422eb07eab
Version: 2
Effective Time: 20171125
Major Pharmaceuticals