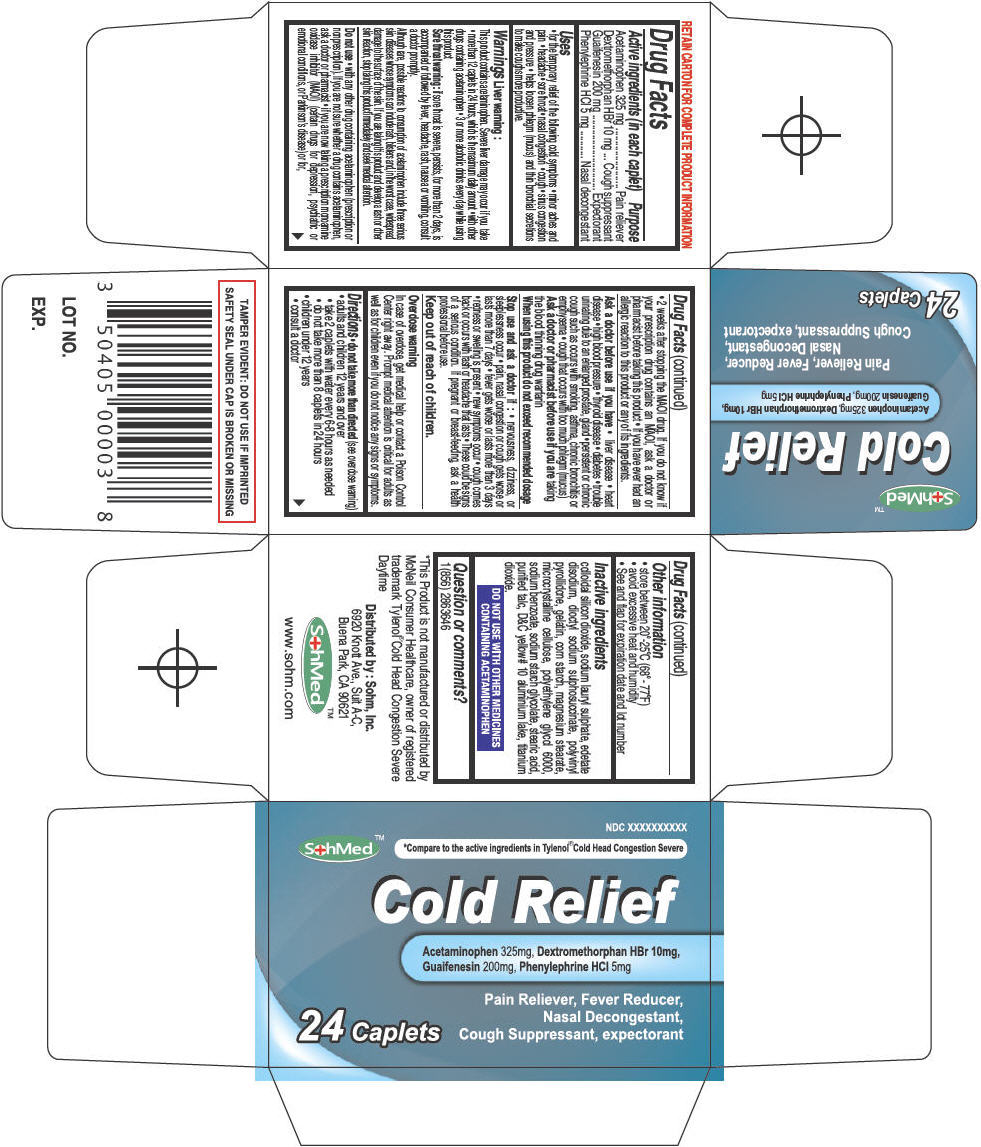
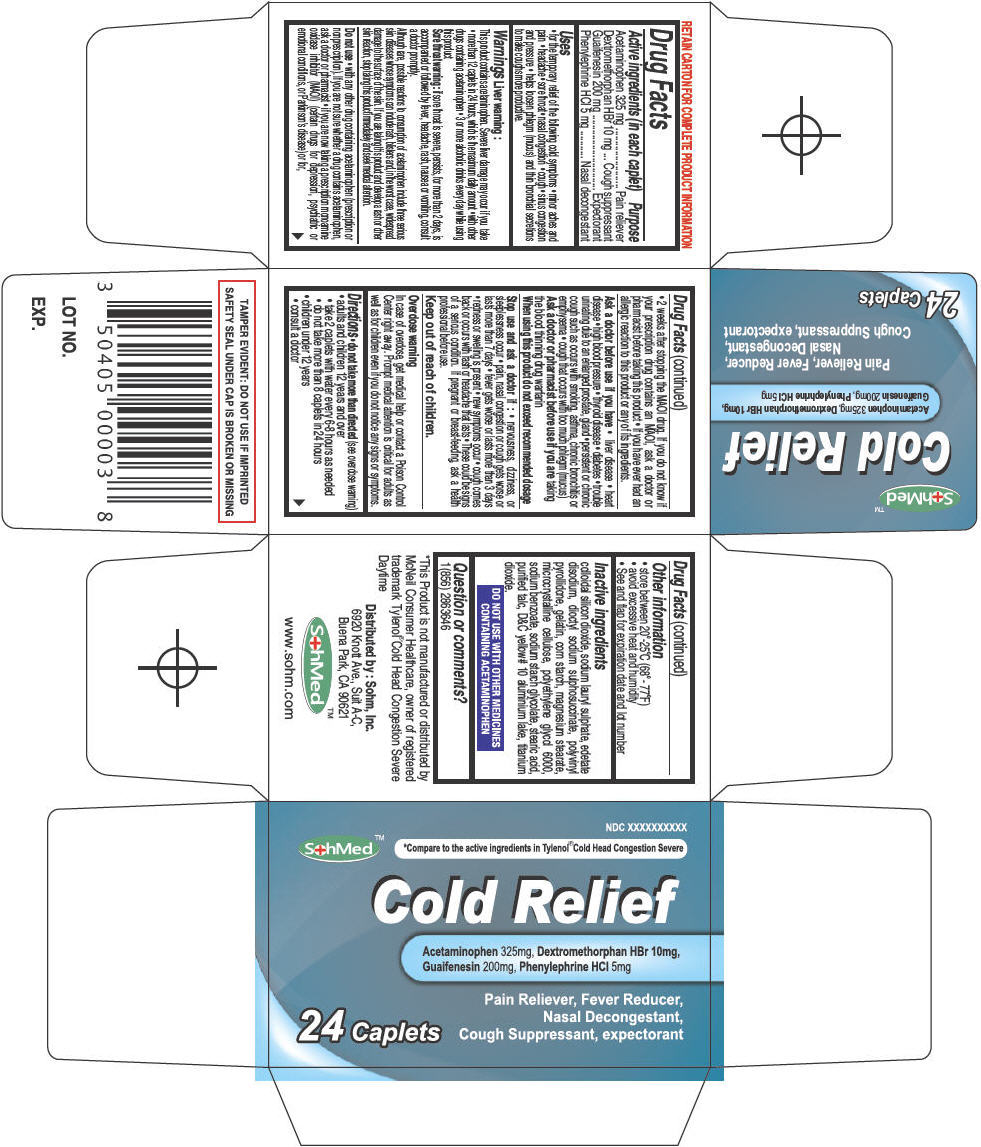
Label: SOHMED COLD RELIEF- acetaminophen, dextromethorphan hydrobromide, guaifenesin, and phenylephrine hydrochloride tablet
-
Contains inactivated NDC Code(s)
NDC Code(s): 50405-006-24 - Packager: SOHM Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated August 20, 2013
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
- Uses
-
Warnings
Liver warning
This product contains acetaminophen. Severe liver damage may occur if you take
- more than 12 caplets in 24 hours, which is the maximum daily amount
- with other drugs containing acetaminophen
- 3 or more alcoholic drinks every day while using this product
Sore throat warning : if sore throat is severe, persists, for more than 2 days, is accompanied or followed by fever, headache, rash, nausea or vomiting, consult a doctor promptly.
Although rare, possible reactions to consumption of acetaminophen include three serious skin diseases whose symptoms can include rash, blisters and, in the worst case, widespread damage to the surface of the skin. If you are taking this product and develop a rash or other skin reaction, stop taking this product immediately and seek medical attention.
Do not use
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson's disease) or for,
- 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product
- If you have ever had an allergic reaction to this product or any of its ingredients.
Ask a doctor before use if you have
- liver disease
- heart disease
- high blood pressure
- thyroid disease
- diabetes
- trouble urinating due to an enlarged prostate, gland
- persistent or chronic cough such as occurs with smoking, asthma, chronic bronchitis or emphysema
- cough that occurs with too much phlegm (mucus)
Stop use and ask a doctor if
- nervousness, dizziness, or sleeplessness occur
- pain, nasal congestion or cough gets worse or lasts more than 7 days
- fever gets worse or lasts more than 3 days
- redness or swelling is present
- new symptoms occur
- cough comes back or occurs with rash or headache that lasts
- These could be signs of a serious condition.
-
Directions
- do not take more than directed (see overdose warning)
- adults and children 12 years and over
- take 2 caplets with water every 6-8 hours as needed
- do not take more than 8 caplets in 24 hours
- children under 12 years
- consult a doctor
- Other information
-
Inactive ingredients
colloidal silicon dioxide, sodium lauryl sulphate, edetate disodium, dioctyl sodium sulphosuccinate, polyvinyl pyrollidone, gelatin, corn starch, magnesium stearate, microcrystalline cellulose, polyethylene glycol 6000, sodium benzoate, sodium starch glycolate, stearic acid, purified talc, D&C yellow# 10 aluminium lake, titanium dioxide.
DO NOT USE WITH OTHER MEDICINES CONTAINING ACETAMINOPHEN
- Question or comments?
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 24 Tablet Bottle Carton
-
INGREDIENTS AND APPEARANCE
SOHMED COLD RELIEF
acetaminophen, dextromethorphan hydrobromide, guaifenesin, and phenylephrine hydrochloride tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:50405-006 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Acetaminophen (UNII: 362O9ITL9D) (Acetaminophen - UNII:362O9ITL9D) Acetaminophen 325 mg Dextromethorphan Hydrobromide (UNII: 9D2RTI9KYH) (Dextromethorphan - UNII:7355X3ROTS) Dextromethorphan Hydrobromide 10 mg Guaifenesin (UNII: 495W7451VQ) (Guaifenesin - UNII:495W7451VQ) Guaifenesin 200 mg Phenylephrine Hydrochloride (UNII: 04JA59TNSJ) (Phenylephrine - UNII:1WS297W6MV) Phenylephrine Hydrochloride 5 mg Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM LAURYL SULFATE (UNII: 368GB5141J) EDETATE DISODIUM (UNII: 7FLD91C86K) DOCUSATE SODIUM (UNII: F05Q2T2JA0) STARCH, CORN (UNII: O8232NY3SJ) POVIDONE K90 (UNII: RDH86HJV5Z) GELATIN (UNII: 2G86QN327L) MAGNESIUM STEARATE (UNII: 70097M6I30) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) POLYETHYLENE GLYCOL 6000 (UNII: 30IQX730WE) SODIUM BENZOATE (UNII: OJ245FE5EU) SODIUM STARCH GLYCOLATE TYPE A CORN (UNII: AG9B65PV6B) STEARIC ACID (UNII: 4ELV7Z65AP) TALC (UNII: 7SEV7J4R1U) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color YELLOW Score 2 pieces Shape OVAL Size 18mm Flavor Imprint Code CRX Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:50405-006-24 1 in 1 CARTON 1 24 in 1 BOTTLE, PLASTIC Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part341 08/25/2013 Labeler - SOHM Inc. (009303848)