POTASSIUM CHLORIDE- potassium chloride solution
Qualitest Pharmaceuticals
Disclaimer: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
----------
POTASSIUM CHLORIDE ORAL SOLUTION, USP
10% AND 20%
Rx only
DESCRIPTION
Potassium Chloride Oral Solution, USP 10% Sugar Free, Dye Free, Alcohol Free – Each 15 mL (tablespoonful) contains: 20 mEq of potassium chloride (provided by potassium chloride 1.5 g), in a palatable, cherry flavored, sugar free, alcohol free vehicle.
Inactive Ingredients: Flavor, glycerin, methylparaben, propylene glycol, propylparaben, purified water, saccharin sodium, sodium citrate.
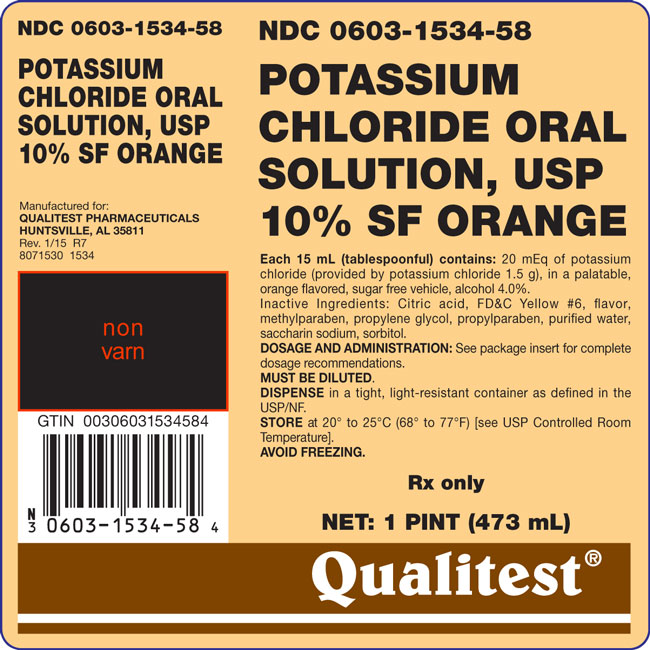
Potassium Chloride Oral Solution, USP 10% Sugar Free Orange – Each 15 mL (tablespoonful) contains: 20 mEq of potassium chloride (provided by potassium chloride 1.5 g), in a palatable, orange flavored, sugar free vehicle, alcohol 4.0%.
Inactive Ingredients: Citric acid, FD&C Yellow #6, flavor, methylparaben, propylene glycol, propylparaben, purified water, saccharin sodium, sorbitol.
Potassium Chloride Oral Solution, USP 10% Yellow – Each 15 mL (tablespoonful) contains: 20 mEq of potassium chloride (provided by potassium chloride 1.5 g), in a palatable lemon flavored vehicle. It also contains sugar and 4.0% alcohol.
Inactive Ingredients: Citric acid, D&C Yellow #10, flavor, methylparaben, propylene glycol, propylparaben, purified water, sucrose.
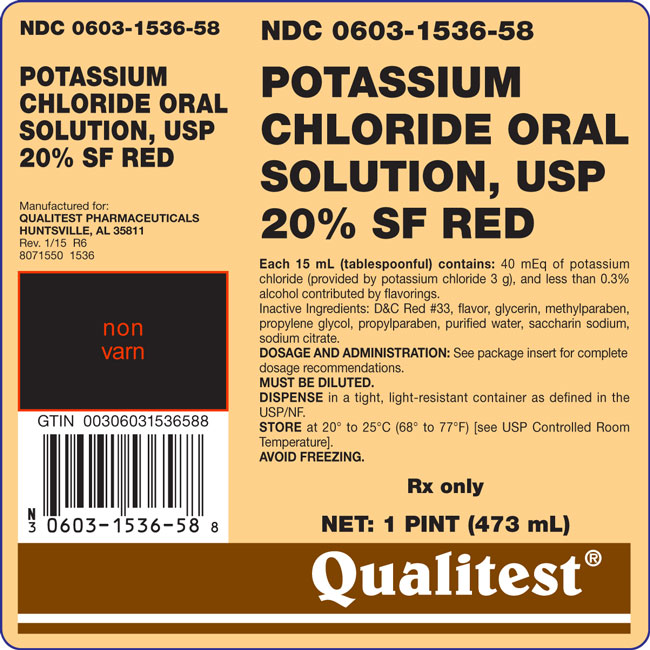
Potassium Chloride Oral Solution, USP 20% Sugar Free Red – Each 15 mL (tablespoonful) contains: 40 mEq of potassium chloride (provided by potassium chloride 3 g), and less than 0.3% alcohol contributed by flavorings.
Inactive Ingredients: D&C Red #33, flavor, glycerin, methylparaben, propylene glycol, propylparaben, purified water, saccharin sodium, sodium citrate.
Potassium chloride oral solution 10% and 20% are electrolyte replenishers. The chemical name is potassium chloride and the structural formula is KCI. Potassium chloride, USP occurs as a white, granular powder or as colorless crystals. It is odorless and has a saline taste. Its solutions are neutral to litmus. It is freely soluble in water and insoluble in alcohol.
CLINICAL PHARMACOLOGY
The potassium ion is the principal intracellular cation of most body tissues. Potassium ions participate in a number of essential physiological processes including the maintenance of intracellular tonicity, the transmission of nerve impulses, the contraction of cardiac, skeletal and smooth muscle and the maintenance of normal renal function.
The intracellular concentration of potassium is approximately 150 to 160 mEq per liter. The normal adult plasma concentration is 3.5 to 5 mEq per liter. An active ion transport system maintains this gradient across the plasma membrane.
Potassium is a normal dietary constituent and under steady state conditions the amount of potassium absorbed from the gastrointestinal tract is equal to the amount excreted in the urine. The usual dietary intake of potassium is 50 to 100 mEq per day.
Potassium depletion will occur whenever the rate of potassium loss through renal excretion and/or loss from the gastrointestinal tract exceeds the rate of potassium intake. Such depletion usually develops as a consequence of therapy with diuretics, primary or secondary hyperaldosteronism, diabetic ketoacidosis, or inadequate replacement of potassium in patients on prolonged parenteral nutrition. Depletion can develop rapidly with severe diarrhea, especially if associated with vomiting. Potassium depletion due to these causes is usually accompanied by a concomitant loss of chloride and is manifested by hypokalemia and metabolic alkalosis. Potassium depletion may produce weakness, fatigue, disturbances of cardiac rhythm (primarily ectopic beats), prominent U-waves in the electrocardiogram, and, in advanced cases, flaccid paralysis and/or impaired ability to concentrate urine.
If potassium depletion associated with metabolic alkalosis cannot be managed by correcting the fundamental cause of the deficiency, e.g., where the patient requires long term diuretic therapy, supplemental potassium in the form of high potassium food or potassium chloride may be able to restore normal potassium levels.
In rare circumstances (e.g., patients with renal tubular acidosis) potassium depletion may be associated with metabolic acidosis and hyperchloremia. In such patients potassium replacement should be accomplished with potassium salts other than chloride, such as potassium bicarbonate, potassium citrate, potassium acetate, or potassium gluconate.
INDICATIONS AND USAGE
- For the treatment of patients with hypokalemia, with or without metabolic alkalosis; in digitalis intoxication; and in patients with hypokalemic familial periodic paralysis. If hypokalemia is the result of diuretic therapy, consideration should be given to the use of a lower dose of diuretic, which may be sufficient without leading to hypokalemia.
- For the prevention of hypokalemia in patients who would be at particular risk if hypokalemia were to develop, e.g., digitalized patients or patients with significant cardiac arrhythmias.
The use of potassium salts in patients receiving diuretics for uncomplicated essential hypertension is often unnecessary when such patients have a normal dietary pattern and when low doses of the diuretic are used. Serum potassium should be checked periodically, however, and if hypokalemia occurs, dietary supplementation with potassium-containing foods may be adequate to control milder cases. In more severe cases, and if dose adjustment of the diuretic is ineffective or unwarranted, supplementation with potassium salts may be indicated.
CONTRAINDICATIONS
Potassium supplements are contraindicated in patients with hyperkalemia since further increase in serum potassium concentration in such patients can produce cardiac arrest. Hyperkalemia may complicate any of the following conditions: chronic renal failure, systemic acidosis such as diabetic acidosis, acute dehydration, extensive tissue breakdown as in severe burns, adrenal insufficiency, or the administration of a potassium-sparing diuretic (e.g., spironolactone, triamterene, amiloride)(see OVERDOSAGE).
WARNINGS
DO NOT ADMINISTER FULL STRENGTH. POTASSIUM CHLORIDE ORAL SOLUTION 10% AND 20% LIQUIDS WILL CAUSE GASTROINTESTINAL IRRITATION IF ADMINISTERED UNDILUTED. FOR DETAILS REGARDING ADEQUATE DILUTION, SEE DOSAGE AND ADMINISTRATION.
Hyperkalemia (see OVERDOSAGE)
In patients with impaired mechanisms for excreting potassium, the administration of potassium salts can produce hyperkalemia and cardiac arrest. This occurs most commonly in patients given potassium by the intravenous route but may also occur in patients given potassium orally. Potentially fatal hyperkalemia can develop rapidly and be asymptomatic. The use of potassium salts in patients with chronic renal disease, or any other condition which impairs potassium excretion, requires particularly careful monitoring of the serum potassium concentration and appropriate dosage adjustment.
Interaction with Potassium Sparing Diuretics
Hypokalemia should not be treated by the concomitant administration of potassium salts and a potassium-sparing diuretic (e.g., spironolactone, triamterene or amiloride) since the simultaneous administration of these agents can produce severe hyperkalemia.
Interaction with Angiotensin Converting Enzyme Inhibitors
Angiotensin converting enzyme (ACE) inhibitors (e.g., captopril, enalapril) will produce some potassium retention by inhibiting aldosterone production. Potassium supplements should be given to patients receiving ACE inhibitors only with close monitoring.
Metabolic Acidosis
Hypokalemia in patients with metabolic acidosis should be treated with an alkalinizing potassium salt such as potassium bicarbonate, potassium citrate, potassium acetate or potassium gluconate.
PRECAUTIONS
General
The diagnosis of potassium depletion is ordinarily made by demonstrating hypokalemia in a patient with a clinical history suggesting some cause for potassium depletion. In interpreting serum potassium level, the physician should bear in mind that acute alkalosis per se can produce hypokalemia in the absence of a deficit in total body potassium while acute acidosis per se can increase the serum potassium concentration into the normal range even in the presence of a reduced total body potassium. The treatment of potassium depletion, particularly in the presence of cardiac disease, renal disease, or acidosis requires careful attention to acid-base balance and appropriate monitoring of serum electrolytes, the electrocardiogram, and the clinical status of the patient.
Potassium chloride oral solution 10% SF orange contains FD&C Yellow No. 6 as a color additive.
Laboratory Tests
When blood is drawn for analysis of plasma potassium it is important to recognize that artifactual elevations can occur after improper venipuncture technique or as a result of in vitro hemolysis of the sample.
Drug Interactions
Potassium-sparing diuretic, angiotensin converting enzyme inhibitors: see WARNINGS.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity, mutagenicity and fertility studies in animals have not been performed. Potassium is a normal dietary constituent.
Pregnancy Category C
Animal reproduction studies have not been conducted with potassium chloride oral solution 10% and 20%. It is unlikely that potassium supplementation that does not lead to hyperkalemia would have an adverse effect on the fetus or would affect reproduction capacity.
Nursing Mothers
The normal potassium content of human milk is about 13 mEq per liter. Since oral potassium becomes part of the body potassium pool, so long as body potassium is not excessive, the contribution of potassium chloride supplementation should have little or no effect on the level in human milk.
ADVERSE REACTIONS
One of the most severe adverse effects is hyperkalemia (see CONTRAINDICATIONS, WARNINGS, and OVERDOSAGE). There also have been reports of upper and lower gastrointestinal conditions including obstruction, bleeding, ulceration and perforation (see CONTRAINDICATIONS and WARNINGS).
The most common adverse reactions to the potassium salts are nausea, vomiting, flatulence, abdominal pain/discomfort and diarrhea. These symptoms are due to irritation to the gastrointestinal tract and are best managed by diluting the preparation further, taking the dose with meals, or reducing the amount taken at one time.
OVERDOSAGE
The administration of oral potassium salts to persons with normal excretory mechanisms for potassium rarely causes serious hyperkalemia. However, if excretory mechanisms are impaired, or if potassium is administered too rapidly intravenously, potentially fatal hyperkalemia can result (see CONTRAINDICATIONS and WARNINGS). It is important to recognize that hyperkalemia is usually asymptomatic and may be manifested only by an increased serum potassium concentration (6.5-8.0 mEq/L) and characteristic electrocardiographic changes (peaking of T-waves, loss of P-wave, depression of S-T segment, and prolongation of the QT interval). Late manifestations include paralysis and cardiovascular collapse from cardiac arrest (9-12 mEq/L).
Treatment measures for hyperkalemia include the following:
- Elimination of foods and medications containing potassium and of any agents with potassium-sparing properties;
- Intravenous administration of 300 to 500 mL/hr of 10% dextrose solution containing 10-20 units of crystalline insulin per 1,000 mL;
- Correction of acidosis, if present, with intravenous sodium bicarbonate;
- Use of exchange resins, hemodialysis, or peritoneal dialysis.
In treating hyperkalemia, it should be recalled that in patients who have been stabilized on digitalis, too rapid a lowering of the serum potassium concentration can produce digitalis toxicity.
DOSAGE AND ADMINISTRATION
The usual dietary potassium intake by the average adult is 50 to 100 mEq per day. Potassium depletion sufficient to cause hypokalemia usually requires the loss of 200 or more mEq of potassium from the total body store. Dosage must be adjusted to the individual needs of each patient. The dose for the prevention of hypokalemia is typically in the range of 20 mEq per day. Doses of 40-100 mEq per day or more are used for the treatment of potassium depletion.
Potassium Chloride Oral Solution 10%
To minimize gastrointestinal irritation, patients must follow directions regarding dilution. Each tablespoonful (15 mL) should be diluted with three (3) fluid ounces or more of water or other liquid.
One (1) tablespoonful (15 mL) twice daily (after morning and evening meals) supplies 40 mEq of potassium chloride.
Potassium Chloride Oral Solutions 20%
To minimize gastrointestinal irritation, patients must follow directions regarding dilution. Each tablespoonful should be diluted with six (6) fluid ounces or more of water or other liquid.
One (1) tablespoonful (15 mL) per day (after the morning meal) supplies 40 mEq of potassium chloride. One tablespoonful twice a day, provides 80 mEq of potassium chloride.
Deviations from these recommendations may be indicated, since no average total daily dose can be defined but must be governed by close observation for clinical effects. However, potassium intoxication may result from any therapeutic dosage. See OVERDOSAGE and PRECAUTIONS.
| POTASSIUM CHLORIDE
potassium chloride solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| POTASSIUM CHLORIDE
potassium chloride solution |
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
| POTASSIUM CHLORIDE
potassium chloride solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| POTASSIUM CHLORIDE
potassium chloride solution |
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
| Labeler - Qualitest Pharmaceuticals (011103059) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Vintage Pharmaceuticals-Huntsville | 825839835 | MANUFACTURE(0603-1532, 0603-1534, 0603-1535, 0603-1536) | |