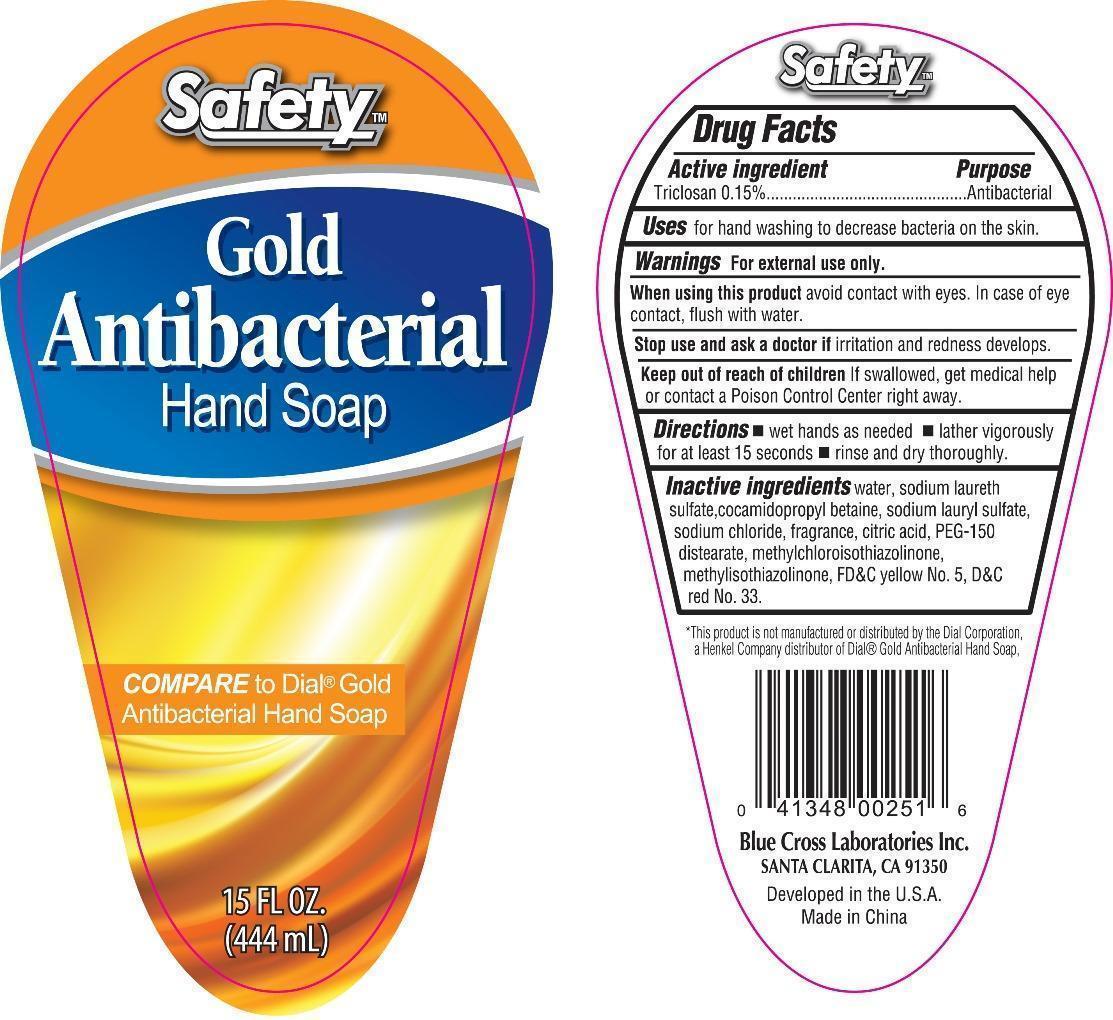
SAFETY GOLD ANTIBACTERIAL HAND- triclosan liquid
Blue Cross Laboratories, Inc
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Gold Antibacterial Soap
Active ingredient Purpose
Triclosan 0.15% Antibacterial
Uses for hand washing to decrease bacteria on the skin
keep out of reach of children
If swallowed, get medical help or contact a poison control center right away
| SAFETY GOLD ANTIBACTERIAL HAND
triclosan liquid |
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
| Labeler - Blue Cross Laboratories, Inc (008298879) |
| Registrant - Ningbo Liyuan Daily Chemical Products Co., Ltd. (530766098) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Ningbo Liyuan Daily Chemical Products Co., Ltd. | 530766098 | manufacture(22431-583) | |
Revised: 1/2021
Document Id: b82b6db1-f4dd-c296-e053-2a95a90a9093
Set id: 316f6e17-d574-4c0a-bb12-aa589840d586
Version: 5
Effective Time: 20210105
Blue Cross Laboratories, Inc