PREVPAC- lansoprazole, amoxicillin and clarithromycin
Takeda Pharmaceuticals America, Inc.
----------
PREVPAC
(lansoprazole 30 mg delayed release capsules, amoxicillin 500 mg capsules, USP, and clarithromycin 500 mg tablets, USP)
Rx only
To reduce the development of drug-resistant bacteria and maintain the effectiveness of PREVPAC and other antibacterial drugs, PREVPAC should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria.
THESE PRODUCTS ARE INTENDED ONLY FOR USE AS DESCRIBED. The individual products contained in this package should not be used alone or in combination for other purposes. The information described in this labeling concerns only the use of these products as indicated in this daily administration pack. For information on use of the individual components when dispensed as individual medications outside this combined use for treating Helicobacter pylori (H. pylori), please see the package inserts for each individual product.
DESCRIPTION
PREVPAC consists of a daily administration card containing two PREVACID 30 mg delayed release capsules, four amoxicillin 500 mg capsules, USP, and two clarithromycin 500 mg tablets, USP, for oral administration.
PREVACID (lansoprazole) Delayed-Release Capsules
The active ingredient in PREVACID delayed-release capsules is lansoprazole, a proton pump inhibitor. Its empirical formula is C16H14F3N3O2S with a molecular weight of 369.37. PREVACID has the following structure:
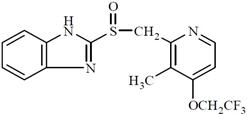
Lansoprazole is a white to brownish-white odorless crystalline powder which melts with decomposition at approximately 166°C. Lansoprazole is freely soluble in dimethylformamide; soluble in methanol; sparingly soluble in ethanol; slightly soluble in ethyl acetate, dichloromethane and acetonitrile; very slightly soluble in ether; and practically insoluble in hexane and water.
Each delayed-release capsule contains enteric-coated granules consisting of 30 mg of lansoprazole (active ingredient) and the following inactive ingredients: sugar sphere, sucrose, methacrylic acid copolymer, low substituted hydroxypropyl cellulose, starch, magnesium carbonate, talc, polyethylene glycol, titanium dioxide, polysorbate 80, hydroxypropyl cellulose, colloidal silicon dioxide D&C Red No. 28, FD&C Blue No. 1, and FD&C Red No. 40.
Amoxicillin Capsules, USP
Amoxicillin is a penicillin class antibacterial, with a broad spectrum of bactericidal activity against many gram-positive and gram-negative microorganisms. Chemically it is (2S, 5R, 6R)-6-[(R)-(-)-2-amino-2-(p-hydroxyphenyl)acetamido]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo [3.2.0] heptane-2-carboxylic acid trihydrate. The molecular formula is C16H19N3O5S ∙ 3H2O and the molecular weight is 419.45. Amoxicillin has the following structure:
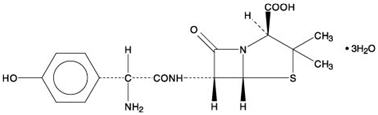
Amoxicillin capsules are intended for oral administration.
Each capsule, with yellow opaque cap and body, contains 500 mg amoxicillin trihydrate. Inactive ingredients: Capsule shells – yellow ferric oxide, titanium dioxide, gelatin, black ferric oxide; Capsule contents – cellulose microcrystalline and magnesium stearate.
Meets USP Dissolution Test 2.
BIAXIN Filmtab (clarithromycin tablets, USP)
Clarithromycin is a macrolide antimicrobial. Chemically, it is 6-0-methylerythromycin. The molecular formula is C38H69NO13, and the molecular weight is 747.96. Clarithromycin has the following structure:
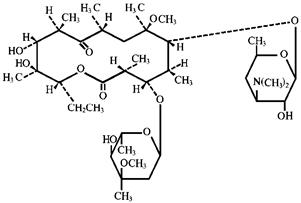
Clarithromycin is a white to off-white crystalline powder. It is soluble in acetone, slightly soluble in methanol, ethanol, and acetonitrile, and practically insoluble in water.
Each yellow oval film-coated immediate-release tablet contains 500 mg of clarithromycin and the following inactive ingredients: hypromellose, hydroxypropyl cellulose, colloidal silicon dioxide, croscarmellose sodium, D&C Yellow No. 10, magnesium stearate, microcrystalline cellulose, povidone, propylene glycol, sorbic acid, sorbitan monooleate, titanium dioxide, and vanillin.
CLINICAL PHARMACOLOGY
Pharmacokinetics
Pharmacokinetics when all three of the PREVPAC components (PREVACID capsules, amoxicillin capsules, clarithromycin tablets) were coadministered has not been studied. Studies have shown no clinically significant interactions of PREVACID and amoxicillin or PREVACID and clarithromycin when administered together. There is no information about the gastric mucosal concentrations of PREVACID, amoxicillin and clarithromycin after administration of these agents concomitantly. The systemic pharmacokinetic information presented below is based on studies in which each product was administered alone.
PREVACID
Absorption
PREVACID capsules contain an enteric-coated granule formulation of lansoprazole (because lansoprazole is acid-labile), so that absorption of lansoprazole begins only after the granules leave the stomach.The mean peak plasma levels of lansoprazole occur at approximately 1.7 hours. After a single-dose administration of 15 to 60 mg of oral lansoprazole, the peak plasma concentrations (Cmax) of lansoprazole and the area under the plasma concentration curves (AUCs) of lansoprazole were approximately proportional to the administered dose. Lansoprazole does not accumulate and its pharmacokinetics are unaltered by multiple dosing. The absolute bioavailability is over 80%. In healthy subjects, the mean (±SD) plasma half-life was 1.5 (±1.0) hours. Both the Cmax and AUC are diminished by about 50 to 70% if lansoprazole is given 30 minutes after food, compared to the fasting condition. There is no significant food effect if lansoprazole is given before meals.
Distribution
Lansoprazole is 97% bound to plasma proteins. Plasma protein binding is constant over the concentration range of 0.05 to 5 mcg/mL.
Elimination
Metabolism
Lansoprazole is extensively metabolized in the liver. Two metabolites have been identified in measurable quantities in plasma (the hydroxylated sulfinyl and sulfone derivatives of lansoprazole). These metabolites have very little or no antisecretory activity. Lansoprazole is thought to be transformed into two active species which inhibit acid secretion by blocking the proton pump [(H+, K+)-ATPase enzyme system] at the secretory surface of the gastric parietal cell. The two active species are not present in the systemic circulation. The plasma elimination half-life of lansoprazole is less than two hours while the acid inhibitory effect lasts more than 24 hours. Therefore, the plasma elimination half-life of lansoprazole does not reflect its duration of suppression of gastric acid secretion.
Excretion
Following single-dose oral administration of PREVACID, virtually no unchanged lansoprazole was excreted in the urine. In one study, after a single oral dose of 14C-lansoprazole, approximately one-third of the administered radiation was excreted in the urine and two-thirds was recovered in the feces. This implies a significant biliary excretion of the lansoprazole metabolites.
Amoxicillin
Absorption
Amoxicillin is stable in the presence of gastric acid and may be given without regard to meals. It is rapidly absorbed after oral administration. Orally administered doses of 500 mg amoxicillin capsules result in average peak blood levels one to two hours after administration in the range of 5.5 to 7.5 mcg/mL.
Distribution
Amoxicillin diffuses readily into most body tissues and fluids, with the exception of brain and spinal fluid, except when meninges are inflamed. In blood serum, amoxicillin is approximately 20% protein-bound.
Metabolism/Elimination
The elimination half-life of amoxicillin is 61.3 minutes. Detectable serum levels are observed up to eight hours after an orally administered dose of amoxicillin. Approximately 60% of the orally administered dose of amoxicillin is excreted unchanged in the urine within six to eight hours post-dose; its excretion can be delayed by concurrent administration of probenecid.
Clarithromycin
Absorption
Clarithromycin is rapidly absorbed from the gastrointestinal tract after oral administration. The absolute bioavailability of 250 mg clarithromycin tablets was approximately 50%. For a single 500 mg dose of clarithromycin, food slightly delays the onset of clarithromycin absorption, increasing the peak time from approximately two to 2.5 hours. Food also increases the clarithromycin peak plasma concentration by about 24%, but does not affect the extent of clarithromycin bioavailability. Food does not affect the onset of formation of the antimicrobially active metabolite, 14-OH clarithromycin or its peak plasma concentration but does slightly decrease the extent of metabolite formation, indicated by an 11% decrease in area under the plasma concentration-time curve (AUC). Therefore, clarithromycin tablets may be given without regard to food.
In nonfasting, healthy human subjects (males and females), peak plasma concentrations were attained within two to three hours after oral dosing. Steady-state peak plasma clarithromycin concentrations were attained within three days and were approximately 3 to 4 mcg/mL with a 500 mg dose administered every eight to 12 hours.
Metabolism/Elimination
The elimination half-life of clarithromycin was five to seven hours with 500 mg administered every eight to 12 hours. The nonlinearity of clarithromycin pharmacokinetics is slight at the recommended dose of 500 mg administered every eight to 12 hours. With a 500 mg every eight to 12 hours dosing, the peak steady-state concentration of 14-OH clarithromycin is up to 1 mcg/mL, and its elimination half-life is about seven to nine hours. The steady-state concentration of this metabolite is generally attained within three to four days.
After a 500 mg tablet every 12 hours, the urinary excretion of clarithromycin is approximately 30%. The renal clearance of clarithromycin approximates the normal glomerular filtration rate. The major metabolite found in urine is 14-OH clarithromycin, which accounts for an additional 10 to 15% of the dose with a 500 mg tablet administered every 12 hours.
Steady-state concentrations of clarithromycin and 14-OH clarithromycin observed following administration of 500 mg doses of clarithromycin every 12 hours to adult patients with HIV infection were similar to those observed in healthy volunteers. In adult HIV-infected patients taking 500 mg doses of clarithromycin every 12 hours, steady-state clarithromycin Cmax values ranged from 2 to 4 mcg/mL.
The steady-state concentrations of clarithromycin in subjects with impaired hepatic function did not differ from those in normal subjects; however, the 14-OH clarithromycin concentrations were lower in the hepatically impaired subjects. The decreased formation of 14-OH clarithromycin was at least partially offset by an increase in renal clearance of clarithromycin in the subjects with impaired hepatic function when compared to healthy subjects.
The pharmacokinetics of clarithromycin was also altered in subjects with impaired renal function (see PRECAUTIONS and DOSAGE AND ADMINISTRATION).
Clarithromycin and the 14-OH clarithromycin metabolite distribute readily into body tissues and fluids. There are no data available on cerebrospinal fluid penetration. Because of high intracellular concentrations, tissue concentrations are higher than serum concentrations.
Specific Populations
Geriatric Patients
The clearance of lansoprazole is decreased in the elderly, with elimination half-life increased approximately 50 to 100%. Because the mean half-life in the elderly remains between 1.9 to 2.9 hours, repeated once daily dosing does not result in accumulation of lansoprazole. Peak plasma levels were not increased in the elderly.
Male and Female Patients
In a study comparing 12 male and six female human subjects who received lansoprazole, no sex-related differences were found in pharmacokinetics and intragastric pH results.
Racial or Ethnic Groups
The pooled mean pharmacokinetic parameters of PREVACID from twelve U.S. studies (N=513) were compared to the mean pharmacokinetic parameters from two Asian studies (N=20). The mean AUCs of PREVACID in Asian subjects were approximately twice those seen in pooled U.S. data; however, the inter-individual variability was high. The Cmax values were comparable.
Patients with Renal Impairment
In patients with severe renal impairment, plasma protein binding decreased by 1 to 1.5% after administration of 60 mg of lansoprazole. Patients with renal impairment had a shortened elimination half-life and decreased total AUC (free and bound). The AUC for free lansoprazole in plasma, however, was not related to the degree of renal impairment; and the Cmax and Tmax (time to reach the maximum concentration) were not different than the Cmax and Tmax from subjects with normal renal function. Therefore, the pharmacokinetics of lansoprazole were not clinically different in patients with mild, moderate or severe renal impairment compared to healthy subjects with normal renal function.
Patients with Hepatic Impairment
In patients with mild (Child-Pugh Class A) or moderate (Child-Pugh Class B) hepatic impairment there was an approximate 3-fold increase in mean AUC compared to healthy subjects with normal hepatic function following multiple oral doses of 30 mg PREVACID for 7 days. The corresponding mean plasma half-life of lansoprazole was prolonged from 1.5 hours to 4 hours (Child-Pugh A) or 5 hours (Child-Pugh B).
In patients with compensated and decompensated cirrhosis, there was an approximate 6- and 5-fold increase in AUC, respectively, compared to healthy subjects with normal hepatic function following a single oral dose of 30 mg PREVACID (see DOSAGE AND ADMINISTRATION).
Drug Interaction Studies
Effect of Lansoprazole on Other Drugs
Cytochrome P450 Interactions
Lansoprazole is metabolized through the cytochrome P450 system, specifically through the CYP3A and CYP2C19 isozymes. Studies have shown that PREVACID does not have clinically significant interactions with other drugs metabolized by the cytochrome P450 system, such as warfarin, antipyrine, indomethacin, ibuprofen, phenytoin, propranolol, prednisone, diazepam, or clarithromycin in healthy subjects. These compounds are metabolized through various cytochrome P450 isozymes including CYP1A2, CYP2C9, CYP2C19, CYP2D6, and CYP3A.
Theophylline
When PREVACID was administered concomitantly with theophylline (CYP1A2, CYP3A), a minor increase (10%) in the clearance of theophylline was seen. Because of the small magnitude and the direction of the effect on theophylline clearance, this interaction is unlikely to be of clinical concern (see PRECAUTIONS, Drug Interactions).
Methotrexate and 7-hydroxymethotrexate
In an open-label, single-arm, eight day, pharmacokinetic study of 28 adult rheumatoid arthritis patients (who required the chronic use of 7.5 to 15 mg of methotrexate given weekly), administration of seven days of naproxen 500 mg twice daily and PREVACID 30 mg daily had no effect on the pharmacokinetics of methotrexate and 7-hydroxymethotrexate. While this study was not designed to assess the safety of this combination of drugs, no major adverse reactions were noted. However, this study was conducted with low doses of methotrexate. A drug interaction study with high doses of methotrexate has not been conducted (see WARNINGS).
Amoxicillin
PREVACID has also been shown to have no clinically significant interaction with amoxicillin.
Sucralfate
In a single-dose crossover study examining PREVACID 30 mg administered alone and concomitantly with sucralfate 1 gram, absorption of lansoprazole was delayed and the bioavailability was reduced by 17% when administered concomitantly with sucralfate (see PRECAUTIONS, Drug Interactions).
Antacids
In clinical trials, antacids were administered concomitantly with PREVACID and there was no evidence of a change in the efficacy of PREVACID.
Clopidogrel
Clopidogrel is metabolized to its active metabolite in part by CYP2C19. A study of healthy subjects who were CYP2C19 extensive metabolizers, receiving once daily administration of clopidogrel 75 mg alone or concomitantly with PREVACID 30 mg (n=40), for nine days was conducted. The mean AUC of the active metabolite of clopidogrel was reduced by approximately 14% (mean AUC ratio was 86%, with 90% CI of 80 to 92%) when PREVACID was coadministered compared to administration of clopidogrel alone.
Pharmacodynamic parameters were also measured and demonstrated that the change in inhibition of platelet aggregation (induced by 5 mcM ADP) was related to the change in the exposure to clopidogrel active metabolite. The effect on exposure to the active metabolite of clopidogrel and on clopidogrel-induced platelet inhibition is not considered clinically important.
Pharmacodynamics
Microbiology
Lansoprazole, clarithromycin and/or amoxicillin have been shown to be active against most strains of Helicobacter pylori in vitro and in clinical infections as described in the INDICATIONS AND USAGE section.
Helicobacter pylori Pretreatment Resistance
Clarithromycin pretreatment resistance rates were 9.5% (91/960) by E-test and 11.3% (12/106) by agar dilution in the dual and triple therapy clinical trials (M93-125, M93-130, M93-131, M95-392, and M95-399).
Amoxicillin pretreatment susceptible isolates (≤0.25 mcg/mL) occurred in 97.8% (936/957) and 98.0% (98/100) of the patients in the dual and triple therapy clinical trials by E-test and agar dilution, respectively. Twenty-one of 957 patients (2.2%) by E-test, and two of 100 patients (2.0%) by agar dilution, had amoxicillin pretreatment MICs of greater than 0.25 mcg/mL. One patient on the 14 day triple therapy regimen had an unconfirmed pretreatment amoxicillin minimum inhibitory concentration (MIC) of greater than 256 mcg/mL by E-test and the patient was eradicated of H. pylori.
| Clarithromycin Pretreatment Results | Clarithromycin Post-treatment Results | |||||
|---|---|---|---|---|---|---|
| H. pylori negative - eradicated | H. pylori positive – not eradicated Post-treatment susceptibility results |
|||||
| S† | I† | R† | No MIC | |||
|
||||||
| Triple Therapy 14 Day (lansoprazole 30 mg twice daily/amoxicillin 1 g twice daily/clarithromycin 500 mg twice daily) (M95-399, M93-131, M95-392) | ||||||
| Susceptible† | 112 | 105 | 7 | |||
| Intermediate† | 3 | 3 | ||||
| Resistant† | 17 | 6 | 7 | 4 | ||
| Triple Therapy 10 Day (lansoprazole 30 mg twice daily/amoxicillin 1 g twice daily/clarithromycin 500 mg twice daily) (M95-399) | ||||||
| Susceptible† | 42 | 40 | 1 | 1 | ||
| Intermediate† | ||||||
| Resistant† | 4 | 1 | 3 | |||
Patients not eradicated of H. pylori following lansoprazole/amoxicillin/clarithromycin triple therapy will likely have clarithromycin resistant H. pylori isolates. Therefore, for those patients who fail therapy, clarithromycin susceptibility testing should be done if possible. Patients with clarithromycin resistant H. pylori should not be treated with lansoprazole/amoxicillin/clarithromycin triple therapy or other regimens which include clarithromycin as the sole antimicrobial agent.
Amoxicillin Susceptibility Test Results and Clinical/Bacteriological Outcomes
In the dual and triple therapy clinical trials, 82.6% (195/236) of the patients that had pretreatment amoxicillin susceptible MICs (≤0.25 mcg/mL) were eradicated of H. pylori. Of those with pretreatment amoxicillin MICs of greater than 0.25 mcg/mL, three of six had the H. pylori eradicated. A total of 30% (21/70) of the patients failed lansoprazole 30 mg three times daily per amoxicillin 1 g three times daily dual therapy and a total of 12.8% (22/172) of the patients failed the 10 and 14 day triple therapy regimens. Post-treatment susceptibility results were not obtained on 11 of the patients who failed therapy. Nine of the 11 patients with amoxicillin post-treatment MICs that failed the triple therapy regimen also had clarithromycin resistant H. pylori isolates.
Susceptibility Test for Helicobacter pylori
The reference methodology for susceptibility testing of H. pylori is agar dilution MICs.1 One to three microliters of an inoculum equivalent to a No. 2 McFarland standard (1 × 107-1 × 108 CFU/mL for H. pylori) are inoculated directly onto freshly prepared antimicrobial containing Mueller-Hinton agar plates with 5% aged defibrinated sheep blood (greater than two weeks old). The agar dilution plates are incubated at 35°C in a microaerobic environment produced by a gas generating system suitable for Campylobacter species. After three days of incubation, the MICs are recorded as the lowest concentration of antimicrobial agent required to inhibit growth of the organism. The clarithromycin and amoxicillin MIC values should be interpreted according to the following criteria:
| Susceptibility Test Interpretive Criteria for H. pylori | |
| Clarithromycin MIC (mcg/mL)* | Interpretation |
| ≤0.25 | Susceptible (S) |
| 0.5 | Intermediate (I) |
| ≥1.0 | Resistant (R) |
| Susceptibility Test Interpretive Criteria for H. pylori | |
| Amoxicillin MIC (mcg/mL)*,† | Interpretation |
| <0.25 | Susceptible (S) |
Standardized susceptibility test procedures require the use of laboratory control bacteria to monitor and ensure the accuracy and precision of supplies and reagents in the assay, and the techniques of the individual performing the test. Standard clarithromycin or amoxicillin powder should provide the following MIC ranges.
| Acceptable Quality Control Ranges | Antimicrobial Agent | MIC (mcg/mL)* |
|---|---|---|
|
||
| H. pylori ATCC 43504 | Clarithromycin | 0.015 - 0.12 |
| H. pylori ATCC 43504 | Amoxicillin | 0.015 - 0.12 |
Antisecretory activity
After oral administration, lansoprazole was shown to significantly decrease the basal acid output and significantly increase the mean gastric pH and percent of time the gastric pH was greater than three and greater than four. Lansoprazole also significantly reduced meal-stimulated gastric acid output and secretion volume, as well as pentagastrin-stimulated acid output. In patients with hypersecretion of acid, lansoprazole significantly reduced basal and pentagastrin-stimulated gastric acid secretion. Lansoprazole inhibited the normal increases in secretion volume, acidity and acid output induced by insulin.
The intragastric pH results of a five day, pharmacodynamic, crossover study of 15 mg and 30 mg of once daily lansoprazole are presented in Table 2.
| PREVACID | |||||
|---|---|---|---|---|---|
| Parameter | Baseline Value | 15 mg | 30 mg | ||
| Day 1 | Day 5 | Day 1 | Day 5 | ||
| NOTE: An intragastric pH of greater than 4 reflects a reduction in gastric acid by 99%. | |||||
| Mean 24 Hour pH | 2.1 | 2.7* | 4.0* | 3.6† | 4.9† |
| Mean Nighttime Hour pH | 1.9 | 2.4 | 3.0* | 2.6 | 3.8† |
| % Time Gastric pH>3 | 18 | 33* | 59* | 51† | 72† |
| % Time Gastric pH>4 | 12 | 22* | 49* | 41† | 66† |
After the initial dose in this study, increased gastric pH was seen within one to two hours with 30 mg of lansoprazole and two to three hours with 15 mg of lansoprazole. After multiple daily dosing, increased gastric pH was seen within the first hour post dosing with 30 mg of lansoprazole and within one to two hours post dosing with 15 mg of lansoprazole.
Acid suppression may enhance the effect of antimicrobials in eradicating Helicobacter pylori (H. pylori). The percentage of time gastric pH was elevated above five and six was evaluated in a crossover study of PREVACID given daily, twice daily and three times daily.
| PREVACID | ||||
|---|---|---|---|---|
| Parameter | 30 mg daily | 15 mg twice daily | 30 mg twice daily | 30 mg three times daily |
| % Time Gastric pH>5 | 43 | 47 | 59* | 77† |
| % Time Gastric pH>6 | 20 | 23 | 28 | 45† |
The inhibition of gastric acid secretion as measured by intragastric pH gradually returned to normal over two to four days after multiple doses. There was no indication of rebound gastric acidity.
Enterochromaffin-like (ECL) Cell Effects
During lifetime exposure of rats with up to 150 mg/kg/day of lansoprazole dosed seven days per week, marked hypergastrinemia was observed followed by ECL cell proliferation and formation of carcinoid tumors, especially in female rats. Gastric biopsy specimens from the body of the stomach from approximately 150 patients treated continuously with lansoprazole for at least one year did not show evidence of ECL cell effects similar to those seen in rat studies. Longer term data are needed to rule out the possibility of an increased risk of the development of gastric tumors in patients receiving long-term therapy with lansoprazole (see PRECAUTIONS, Carcinogenesis, Mutagenesis, Impairment of Fertility).
Other Gastric Effects in Humans
Lansoprazole did not significantly affect mucosal blood flow in the fundus of the stomach. Due to the normal physiologic effect caused by the inhibition of gastric acid secretion, a decrease of about 17% in blood flow in the antrum, pylorus, and duodenal bulb was seen. Lansoprazole significantly slowed the gastric emptying of digestible solids. Lansoprazole increased serum pepsinogen levels and decreased pepsin activity under basal conditions and in response to meal stimulation or insulin injection. As with other agents that elevate intragastric pH, increases in gastric pH were associated with increases in nitrate-reducing bacteria and elevation of nitrite concentration in gastric juice in patients with gastric ulcer. No significant increase in nitrosamine concentrations was observed.
Serum Gastrin Effects
In over 2100 patients, median fasting serum gastrin levels increased 50 to 100% from baseline but remained within normal range after treatment with 15 to 60 mg of oral lansoprazole. These elevations reached a plateau within two months of therapy and returned to pretreatment levels within four weeks after discontinuation of therapy.
Increased gastrin causes enterochromaffin-like cell hyperplasia and increased serum CgA levels. The increased CgA levels may cause false positive results in diagnostic investigations for neuroendocrine tumors (see WARNINGS).
Endocrine Effects
Human studies for up to one year have not detected any clinically significant effects on the endocrine system. Hormones studied include testosterone, luteinizing hormone (LH), follicle stimulating hormone (FSH), sex hormone binding globulin (SHBG), dehydroepiandrosterone sulfate (DHEA-S), prolactin, cortisol, estradiol, insulin, aldosterone, parathormone, glucagon, thyroid stimulating hormone (TSH), triiodothyronine (T3), thyroxine (T4), and somatotropic hormone (STH). Lansoprazole in oral doses of 15 to 60 mg for up to one year had no clinically significant effect on sexual function. In addition, lansoprazole in oral doses of 15 to 60 mg for two to eight weeks had no clinically significant effect on thyroid function. In 24 month carcinogenicity studies in Sprague-Dawley rats with daily lansoprazole dosages up to 150 mg/kg, proliferative changes in the Leydig cells of the testes, including benign neoplasm, were increased compared to control rats.
Other Effects
No systemic effects of lansoprazole on the central nervous system, lymphoid, hematopoietic, renal, hepatic, cardiovascular, or respiratory systems have been found in humans. Among 56 patients who had extensive baseline eye evaluations, no visual toxicity was observed after lansoprazole treatment (up to 180 mg/day) for up to 58 months. After lifetime lansoprazole exposure in rats, focal pancreatic atrophy, diffuse lymphoid hyperplasia in the thymus, and spontaneous retinal atrophy were seen.
CLINICAL STUDIES
H. pylori Eradication to Reduce the Risk of Duodenal Ulcer Recurrence
Randomized, double-blind clinical studies performed in the U.S. in patients with H. pylori and duodenal ulcer disease (defined as an active ulcer or history of an ulcer within one year) evaluated the efficacy of PREVPAC as triple 14 day therapy for the eradication of H. pylori. The triple therapy regimen (PREVACID 30 mg twice daily/amoxicillin 1 g twice daily/clarithromycin 500 mg twice daily) produced statistically significantly higher eradication rates than PREVACID plus amoxicillin, PREVACID plus clarithromycin, and amoxicillin plus clarithromycin dual therapies.
H. pylori eradication was defined as two negative tests (culture and histology) at four to six weeks following the end of treatment.
Triple therapy was shown to be more effective than all possible dual therapy combinations. The combination of PREVACID plus amoxicillin and clarithromycin as triple therapy was effective in eradicating H. pylori. Eradication of H. pylori has been shown to reduce the risk of duodenal ulcer recurrence.
A randomized, double-blind clinical study performed in the U.S. in patients with H. pylori and duodenal ulcer disease (defined as an active ulcer or history of an ulcer within one year) compared the efficacy of PREVACID triple therapy for ten and 14 days. This study established that the 10 day triple therapy was equivalent to the 14 day triple therapy in eradicating H. pylori.
| Study | Duration | Triple Therapy Evaluable Analysis* | Triple Therapy Intent-to-Treat Analysis† |
|---|---|---|---|
|
|||
| M93-131 | 14 days | 92‡
[80.0-97.7] (N=48) | 86‡
[73.3-93.5] (N=55) |
| M95-392 | 14 days | 86§
[75.7-93.6] (N=66) | 83§
[72.0-90.8] (N=70) |
| M95-399¶ | 14 days | 85 [77.0-91.0] (N=113) | 82 [73.9-88.1] (N=126) |
| 10 days | 84 [76.0-89.8] (N=123) | 81 [73.9-87.6] (N=135) |
|
INDICATIONS AND USAGE
H. pylori Eradication to Reduce the Risk of Duodenal Ulcer Recurrence
The components in PREVPAC (PREVACID, amoxicillin, and clarithromycin) are indicated for the treatment of patients with H. pylori infection and duodenal ulcer disease (active or one year history of a duodenal ulcer) to eradicate H. pylori. Eradication of H. pylori has been shown to reduce the risk of duodenal ulcer recurrence (see CLINICAL STUDIES and DOSAGE AND ADMINISTRATION).
To reduce the development of drug-resistant bacteria and maintain the effectiveness of PREVPAC and other antibacterial drugs, PREVPAC should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
CONTRAINDICATIONS
PREVPAC is contraindicated in patients with known severe hypersensitivity to any component of the formulation of PREVACID. Hypersensitivity reactions may include anaphylaxis, anaphylactic shock, angioedema, bronchospasm, acute interstitial nephritis, and urticaria (see ADVERSE REACTIONS). Proton Pump Inhibitors (PPIs), including PREVACID, are contraindicated with rilpivirine-containing products (see CLINICAL PHARMACOLOGY, Drug Interaction Studies).
A history of severe hypersensitivity reactions (e.g., anaphylaxis or Stevens-Johnson Syndrome) to amoxicillin or other beta-lactam antibiotics (e.g., penicillins and cephalosporins) is a contraindication.
Clarithromycin is contraindicated in patients with a known hypersensitivity to clarithromycin, erythromycin, or any of the macrolide antibiotics.
Clarithromycin is contraindicated in patients with a history of cholestatic jaundice/hepatic dysfunction associated with prior use of clarithromycin.
Clarithromycin should not be given to patients with history of QT prolongation or ventricular cardiac arrhythmia, including torsades de pointes.
Concomitant administration of clarithromycin, a component of PREVPAC, and any of the following drugs is contraindicated: cisapride, pimozide, astemizole, terfenadine, ergotamine or dihydroergotamine (see PRECAUTIONS, Drug Interactions). There have been postmarketing reports of drug interactions when clarithromycin and/or erythromycin are coadministered with cisapride, pimozide, astemizole, or terfenadine resulting in cardiac arrhythmias (QT prolongation, ventricular tachycardia, ventricular fibrillation, and torsades de pointes) most likely due to inhibition of metabolism of these drugs by erythromycin and clarithromycin. Fatalities have been reported.
Concomitant administration of clarithromycin and colchicine is contraindicated in patients with renal or hepatic impairment.
Clarithromycin should not be used concomitantly with HMG-CoA reductase inhibitors (statins) that are extensively metabolized by CYP3A4 (lovastatin or simvastatin), due to the increased risk of myopathy, including rhabdomyolysis (see WARNINGS).
WARNINGS
Acute Hypersensitivity Reactions
Serious and occasionally fatal hypersensitivity (anaphylactic) reactions have been reported in patients on penicillin therapy, including amoxicillin. Although anaphylaxis is more frequent following parenteral therapy, it has occurred in patients on oral penicillins. These reactions are more likely to occur in individuals with a history of penicillin hypersensitivity and/or a history of sensitivity to multiple allergens. There have been reports of individuals with a history of penicillin hypersensitivity who have experienced severe reactions when treated with cephalosporins. Before initiating therapy with PREVPAC careful inquiry should be made regarding previous hypersensitivity reactions to penicillins, cephalosporins, or other allergens. In the event of severe acute hypersensitivity reactions, such as anaphylaxis, Stevens-Johnson Syndrome, toxic epidermal necrolysis, drug rash with eosinophilia and systemic symptoms (DRESS), and Henoch-Schonlein purpura PREVPAC should be discontinued immediately and appropriate treatment should be urgently initiated.
Use in Pregnancy
CLARITHROMYCIN SHOULD NOT BE USED IN PREGNANT WOMEN EXCEPT IN CLINICAL CIRCUMSTANCES WHERE NO ALTERNATIVE THERAPY IS APPROPRIATE. IF PREGNANCY OCCURS WHILE TAKING CLARITHROMYCIN, THE PATIENT SHOULD BE INFORMED OF THE POTENTIAL HAZARD TO THE FETUS. CLARITHROMYCIN HAS DEMONSTRATED ADVERSE EFFECTS OF PREGNANCY OUTCOME AND/OR EMBRYO-FETAL DEVELOPMENT IN MONKEYS, RATS, MICE, AND RABBITS AT DOSES THAT PRODUCED PLASMA LEVELS TWO TO 17 TIMES THE SERUM LEVELS ACHIEVED IN HUMANS TREATED AT THE MAXIMUM RECOMMENDED HUMAN DOSES (see PRECAUTIONS, Pregnancy).
Hepatotoxicity
Hepatic dysfunction, including increased liver enzymes, and hepatocellular and/or cholestatic hepatitis, with or without jaundice, has been reported with clarithromycin. This hepatic dysfunction may be severe and is usually reversible. In some instances, hepatic failure with fatal outcome has been reported and generally has been associated with serious underlying diseases and/or concomitant medications. Symptoms of hepatitis can include anorexia, jaundice, dark urine, pruritus, or tender abdomen. Discontinue clarithromycin immediately if signs and symptoms of hepatitis occur.
QT Prolongation
Clarithromycin has been associated with prolongation of the QT interval and infrequent cases of arrhythmia. Cases of torsades de pointes have been spontaneously reported during postmarketing surveillance in patients receiving clarithromycin. Fatalities have been reported. Clarithromycin should be avoided in patients with ongoing proarrhythmic conditions such as uncorrected hypokalemia or hypomagnesemia, clinically significant bradycardia (see CONTRAINDICATIONS) and in patients receiving Class IA (quinidine, procainamide) or Class III (dofetilide, amiodarone, sotalol) antiarrhythmic agents. Elderly patients may be more susceptible to drug-associated effects on the QT interval.
Presence of Gastric Malignancy
In adults, symptomatic response to therapy with lansoprazole does not preclude the presence of gastric malignancy. Consider additional follow-up and diagnostic testing in adult patients who have a suboptimal response or an early symptomatic relapse after completing treatment with a PPI. In older patients, also consider an endoscopy.
Acute Interstitial Nephritis
Acute interstitial nephritis (AIN) has been observed in patients taking proton pump inhibitors (PPIs) including lansoprazole. Acute interstitial nephritis may occur at any point during PPI therapy and is generally attributed to an idiopathic hypersensitivity reaction. Discontinue lansoprazole if AIN develops (see CONTRAINDICATIONS).
Cutaneous and Systemic Lupus Erythematosus
Cutaneous lupus erythematosus (CLE) and systemic lupus erythematosus (SLE) have been reported in patients taking PPIs, including lansoprazole. These events have occurred as both new onset and an exacerbation of existing autoimmune disease. The majority of PPI-induced lupus erythematosus cases were CLE.
The most common form of CLE reported in patients treated with PPIs was subacute CLE (SCLE) and occurred within weeks to years after continuous drug therapy in patients ranging from infants to the elderly. Generally, histological findings were observed without organ involvement.
Systemic lupus erythematosus (SLE) is less commonly reported than CLE in patients receiving PPIs. PPI associated SLE is usually milder than non-drug induced SLE. Onset of SLE typically occurred within days to years after initiating treatment primarily in patients ranging from young adults to the elderly. The majority of patients presented with rash; however, arthralgia and cytopenia were also reported.
Avoid administration of PPIs for longer than medically indicated. If signs or symptoms consistent with CLE or SLE are noted in patients receiving PREVPAC, discontinue the drug and refer the patient to the appropriate specialist for evaluation. Most patients improve with discontinuation of the PPI alone in four to 12 weeks. Serological testing (e.g., ANA) may be positive and elevated serological test results may take longer to resolve than clinical manifestations.
Drug Interactions
Serious adverse reactions have been reported in patients taking clarithromycin concomitantly with CYP3A4 substrates. These include colchicine toxicity with colchicine; rhabdomyolysis with simvastatin, lovastatin, and atorvastatin; hypoglycemia with disopyramide, and hypotension and acute kidney injury with calcium channel blockers metabolized by CYP3A4 (e.g., verapamil, amlodipine, diltiazem, nifedipine). Most reports of acute kidney injury with calcium channel blockers metabolized by CYP3A4 involved elderly patients 65 years of age or older (see CONTRAINDICATIONS and PRECAUTIONS, Drug Interactions). Clarithromycin should be used with caution when administered concurrently with medications that induce the cytochrome CYP3A4 enzyme (see PRECAUTIONS, Drug Interactions).
Colchicine
Life-threatening and fatal drug interactions have been reported in patients treated with clarithromycin and colchicine. Clarithromycin is a strong CYP3A4 inhibitor and this interaction may occur while using both drugs at their recommended doses. If coadministration of clarithromycin and colchicine is necessary in patients with normal renal and hepatic function, the dose of colchicine should be reduced. Patients should be monitored for clinical symptoms of colchicine toxicity. Concomitant administration of clarithromycin and colchicine is contraindicated in patients with renal or hepatic impairment (see CONTRAINDICATIONS and PRECAUTIONS, Drug Interactions).
Benzodiazepines
Increased sedation and prolongation of sedation have been reported with concomitant administration of clarithromycin and triazolobenzodiazepines, such as triazolam, and midazolam.
Oral Hypoglycemic Agents/Insulin
The concomitant use of clarithromycin and oral hypoglycemic agents and/or insulin can result in significant hypoglycemia. With certain hypoglycemic drugs such as nateglinide, pioglitazone, repaglinide and rosiglitazone, inhibition of CYP3A enzyme by clarithromycin may be involved and could cause hypoglycemia when used concomitantly. Careful monitoring of glucose is recommended.
Oral Anticoagulants
There is a risk of serious hemorrhage and significant elevations in INR and prothrombin time when clarithromycin is coadministered with warfarin. INR and prothrombin times should be frequently monitored while patients are receiving clarithromycin and oral anticoagulants concurrently.
HMG-CoA Reductase Inhibitors (statins)
Concomitant use of clarithromycin with lovastatin or simvastatin is contraindicated (see CONTRAINDICATIONS) as these statins are extensively metabolized by CYP3A4, and concomitant treatment with clarithromycin increases their plasma concentration, which increases the risk of myopathy, including rhabdomyolysis. Cases of rhabdomyolysis have been reported in patients taking clarithromycin concomitantly with these statins. If treatment with clarithromycin cannot be avoided, therapy with lovastatin or simvastatin must be suspended during the course of treatment.
Caution should be exercised when prescribing clarithromycin with statins. In situations where the concomitant use of clarithromycin with atorvastatin or pravastatin cannot be avoided, atorvastatin dose should not exceed 20 mg daily and pravastatin dose should not exceed 40 mg daily. Use of a statin that is not dependent on CYP3A metabolism (e.g., fluvastatin) can be considered. It is recommended to prescribe the lowest registered dose if concomitant use cannot be avoided.
Interactions with Investigations for Neuroendocrine Tumors
Serum chromogranin A (CgA) levels increase secondary to drug-induced decreases in gastric acidity. The increased CgA level may cause false positive results in diagnostic investigations for neuroendocrine tumors. Healthcare providers should temporarily stop lansoprazole treatment at least 14 days before assessing CgA levels and consider repeating the test if initial CgA levels are high. If serial tests are performed (e.g., for monitoring), the same commercial laboratory should be used for testing, as reference ranges between tests may vary (see CLINICAL PHARMACOLOGY, Drug Interaction Studies).
Interaction with Methotrexate
Literature suggests that concomitant use of PPIs with methotrexate (primarily at high dose) may elevate and prolong serum levels of methotrexate and/or its metabolite, possibly leading to methotrexate toxicities. In high-dose methotrexate administration, a temporary withdrawal of the PPI may be considered in some patients (see CLINICAL PHARMACOLOGY, Drug Interaction Studies).
Clostridium Difficile-Associated Diarrhea
Clostridium difficile-associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including clarithromycin and/or amoxicillin, and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibiotic use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibiotic use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibiotic treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
In addition, published observational studies suggest that PPI therapy, may be associated with an increased risk of CDAD, especially in hospitalized patients. This diagnosis should be considered for diarrhea that does not improve.
PRECAUTIONS
General
The possibility of superinfections with fungal or bacterial pathogens should be considered during therapy. If superinfections occur, PREVPAC should be discontinued and appropriate therapy instituted.
Prescribing PREVPAC either in the absence of a proven or strongly suspected bacterial infection is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria.
Clarithromycin is principally excreted via the liver and kidney. Clarithromycin may be administered without dosage adjustment to patients with hepatic impairment and normal renal function. However, in the presence of severe renal impairment with or without coexisting hepatic impairment, decreased dosage or prolonged dosing intervals may be appropriate.
Exacerbation of symptoms of myasthenia gravis and new onset of symptoms of myasthenic syndrome has been reported in patients receiving clarithromycin therapy.
Information for Patients
Each dose of PREVPAC contains four pills: one pink and black capsule (PREVACID), two opaque, yellow capsules (amoxicillin) and one yellow tablet (clarithromycin). Each dose should be taken twice per day before eating. Patients should be instructed to swallow each pill whole.
PREVPAC may interact with some drugs; therefore patients should be advised to report to their doctor the use of any other medications.
Patients should be counseled that antibacterial drugs including PREVPAC should only be used to treat bacterial infections. They do not treat viral infections (e.g., the common cold). When PREVPAC is prescribed to treat a bacterial infection, patients should be told that although it is common to feel better early in the course of therapy, the medication should be taken exactly as directed. Skipping doses or not completing the full course of therapy may (1) decrease the effectiveness of the immediate treatment and (2) increase the likelihood that bacteria will develop resistance and will not be treatable by PREVPAC or other antibacterial drugs in the future.
Diarrhea is a common problem caused by antibiotics which usually ends when the antibiotic is discontinued. Sometimes after starting treatment with antibiotics, patients can develop watery and bloody stools (with or without stomach cramps and fever) even as late as two or more months after having taken the last dose of the antibiotic. If this occurs, patients should contact their physician as soon as possible.
Patients should be advised to immediately report and seek care for diarrhea that does not improve. This may be a sign of Clostridium difficile-associated diarrhea (see WARNINGS).
Patients should be advised to immediately report and seek care for any cardiovascular or neurological symptoms including palpitations, dizziness, seizures, and tetany as these may be signs of hypomagnesemia (see WARNINGS).
Advise patients to report any symptoms associated with cutaneous or systemic lupus erythematosus (see WARNINGS).
Advise patients to report to their healthcare provider if they are taking rilpivirine-containing products or high-dose methotrexate (see WARNINGS).
Drug Interactions
No drug interaction studies have been conducted specifically with PREVPAC. The following drug interactions are for the individual drug components: PREVACID (lansoprazole), amoxicillin, and clarithromycin. Therefore, the decision to adjust dosage should depend on the clinician's assessment of among other things, the cumulative or net effect of the drug components of PREVPAC.
PREVACID
Tables 5 and 6 include drugs with clinically important drug interactions and interaction with diagnostics when administered concomitantly with PREVACID and instructions for preventing or managing them.
Consult the labeling of concomitantly used drugs to obtain further information about interactions with PPIs.
| Antiretrovirals | |
| Clinical Impact: | The effect of PPIs on antiretroviral drugs is variable. The clinical importance and the mechanisms behind these interactions are not always known.
|
| Intervention: | Rilpivirine-containing products: Concomitant use with PREVACID is contraindicated (see CONTRAINDICATIONS). See prescribing information. Atazanavir: See prescribing information for atazanavir for dosing information. Nelfinavir: Avoid concomitant use with PREVACID. See prescribing information for nelfinavir. Saquinavir: See the prescribing information for saquinavir and monitor for potential saquinavir toxicities. Other antiretrovirals: See prescribing information. |
| Warfarin | |
| Clinical Impact: | Increased INR and prothrombin time in patients receiving PPIs and warfarin concomitantly. Increases in INR and prothrombin time may lead to abnormal bleeding and even death. |
| Intervention: | Monitor INR and prothrombin time. Dose adjustment of warfarin may be needed to maintain target INR range. See prescribing information for warfarin. |
| Methotrexate | |
| Clinical Impact: | Concomitant use of PPIs with methotrexate (primarily at high dose) may elevate and prolong serum concentrations of methotrexate and/or its metabolite hydroxymethotrexate, possibly leading to methotrexate toxicities. No formal drug interaction studies of high-dose methotrexate with PPIs have been conducted (see WARNINGS). |
| Intervention: | A temporary withdrawal of PREVACID may be considered in some patients receiving high-dose methotrexate. |
| Digoxin | |
| Clinical Impact: | Potential for increased exposure of digoxin. |
| Intervention: | Monitor digoxin concentrations. Dose adjustment of digoxin may be needed to maintain therapeutic drug concentrations. See prescribing information for digoxin. |
| Theophylline | |
| Clinical Impact: | Increased clearance of theophylline (see CLINICAL PHARMACOLOGY). |
| Intervention: | Individual patients may require additional titration of their theophylline dosage when PREVACID is started or stopped to ensure clinically effective blood concentrations. |
| Drugs Dependent on Gastric pH for Absorption (e.g., iron salts, erlotinib, dasatinib, nilotinib, mycophenolate mofetil, ketoconazole/itraconazole) | |
| Clinical Impact: | Lansoprazole can reduce the absorption of other drugs due to its effect on reducing intragastric acidity. |
| Intervention: | Mycophenolate mofetil (MMF): Coadministration of PPIs in healthy subjects and in transplant patients receiving MMF has been reported to reduce the exposure to the active metabolite, mycophenolic acid (MPA), possibly due to a decrease in MMF solubility at an increased gastric pH. The clinical relevance of reduced MPA exposure on organ rejection has not been established in transplant patients receiving PREVACID and MMF. Use PREVACID with caution in transplant patients receiving MMF. See the prescribing information for other drugs dependent on gastric pH for absorption. |
| Combination Therapy with Clarithromycin and Amoxicillin | |
| Clinical Impact: | Concomitant administration of clarithromycin with other drugs can lead to serious adverse reactions, including potentially fatal arrhythmias, and are contraindicated. Amoxicillin also has drug interactions. |
| Intervention: |
|
| Tacrolimus | |
| Clinical Impact: | Potentially increased exposure of tacrolimus, especially in transplant patients who are intermediate or poor metabolizers of CYP2C19. |
| Intervention: | Monitor tacrolimus whole blood trough concentrations. Dose adjustment of tacrolimus may be needed to maintain therapeutic drug concentrations. See prescribing information for tacrolimus. |
| Interactions with Investigations of Neuroendocrine Tumors | |
| Clinical Impact: | CgA levels increase secondary to PPI-induced decreases in gastric acidity. The increased CgA level may cause false positive results in diagnostic investigations for neuroendocrine tumors (see WARNINGS, CLINICAL PHARMACOLOGY). |
| Intervention: | Temporarily stop PREVACID treatment at least 14 days before assessing CgA levels and consider repeating the test if initial CgA levels are high. If serial tests are performed (e.g., for monitoring), the same commercial laboratory should be used for testing, as reference ranges between tests may vary. |
| Interaction with Secretin Stimulation Test | |
| Clinical Impact: | Hyper-response in gastrin secretion in response to secretin stimulation test, falsely suggesting gastrinoma. |
| Intervention: | Temporarily stop PREVACID treatment at least 28 days before assessing to allow gastrin levels to return to baseline (see CLINICAL PHARMACOLOGY). |
| False Positive Urine Tests for THC | |
| Clinical Impact: | There have been reports of false positive urine screening tests for tetrahydrocannabinol (THC) in patients receiving PPIs. |
| Intervention: | An alternative confirmatory method should be considered to verify positive results. |
| CYP2C19 OR CYP3A4 Inducers | |
| Clinical Impact: | Decreased exposure of lansoprazole when used concomitantly with strong inducers (see CLINICAL PHARMACOLOGY). |
| Intervention: | St John's Wort, rifampin: Avoid concomitant use with PREVACID. Ritonavir-containing products: See prescribing information. |
| CYP2C19 or CYP3A4 Inhibitors | |
| Clinical Impact: | Increased exposure of lansoprazole is expected when used concomitantly with strong inhibitors (see CLINICAL PHARMACOLOGY). |
| Intervention: | Voriconazole: See prescribing information. |
| Sucralfate | |
| Clinical Impact: | Decreased and delayed absorption of lansoprazole (see CLINICAL PHARMACOLOGY). |
| Intervention: | Take PREVACID at least 30 minutes prior to sucralfate (see CLINICAL PHARMACOLOGY). |
Amoxicillin
Probenecid
Probenecid decreases the renal tubular secretion of amoxicillin. Concurrent use of amoxicillin and probenecid may result in increased and prolonged blood levels of amoxicillin.
Antibiotics
Chloramphenicol, macrolides, sulfonamides, and tetracyclines may interfere with bactericidal effects of penicillin. This has been demonstrated in vitro; however, the clinical significance of this interaction is not well documented.
Amoxicillin may affect the gut flora, leading to lower estrogen reabsorption and reduced efficacy of combined oral estrogen/progesterone contraceptives.
| Drugs That Are Affected By BIAXIN | ||
| Drug(s) with Pharmacokinetics Affected by BIAXIN | Recommendation | Comments |
| Antiarrhythmics: | ||
| Disopyramide Quinidine Dofetilide Amiodarone Sotalol Procainamide | Not Recommended | Disopyramide, Quinidine: There have been postmarketing reports of torsades de pointes occurring with concurrent use of clarithromycin and quinidine or disopyramide. Electrocardiograms should be monitored for QTc prolongation during coadministration of clarithromycin with these drugs (see WARNINGS). Serum concentrations of these medications should also be monitored. There have been spontaneous or published reports of CYP3A based interactions of clarithromycin with disopyramide and quinidine. There have been postmarketing reports of hypoglycemia with the concomitant administration of clarithromycin and disopyramide. Therefore, blood glucose levels should be monitored during concomitant administration of clarithromycin and disopyramide. |
| Digoxin | Use With Caution | Digoxin: Digoxin is a substrate for P-glycoprotein (Pgp) and clarithromycin is known to inhibit Pgp. When clarithromycin and digoxin are coadministered, inhibition of Pgp by clarithromycin may lead to increased exposure of digoxin. Elevated digoxin serum concentrations in patients receiving clarithromycin and digoxin concomitantly have been reported in postmarketing surveillance. Some patients have shown clinical signs consistent with digoxin toxicity, including potentially fatal arrhythmias. Monitoring of serum digoxin concentrations should be considered, especially for patients with digoxin concentrations in the upper therapeutic range. |
| Oral Anticoagulants: | ||
| Warfarin | Use With Caution | Oral anticoagulants: Spontaneous reports in the postmarketing period suggest that concomitant administration of clarithromycin and oral anticoagulants may potentiate the effects of the oral anticoagulants. Prothrombin times should be carefully monitored while patients are receiving clarithromycin and oral anticoagulants simultaneously (see WARNINGS). |
| Antiepileptics: | ||
| Carbamazepine | Use With Caution | Carbamazepine: Concomitant administration of single doses of clarithromycin and carbamazepine has been shown to result in increased plasma concentrations of carbamazepine. Blood level monitoring of carbamazepine may be considered. Increased serum concentrations of carbamazepine were observed in clinical trials with clarithromycin. There have been spontaneous or published reports of CYP3A based interactions of clarithromycin with carbamazepine. |
| Antifungals: | ||
| Itraconazole | Use With Caution |
Itraconazole: Both clarithromycin and itraconazole are substrates and inhibitors of CYP3A, potentially leading to a bi-directional drug interaction when administered concomitantly (see also Itraconazole under "Drugs That Affect BIAXIN" in the table below). Clarithromycin may increase the plasma concentrations of itraconazole. Patients taking itraconazole and clarithromycin concomitantly should be monitored closely for signs or symptoms of increased or prolonged adverse reactions. |
| Fluconazole | No Dose Adjustment | Fluconazole: No dosage adjustment of clarithromycin is necessary when coadministered with fluconazole. |
| Anti-Gout Agents: | ||
| Colchicine (in patients with renal or hepatic impairment) | Contraindicated | Colchicine: Colchicine is a substrate for both CYP3A and the efflux transporter, P-glycoprotein (Pgp). Clarithromycin and other macrolides are known to inhibit CYP3A and Pgp. The dose of colchicine should be reduced when coadministered with clarithromycin in patients with normal renal and hepatic function (see CONTRAINDICATIONS, WARNINGS). |
| Colchicine (in patients with normal renal and hepatic function) | Use With Caution | |
| Antipsychotics: | ||
| Pimozide | Contraindicated | Pimozide: (see CONTRAINDICATIONS). |
| Quetiapine | Quetiapine: Quetiapine is a substrate for CYP3A4, which is inhibited by clarithromycin. Coadministration with clarithromycin could result in increased quetiapine exposure and possible quetiapine related toxicities. There have been postmarketing reports of somnolence, orthostatic hypotension, altered state of consciousness, neuroleptic malignant syndrome, and QT prolongation during concomitant administration. Refer to quetiapine prescribing information for recommendations on dose reduction if coadministered with CYP3A4 inhibitors such as clarithromycin. | |
| Antispasmodics: | ||
| Tolterodine (patients deficient in CYP2D6 activity) | Use With Caution | Tolterodine: The primary route of metabolism for tolterodine is via CYP2D6. However, in a subset of the population devoid of CYP2D6, the identified pathway of metabolism is via CYP3A. In this population subset, inhibition of CYP3A results in significantly higher serum concentrations of tolterodine. Tolterodine 1 mg twice daily is recommended in patients deficient in CYP2D6 activity (poor metabolizers) when coadministered with clarithromycin. |
| Antivirals: | ||
| Atazanavir | Use With Caution | Atazanavir: Both clarithromycin and atazanavir are substrates and inhibitors of CYP3A, and there is evidence of a bi-directional drug interaction (see Atazanavir under "Drugs That Affect BIAXIN" in the table below). |
| Saquinavir (in patients with decreased renal function) | Saquinavir: Both clarithromycin and saquinavir are substrates and inhibitors of CYP3A and there is evidence of a bi-directional drug interaction (see Saquinavir under "Drugs That Affect BIAXIN" in the table below). | |
| Ritonavir Etravirine | Ritonavir, Etravirine: (see Ritonavir and Etravirine under "Drugs That Affect BIAXIN" in the table below). | |
| Maraviroc | Maraviroc: Clarithromycin may result in increases in maraviroc exposures by inhibition of CYP3A metabolism. See Selzentry® prescribing information for dose recommendation when given with strong CYP3A inhibitors such as clarithromycin. | |
| Boceprevir (in patients with normal renal function) | No Dose Adjustment | Boceprevir: Both clarithromycin and boceprevir are substrates and inhibitors of CYP3A, potentially leading to a bi-directional drug interaction when coadministered. No dose adjustments are necessary for patients with normal renal function (see Victrelis® prescribing information). |
| Didanosine | ||
| Zidovudine | Zidovudine: Simultaneous oral administration of clarithromycin immediate-release tablets and zidovudine to HIV-infected adult patients may result in decreased steady-state zidovudine concentrations. Administration of clarithromycin and zidovudine should be separated by at least two hours. The impact of coadministration of clarithromycin extended-release tablets or granules and zidovudine has not been evaluated. |
|
| Calcium Channel Blockers: | ||
| Verapamil | Use With Caution | Verapamil: Hypotension, bradyarrhythmias, and lactic acidosis have been observed in patients receiving concurrent verapamil (see WARNINGS). |
| Amlodipine Diltiazem | Amlodipine, Diltiazem: (see WARNINGS). | |
| Nifedipine | Nifedipine: Nifedipine is a substrate for CYP3A. Clarithromycin and other macrolides are known to inhibit CYP3A. There is potential of CYP3A-mediated interaction between nifedipine and clarithromycin. Hypotension and peripheral edema were observed when clarithromycin was taken concomitantly with nifedipine (see WARNINGS). | |
| Ergot Alkaloids: | ||
| Ergotamine Dihydroergotamine | Contraindicated | Ergotamine, Dihydroergotamine: Postmarketing reports indicate that coadministration of clarithromycin with ergotamine or dihydroergotamine has been associated with acute ergot toxicity characterized by vasospasm and ischemia of the extremities and other tissues including the central nervous system (see CONTRAINDICATIONS). |
| Gastroprokinetic Agents: | ||
| Cisapride | Contraindicated | Cisapride: (see CONTRAINDICATIONS). |
| HMG-CoA Reductase Inhibitors: | ||
| Lovastatin Simvastatin | Contraindicated | Lovastatin, Simvastatin, Atorvastatin, Pravastatin, Fluvastatin: (see CONTRAINDICATIONS, WARNINGS). |
| Atorvastatin Pravastatin | Use With Caution | |
| Fluvastatin | No Dose Adjustment | |
| Hypoglycemic Agents: | ||
| Nateglinide Pioglitazone Repaglinide Rosiglitazone | Use With Caution | Nateglinide, Pioglitazone, Repaglinide, Rosiglitazone: (see WARNINGS, ADVERSE REACTIONS). |
| Insulin | Insulin: (see WARNINGS, ADVERSE REACTIONS). | |
| Immunosuppressants: | ||
| Cyclosporine | Use With Caution | Cyclosporine: There have been spontaneous or published reports of CYP3A based interactions of clarithromycin with cyclosporine. |
| Tacrolimus | Tacrolimus: There have been spontaneous or published reports of CYP3A based interactions of clarithromycin with tacrolimus. | |
| Phosphodiesterase Inhibitors: | ||
| Sildenafil Tadalafil Vardenafil | Use With Caution | Sildenafil, Tadalafil, Vardenafil: Each of these phosphodiesterase inhibitors is primarily metabolized by CYP3A, and CYP3A will be inhibited by concomitant administration of clarithromycin. Coadministration of clarithromycin with sildenafil, tadalafil, or vardenafil will result in increased exposure of these phosphodiesterase inhibitors. Coadministration of these phosphodiesterase inhibitors with clarithromycin is not recommended. Increased systemic exposure of these drugs may occur with clarithromycin; reduction of dosage for phosphodiesterase inhibitors should be considered (see their respective prescribing information). |
| Proton Pump Inhibitors: | ||
| Omeprazole | No Dose Adjustment | Omeprazole: The mean 24 hour gastric pH value was 5.2 when omeprazole was administered alone and 5.7 when coadministered with clarithromycin as a result of increased omeprazole exposures (see also Omeprazole under "Drugs That Affect BIAXIN" in the table below). |
| Xanthine Derivatives: | ||
| Theophylline | Use With Caution | Theophylline: Clarithromycin use in patients who are receiving theophylline may be associated with an increase of serum theophylline concentrations. Monitoring of serum theophylline concentrations should be considered for patients receiving high doses of theophylline or with baseline concentrations in the upper therapeutic range. |
| Triazolobenzodiazepines and Other Related Benzodiazepines: | ||
| Midazolam | Use With Caution | Midazolam: When oral midazolam is coadministered with clarithromycin, dose adjustments may be necessary and possible prolongation and intensity of effect should be anticipated (see WARNINGS). |
| Alprazolam Triazolam | Triazolam, Alprazolam: Caution and appropriate dose adjustments should be considered when triazolam or alprazolam is coadministered with clarithromycin. There have been postmarketing reports of drug interactions and central nervous system (CNS) effects (e.g., somnolence and confusion) with the concomitant use of clarithromycin and triazolam. Monitoring the patient for increased CNS pharmacological effects is suggested. In postmarketing experience, erythromycin has been reported to decrease the clearance of triazolam and midazolam, and thus, may increase the pharmacologic effect of these benzodiazepines. |
|
| Temazepam Nitrazepam Lorazepam | No Dose Adjustment | Temazepam, Nitrazepam, Lorazepam: For benzodiazepines which are not metabolized by CYP3A (e.g., temazepam, nitrazepam, lorazepam), a clinically important interaction with clarithromycin is unlikely. |
| Cytochrome P450 Inducers: | ||
| Rifabutin | Use With Caution |
Rifabutin: Concomitant administration of rifabutin and clarithromycin resulted in an increase in rifabutin, and decrease in clarithromycin serum levels together with an increased risk of uveitis (see Rifabutin under "Drugs That Affect BIAXIN" in the table below). |
| Other Drugs Metabolized by CYP3A: | ||
| Alfentanil Bromocriptine Cilostazol Methylprednisolone Vinblastine Phenobarbital St. John's Wort | Use With Caution | There have been spontaneous or published reports of CYP3A based interactions of clarithromycin with alfentanil, methylprednisolone, cilostazol, bromocriptine, vinblastine, phenobarbital, and St. John's Wort. |
| Other Drugs Metabolized by CYP450 Isoforms Other than CYP3A: | ||
| Hexobarbital Phenytoin Valproate | Use With Caution | There have been postmarketing reports of interactions of clarithromycin with drugs not thought to be metabolized by CYP3A, including hexobarbital, phenytoin, and valproate. |
| Drugs That Affect BIAXIN | ||
| Drug(s) that Affect the Pharmacokinetics of BIAXIN | Recommendation | Comments |
| Antifungals: | ||
| Itraconazole | Use With Caution |
Itraconazole: Itraconazole may increase the plasma concentrations of clarithromycin. Patients taking itraconazole and clarithromycin concomitantly should be monitored closely for signs or symptoms of increased or prolonged adverse reactions (see also Itraconazole under "Drugs That Are Affected By BIAXIN" in the table above). |
| Antivirals: | ||
| Atazanavir | Use With Caution |
Atazanavir: When clarithromycin is coadministered with atazanavir, the dose of clarithromycin should be decreased by 50%. |
| Ritonavir (in patients with decreased renal function) |
Ritonavir: Since concentrations of 14-OH clarithromycin are significantly reduced when clarithromycin is coadministered with ritonavir, alternative antibacterial therapy should be considered for indications other than infections due to Mycobacterium avium. |
|
| Saquinavir (in patients with decreased renal function) |
Saquinavir: When saquinavir is coadministered with ritonavir, consideration should be given to the potential effects of ritonavir on clarithromycin (refer to ritonavir above). |
|
| Etravirine |
Etravirine: Clarithromycin exposure was decreased by etravirine; however, concentrations of the active metabolite, 14-OH-clarithromycin, were increased. Because 14-OH-clarithromycin has reduced activity against Mycobacterium avium complex (MAC), overall activity against this pathogen may be altered; therefore alternatives to clarithromycin should be considered for the treatment of MAC. |
|
| Saquinavir (in patients with normal renal function) Ritonavir (in patients with normal renal function) | No Dose Adjustment | |
| Proton Pump Inhibitors: | ||
| Omeprazole | Use With Caution |
Omeprazole: Clarithromycin concentrations in the gastric tissue and mucus were also increased by concomitant administration of omeprazole. |
| Miscellaneous Cytochrome P450 Inducers: | ||
| Efavirenz Nevirapine Rifampicin Rifabutin Rifapentine | Use With Caution |
Inducers of CYP3A enzymes, such as efavirenz, nevirapine, rifampicin, rifabutin, and rifapentine will increase the metabolism of clarithromycin, thus decreasing plasma concentrations of clarithromycin, while increasing those of 14-OH-clarithromycin. Since the microbiological activities of clarithromycin and 14-OH-clarithromycin are different for different bacteria, the intended therapeutic effect could be impaired during concomitant administration of clarithromycin and enzyme inducers. Alternative antibacterial treatment should be considered when treating patients receiving inducers of CYP3A. There have been spontaneous or published reports of CYP3A based interactions of clarithromycin with rifabutin (see Rifabutin under "Drugs That Are Affected By BIAXIN" in the table above). |
Drug/Laboratory Test Interactions
High urine concentrations of ampicillin may result in false-positive reactions when testing for the presence of glucose in urine using CLINITEST, Benedict's Solution or Fehling's Solution. Since this effect may also occur with amoxicillin, it is recommended that glucose tests based on enzymatic glucose oxidase reactions (such as CLINISTIX) be used.
Following administration of ampicillin or amoxicillin to pregnant women, a transient decrease in plasma concentration of total conjugated estriol, estriol-glucuronide, conjugated estrone, and estradiol has been noted.
Carcinogenesis, Mutagenesis, Impairment of Fertility
PREVACID
In two 24-month carcinogenicity studies, Sprague-Dawley rats were treated with oral lansoprazole at doses of 5 to 150 mg/kg/day, about 0.5 to 20 times the recommended human dose of 60 mg/day, based on body surface area (BSA). Lansoprazole produced dose-related gastric enterochromaffin-like (ECL) cell hyperplasia and ECL cell carcinoids in both male and female rats. The incidences of intestinal metaplasia of the gastric epithelium were also increased in both sexes. In male rats, lansoprazole produced a dose-related increase in the incidence of testicular interstitial cell adenomas at doses two to 20 times the recommended human dose of 60 mg/day based on BSA.
In a 24-month carcinogenicity study, CD-1 mice were treated with oral lansoprazole at doses of 15 to 600 mg/kg/day (one to 40 times the recommended human dose of 60 mg/day based on BSA comparisons). Lansoprazole produced a dose-related increased incidence of gastric ECL cell hyperplasia. The incidence of liver tumors (hepatocellular adenoma plus carcinoma) was increased in male mice (at doses 20 to 40 times the recommended human dose of 60 mg/day based on BSA) and in female mice (treated at doses ten to 40 times the recommended human dose based on BSA). Lansoprazole treatment produced adenoma of rete testis in male mice receiving doses five to 40 times the recommended human dose of 60 mg/day based on BSA.
A 26 week p53 (+/-) transgenic mouse carcinogenicity study was not positive.
Lansoprazole was positive in the Ames test and the in vitro human lymphocyte chromosomal aberration assay. Lansoprazole was not genotoxic in the ex vivo rat hepatocyte unscheduled DNA synthesis (UDS) test, the in vivo mouse micronucleus test, or the rat bone marrow cell chromosomal aberration test.
Lansoprazole at oral doses up to 150 mg/kg/day (20 times the recommended human dose of 60 mg/day based on BSA) was found to have no effect on fertility and reproductive performance of male and female rats.
Amoxicillin
Long-term studies in animals have not been performed to evaluate the mutagenic or carcinogenic potential of amoxicillin alone. A 4:1 mixture of amoxicillin and potassium clavulanate was non-mutagenic in the Ames bacterial mutation assay, and the yeast gene conversion assay. The amoxicillin/potassium clavulanate mixture was also negative in the mouse micronucleus test, and in the dominant lethal assay in mice, but was weakly positive in the mouse lymphoma assay. In a multi-generation reproduction study in rats, no impairment of fertility or other adverse reproductive effects were seen at doses up to 500 mg/kg, approximately three times the human dose based on BSA comparisons.
Clarithromycin
The following in vitro mutagenicity tests have been conducted with clarithromycin:
- Salmonella/Mammalian Microsomes Test
- Bacterial Induced Mutation Frequency Test
- In Vitro Chromosome Aberration Test
- Rat Hepatocyte DNA Synthesis Assay
- Mouse Lymphoma Assay
- Mouse Dominant Lethal Study
- Mouse Micronucleus Test
All tests had negative results except the In Vitro Chromosome Aberration Test which was weakly positive in one test and negative in another.
In addition, a Bacterial Reverse-Mutation Test (Ames Test) has been performed on clarithromycin metabolites with negative results.
Fertility and reproduction studies have shown that daily doses of up to 160 mg/kg/day (1.3 times the recommended maximum human dose based on mg/m2) to male and female rats caused no adverse effects on the estrous cycle, fertility, parturition, or number and viability of offspring. Plasma levels in rats after 150 mg/kg/day were two times the human serum levels.
In the 150 mg/kg/day monkey studies, plasma levels were three times the human serum levels. When given orally at 150 mg/kg/day (2.4 times the recommended maximum human dose based on mg/m2), clarithromycin was shown to produce embryonic loss in monkeys. This effect has been attributed to marked maternal toxicity of the drug at this high dose.
In rabbits, in utero fetal loss occurred at an intravenous dose of 33 mg/m2, which is 17 times less than the maximum proposed human oral daily dose of 618 mg/m2.
Long-term studies in animals have not been performed to evaluate the carcinogenic potential of clarithromycin.
Pregnancy
Teratogenic Effects
Pregnancy Category C
Category C is based on the pregnancy category for clarithromycin.
There are no adequate and well-controlled studies of lansoprazole, clarithromycin or amoxicillin (used separately or together) in pregnant women. PREVPAC should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus and there is no appropriate alternative therapy (see WARNINGS).
Lansoprazole
No adverse effects on embryo-fetal development occurred in studies performed in pregnant rats at oral lansoprazole doses up to 150 mg/kg/day (40 times the recommended human dose [30 mg/day] based on body surface area) administered during organogenesis and pregnant rabbits at oral lansoprazole doses up to 30 mg/kg/day (16 times the recommended human dose based on body surface area) administered during organogenesis.
A pre- and postnatal developmental toxicity study in rats with additional endpoints to evaluate bone development was performed with lansoprazole at oral doses of 10 to 100 mg/kg/day (0.7 to 6.4 times the maximum recommended human lansoprazole dose of 30 mg based on AUC [area under the plasma concentration-time curve]) administered during organogenesis through lactation. Maternal effects observed at 100 mg/kg/day (6.4 times the maximum recommended human lansoprazole dose of 30 mg based on AUC) included increased gestation period, decreased body weight gain during gestation, and decreased food consumption. The number of stillbirths was increased at this dose, which may have been secondary to maternal toxicity. Body weight of pups was reduced at 100 mg/kg/day starting on postnatal Day 11. Femur weight, femur length, and crown-rump length were reduced at 100 mg/kg/day on postnatal Day 21. Femur weight was still decreased in the 100 mg/kg/day group at age 17 to 18 weeks. Growth plate thickness was decreased in the 100 mg/kg/day males on postnatal Day 21, and was increased in the 30 and 100 mg/kg/day males at age 17 to 18 weeks. The effects on bone parameters were associated with reduction in body weight gain.
Amoxicillin
Reproduction studies with amoxicillin have been performed in mice and rats at doses up to ten times the human dose and revealed no evidence of impaired fertility or harm to the fetus.
Clarithromycin
Four teratogenicity studies in rats with clarithromycin (three with oral doses and one with intravenous doses up to 160 mg/kg/day administered during the period of major organogenesis) and two in rabbits at oral doses up to 125 mg/kg/day (approximately two times the recommended maximum human dose based on mg/m2) or intravenous doses of 30 mg/kg/day administered during gestation days six to 18 failed to demonstrate any teratogenicity from clarithromycin. Two additional oral studies in a different rat strain at similar doses and similar conditions demonstrated a low incidence of cardiovascular anomalies at doses of 150 mg/kg/day administered during gestation days six to 15. Plasma levels after 150 mg/kg/day were two times the human serum levels. Four studies in mice revealed a variable incidence of cleft palate following oral doses of 1000 mg/kg/day (two and four times the recommended maximum human dose based on mg/m2, respectively) during gestation days six to 15. Cleft palate was also seen at 500 mg/kg/day. The 1000 mg/kg/day exposure resulted in plasma levels 17 times the human serum levels. In monkeys, an oral dose of 70 mg/kg/day (an approximate equidose of the recommended maximum human dose based on mg/m2) produced fetal growth retardation at plasma levels that were two times the human serum levels.
Labor and Delivery
Oral ampicillin-class antibiotics are poorly absorbed during labor. Studies in guinea pigs showed that intravenous administration of ampicillin slightly decreased the uterine tone and frequency of contractions, but moderately increased the height and duration of contractions. However, it is not known whether use of these drugs in humans during labor or delivery has immediate or delayed adverse effects on the fetus, prolongs the duration of labor, or increases the likelihood that forceps delivery or other obstetrical intervention or resuscitation of the newborn will be necessary.
Nursing Mothers
Lansoprazole and its metabolites are excreted in the milk of rats. It is not known whether lansoprazole is excreted in human milk. Because of the potential for serious adverse reactions in nursing infants from PREVPAC, and the potential for tumorigenicity shown for lansoprazole in rat carcinogenicity studies, a decision should be made whether to discontinue nursing or to discontinue PREVPAC, taking into account the importance of the therapy to the mother.
Penicillins have been shown to be excreted in human milk. Amoxicillin use by nursing mothers may lead to sensitization of infants. Caution should be exercised when amoxicillin is administered to a nursing woman.
Clarithromycin and its active metabolite 14-hydroxy clarithromycin are excreted in human milk. Serum and milk samples were obtained after three days of treatment, at steady state, from one published study of 12 lactating women who were taking clarithromycin 250 mg orally twice daily. Based on the limited data from this study, and assuming milk consumption of 150 mL/kg/day, an exclusively human milk-fed infant would receive an estimated average of 136 mcg/kg/day of clarithromycin and its active metabolite, with this maternal dosage regimen. This is less than 2% of the maternal weight-adjusted dose (7.8 mg/kg/day, based on the average maternal weight of 64 kg), and less than 1% of the pediatric dose (15 mg/kg/day) for children greater than six months of age.
A prospective observational study of 55 breastfed infants of mothers taking a macrolide antibiotic (six were exposed to clarithromycin) were compared to 36 breastfed infants of mothers taking amoxicillin. Adverse reactions were comparable in both groups. Adverse reactions occurred in 12.7% of infants exposed to macrolides and included rash, diarrhea, loss of appetite, and somnolence.
Caution should be exercised when clarithromycin is administered to nursing women. The development and health benefits of human milk feeding should be considered along with the mother's clinical need for clarithromycin and any potential adverse effects on the human milk-fed child from the drug or from the underlying maternal condition.
Pediatric Use
The safety and effectiveness of PREVPAC in pediatric patients infected with H. pylori have not been established.
Juvenile Animal Toxicity Data
In a juvenile rat study, adverse effects on bone growth and development and heart valves were observed at lansoprazole doses higher than the maximum recommended equivalent human dose.
An eight-week oral toxicity study with a four-week recovery phase was conducted in juvenile rats, with lansoprazole administered from postnatal Day 7 (age equivalent to neonatal humans) through 62 (age equivalent to approximately 14 years in humans) at doses of 40 to 500 mg/kg/day (about 1.2 to 12 times the daily pediatric dose of 15 mg in children age one to 11 years weighing 30 kg or less, based on AUC).
Heart valve thickening occurred at a dose of 500 mg/kg/day (approximately 12 times the daily dose of 15 mg in pediatric patients age one to 11 years weighing 30 kg or less, based on AUC). Heart valve thickening was not observed at the next lower dose (250 mg/kg/day) and below. The findings trended towards reversibility after a four-week drug-free recovery period. The relevance of heart valve thickening in this study to pediatric patients less than approximately 12 years of age is unknown. These findings are not relevant for patients 12 years of age and older. No effects on heart valves were observed in a 13-week intravenous toxicity study of lansoprazole in adolescent rats (approximately 12 years human age equivalence) at systemic exposures similar to those achieved in the eight-week oral toxicity study in juvenile (neonatal) rats.
In the eight-week oral toxicity study, doses equal to or greater than 100 mg/kg/day (about 2.5 times the daily pediatric dose of 15 mg in children age one to 11 years weighing 30 kg or less, based on AUC) produced delayed growth, with impairment of weight gain observed as early as postnatal Day 10 (age equivalent to neonatal humans). At the end of treatment, the signs of impaired growth at 100 mg/kg/day and higher included reductions in body weight (14% to 44% compared to controls), absolute weight of multiple organs, femur weight, femur length, and crown-rump length. Femoral growth plate thickness was reduced only in males and only at the 500 mg/kg/day dose. The effects related to delayed growth persisted through the end of the 4-week recovery period. Longer term data were not collected.
Geriatric Use
Elderly patients may suffer from asymptomatic renal and hepatic dysfunction. Care should be taken when administering PREVPAC to this patient population.
An analysis of clinical studies of amoxicillin was conducted to determine whether subjects aged 65 and over respond differently from younger subjects. These analyses have not identified differences in responses between the elderly and younger patients, but a greater sensitivity of some older individuals cannot be ruled out.
Amoxicillin is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.
In a steady-state study in which healthy elderly subjects (age 65 to 81 years old) were given 500 mg of clarithromycin every 12 hours, the maximum serum concentrations and area under the curves of clarithromycin and 14-OH clarithromycin were increased compared to those achieved in healthy young adults. These changes in pharmacokinetics parallel known age-related decreases in renal function. In clinical trials of clarithromycin, elderly patients did not have an increased incidence of adverse events when compared to younger patients. Dosage adjustment should be considered in elderly patients with severe renal impairment. Elderly patients may be more susceptible to development of torsades de pointes arrhythmias than younger patients (see WARNINGS and PRECAUTIONS).
Most reports of acute kidney injury with calcium channel blockers metabolized by CYP3A4 (e.g., verapamil, amlodipine, diltiazem, nifedipine) involved elderly patients 65 years of age or older (see WARNINGS).
ADVERSE REACTIONS
PREVPAC
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
The most common adverse reactions (≥3%) reported in clinical trials when all three components of this therapy were given concomitantly for 14 days are listed in Table 8.
| Triple Therapy | |
|---|---|
| Adverse Reaction | n=138 (%) |
| Diarrhea | 7.0 |
| Headache | 6.0 |
| Taste Perversion | 5.0 |
The additional adverse reactions which were reported as possibly or probably related to treatment (less than 3%) in clinical trials when all three components of this therapy were given concomitantly are listed below and divided by body system:
Body as a Whole - abdominal pain
Digestive System - dark stools, dry mouth/thirst, glossitis, rectal itching, nausea, oral moniliasis, stomatitis, tongue discoloration, tongue disorder, vomiting
Musculoskeletal System - myalgia
Nervous System - confusion, dizziness
Respiratory System - respiratory disorders
Skin and Appendages - skin reactions
Urogenital System - vaginitis, vaginal moniliasis
There were no statistically significant differences in the frequency of reported adverse events between the 10 and 14 day triple therapy regimens.
PREVACID
The following adverse reactions from the labeling for PREVACID are provided for information:
Worldwide, over 10,000 patients have been treated with PREVACID in Phase 2 or Phase 3 clinical trials involving various dosages and durations of treatment. In general, PREVACID treatment has been well-tolerated in both short-term and long-term trials.
Incidence in Clinical Trials
The following adverse events were reported by the treating physician to have a possible or probable relationship to drug in 1% or more of PREVACID-treated patients and occurred at a greater rate in PREVACID-treated patients than placebo-treated patients:
| Body System/Adverse Event | PREVACID (N= 2768) % | Placebo (N= 1023) % |
|---|---|---|
| Body as a Whole | ||
| Abdominal Pain | 2.1 | 1.2 |
| Digestive System | ||
| Constipation | 1.0 | 0.4 |
| Diarrhea | 3.8 | 2.3 |
| Nausea | 1.3 | 1.2 |
Headache was also seen at greater than 1% incidence but was more common on placebo. The incidence of diarrhea was similar between patients who received placebo and patients who received 30 mg of PREVACID, but higher in the patients who received 60 mg of PREVACID (2.9%, 4.2%, and 7.4%, respectively).
The most commonly reported possibly or probably treatment-related adverse event during maintenance therapy was diarrhea.
Additional adverse experiences occurring in less than 1% of patients or subjects who received PREVACID in domestic trials are shown below:
Body as a Whole – abdomen enlarged, allergic reaction, asthenia, back pain, candidiasis, carcinoma, chest pain (not otherwise specified), chills, edema, fever, flu syndrome, halitosis, infection (not otherwise specified), malaise, neck pain, neck rigidity, pain, pelvic pain
Cardiovascular System – angina, arrhythmia, bradycardia, cerebrovascular accident/cerebral infarction, hypertension/hypotension, migraine, myocardial infarction, palpitations, shock (circulatory failure), syncope, tachycardia, vasodilation
Digestive System – abnormal stools, anorexia, bezoar, cardiospasm, cholelithiasis, colitis, dry mouth, dyspepsia, dysphagia, enteritis, eructation, esophageal stenosis, esophageal ulcer, esophagitis, fecal discoloration, flatulence, gastric nodules/fundic gland polyps, gastritis, gastroenteritis, gastrointestinal anomaly, gastrointestinal disorder, gastrointestinal hemorrhage, glossitis, gum hemorrhage, hematemesis, increased appetite, increased salivation, melena, mouth ulceration, nausea and vomiting, nausea and vomiting and diarrhea, gastrointestinal moniliasis, rectal disorder, rectal hemorrhage, stomatitis, tenesmus, thirst, tongue disorder, ulcerative colitis, ulcerative stomatitis
Endocrine System – diabetes mellitus, goiter, hypothyroidism
Hemic and Lymphatic System – anemia, hemolysis, lymphadenopathy
Metabolism and Nutritional Disorders – avitaminosis, gout, dehydration, hyperglycemia/hypoglycemia, peripheral edema, weight gain/loss
Musculoskeletal System – arthralgia, arthritis, bone disorder, joint disorder, leg cramps, musculoskeletal pain, myalgia, myasthenia, ptosis, synovitis
Nervous System – abnormal dreams, agitation, amnesia, anxiety, apathy, confusion, convulsion, dementia, depersonalization, depression, diplopia, dizziness, emotional lability, hallucinations, hemiplegia, hostility aggravated, hyperkinesia, hypertonia, hypesthesia, insomnia, libido decreased/increased, nervousness, neurosis, paresthesia, sleep disorder, somnolence, thinking abnormality, tremor, vertigo
Respiratory System – asthma, bronchitis, cough increased, dyspnea, epistaxis, hemoptysis, hiccup, laryngeal neoplasia, lung fibrosis, pharyngitis, pleural disorder, pneumonia, respiratory disorder, upper respiratory inflammation/infection, rhinitis, sinusitis, stridor
Skin and Appendages – acne, alopecia, contact dermatitis, dry skin, fixed eruption, hair disorder, maculopapular rash, nail disorder, pruritus, rash, skin carcinoma, skin disorder, sweating, urticaria
Special Senses – abnormal vision, amblyopia, blepharitis, blurred vision, cataract, conjunctivitis, deafness, dry eyes, ear/eye disorder, eye pain, glaucoma, otitis media, parosmia, photophobia, retinal degeneration/disorder, taste loss, taste perversion, tinnitus, visual field defect
Urogenital System – abnormal menses, breast enlargement, breast pain, breast tenderness, dysmenorrhea, dysuria, gynecomastia, impotence, kidney calculus, kidney pain, leukorrhea, menorrhagia, menstrual disorder, penis disorder, polyuria, testis disorder, urethral pain, urinary frequency, urinary retention, urinary tract infection, urinary urgency, urination impaired, vaginitis
Postmarketing
Additional adverse experiences have been reported since PREVACID has been marketed. The majority of these cases are foreign-sourced and a relationship to PREVACID has not been established. Because these events were reported voluntarily from a population of unknown size, estimates of frequency cannot be made. These events are listed below by COSTART body system:
Body as a Whole – anaphylactic/anaphylactoid reactions, systemic lupus erythematosus
Digestive System – hepatotoxicity, pancreatitis, vomiting
Hemic and Lymphatic System – agranulocytosis, aplastic anemia, hemolytic anemia, leukopenia, neutropenia, pancytopenia, thrombocytopenia, and thrombotic thrombocytopenic purpura
Infections and Infestations – Clostridium difficile-associated diarrhea
Metabolism and Nutritional Disorders – hypomagnesemia
Musculoskeletal System – bone fracture, myositis
Skin and Appendages – severe dermatologic reactions including erythema multiforme, Stevens-Johnson Syndrome, toxic epidermal necrolysis, (some fatal), cutaneous lupus erythematosus
Special Senses – speech disorder
Urogenital System – interstitial nephritis, urinary retention
Amoxicillin
The following adverse reactions from the labeling for amoxicillin are provided for information:
The most common adverse reactions (>1%) observed in clinical trials of amoxicillin capsules were diarrhea, rash, vomiting and nausea.
The most frequently reported adverse events for patients who received triple therapy (amoxicillin/clarithromycin/lansoprazole) were diarrhea (7%), headache (6%), and taste perversion (5%).
Infections and Infestations – Mucocutaneous candidiasis
Gastrointestinal – Black hairy tongue, and hemorrhagic/pseudomembranous colitis.
Onset of pseudomembranous colitis symptoms may occur during or after antibiotic treatment (see WARNINGS).
Hypersensitivity Reactions – Anaphylaxis (see WARNINGS), serum sickness-like reactions, erythematous maculopapular rashes, erythema multiforme, Stevens-Johnson Syndrome, exfoliative dermatitis, toxic epidermal necrolysis, acute generalized exanthematous pustulosis, hypersensitivity vasculitis and urticaria have been reported.
Liver – A moderate rise in AST and/or ALT has been noted, but the significance of this finding is unknown. Hepatic dysfunction including cholestatic jaundice, hepatic cholestasis and acute cytolytic hepatitis have been reported.
Renal – Crystalluria has also been reported (see OVERDOSAGE).
Hemic and Lymphatic Systems – Anemia, including hemolytic anemia, thrombocytopenia, thrombocytopenic purpura, eosinophilia, leukopenia and agranulocytosis have been reported. These reactions are usually reversible on discontinuation of therapy and are believed to be hypersensitivity phenomena.
Central Nervous System – Reversible hyperactivity, agitation, anxiety, insomnia, confusion, behavioral changes, and/or dizziness have been reported.
Miscellaneous – Tooth discoloration (brown, yellow, or gray staining) has been reported. Most reports occurred in pediatric patients. Discoloration was reduced or eliminated with brushing or dental cleaning in most cases.
Clarithromycin
The following adverse reactions from the labeling for clarithromycin are provided for information:
The most frequent and common adverse reactions related to clarithromycin therapy for both adult and pediatric populations are abdominal pain, diarrhea, nausea, vomiting, and dysgeusia. These adverse reactions are consistent with the known safety profile of macrolide antibiotics.
There was no significant difference in the incidence of these gastrointestinal adverse reactions during clinical trials between the patient population with or without preexisting mycobacterial infections.
Adverse Reactions Observed During Clinical Trials of Clarithromycin
The following adverse reactions were observed in clinical trials with clarithromycin at a rate greater than or equal to 1%:
Gastrointestinal Disorders – Diarrhea, vomiting, dyspepsia, nausea, abdominal pain
Hepatobiliary Disorders – Liver function test abnormal
Immune System Disorders – Anaphylactoid reaction
Infections and Infestations – Candidiasis
Nervous System Disorders – Dysgeusia, headache
Psychiatric Disorders – Insomnia
Skin and Subcutaneous Tissue Disorders – Rash
Other Adverse Reactions Observed During Clinical Trials of Clarithromycin
The following adverse reactions were observed in clinical trials with clarithromycin at a rate less than 1%:
Blood and Lymphatic System Disorders – Leukopenia, neutropenia, thrombocythemia, eosinophilia
Cardiac Disorders – Electrocardiogram QT prolonged, cardiac arrest, atrial fibrillation, extrasystoles, palpitations
Ear and Labyrinth Disorders – Vertigo, tinnitus, hearing impaired
Gastrointestinal Disorders – Stomatitis, glossitis, esophagitis, gastroesophageal reflux disease, gastritis, proctalgia, abdominal distention, constipation, dry mouth, eructation, flatulence
General Disorders and Administration Site Conditions – Malaise, pyrexia, asthma, chest pain, chills, fatigue
Hepatobiliary Disorders – Cholestasis, hepatitis
Immune System Disorders – Hypersensitivity
Infections and Infestations – Cellulitis, gastroenteritis, infection, vaginal infection
Investigations – Blood bilirubin increased, blood alkaline phosphatase increased, blood lactate dehydrogenase increased, albumin globulin ratio abnormal
Metabolism and Nutrition Disorders – Anorexia, decreased appetite
Musculoskeletal and Connective Tissue Disorders – Myalgia, muscle spasms, nuchal rigidity
Nervous System Disorders – Dizziness, tremor, loss of consciousness, dyskinesia, somnolence
Psychiatric Disorders – Anxiety, nervousness
Renal and Urinary Disorders – Blood creatinine increased, blood urea increased
Respiratory, Thoracic and Mediastinal Disorders – Asthma, epistaxis, pulmonary embolism
Skin and Subcutaneous Tissue Disorders – Urticaria, dermatitis bollus, pruritus, hyperhidrosis, rash maculopapular
The following adverse reactions have been identified during post approval use of clarithromycin. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Blood and Lymphatic System Disorders – Thrombocytopenia, agranulocytosis
Cardiac Disorders – Torsades de pointes, ventricular tachycardia, ventricular arrhythmia
Ear and Labyrinth Disorders – Deafness was reported chiefly in elderly women and was usually reversible.
Gastrointestinal Disorders – Pancreatitis acute, tongue discoloration, tooth discoloration was reported and was usually reversible with professional cleaning upon discontinuation of the drug.
Hepatobiliary Disorders – Hepatic failure, jaundice hepatocellular. Adverse reactions related to hepatic dysfunction have been reported with clarithromycin (see WARNINGS, Hepatotoxicity)
Immune System Disorders – Anaphylactic reaction
Infections and Infestations – Pseudomembranous colitis
Investigations – Prothrombin time prolonged, white blood cell count decreased, international normalized ratio increased. Abnormal urine color has been reported, associated with hepatic failure.
Metabolism and Nutrition Disorders – Hypoglycemia has been reported in patients taking oral hypoglycemic agents or insulin.
Musculoskeletal and Connective Tissue Disorders – Myopathy, rhabdomyolysis was reported and in some of the reports, clarithromycin was administered concomitantly with statins, fibrates, colchicines or allopurinol (see CONTRAINDICATIONS and WARNINGS).
Nervous System Disorders – Convulsion, ageusia, parosmia, anosmia, paresthesia
Psychiatric Disorders – Psychotic disorder, confusional state, depersonalization, depression, disorientation, manic behavior, hallucination, abnormal behavior, abnormal dreams. These disorders usually resolve upon discontinuation of the drug.
There are no data on the effect of clarithromycin on the ability to drive or use machines. The potential for dizziness, vertigo, confusion and disorientation, which may occur with the medication, should be taken into account before patients drive or use machines.
Renal and Urinary Disorders – Nephritis interstitial, renal failure
Skin and Subcutaneous Tissue Disorders – Stevens-Johnson Syndrome, toxic epidermal necrolysis, drug rash with eosinophilia and systemic symptoms (DRESS), Henoch-Schonlein purpura, acne
Vascular Disorders – Hemorrhage
There have been reports of colchicine toxicity with concomitant use of clarithromycin and colchicine, especially in the elderly, some of which occurred in patients with renal insufficiency. Deaths have been reported in some such patients (see WARNINGS and PRECAUTIONS).
Laboratory Values
Prevacid
The following changes in laboratory parameters in patients who received PREVACID were reported as adverse reactions:
Abnormal liver function tests, increased SGOT (AST), increased SGPT (ALT), increased creatinine, increased alkaline phosphatase, increased globulins, increased GGTP, increased/decreased/abnormal WBC, abnormal AG ratio, abnormal RBC, bilirubinemia, blood potassium increased, blood urea increased, crystal urine present, eosinophilia, hemoglobin decreased, hyperlipemia, increased/decreased electrolytes, increased/decreased cholesterol, increased glucocorticoids, increased LDH, increased/decreased/abnormal platelets, increased gastrin levels and positive fecal occult blood. Urine abnormalities such as albuminuria, glycosuria, and hematuria were also reported.
In the placebo-controlled studies, when SGOT (AST) and SGPT (ALT) were evaluated, 0.4% (4/978) and 0.4% (11/2677) patients, who received placebo and PREVACID, respectively, had enzyme elevations greater than three times the upper limit of normal range at the final treatment visit. None of these patients who received PREVACID reported jaundice at any time during the study.
OVERDOSAGE
In case of an overdose, patients should contact a physician, poison control center, or emergency room. There is neither a pharmacologic basis nor data suggesting an increased toxicity of the combination compared to individual components.
In the event of over-exposure, treatment should be symptomatic and supportive.
If over-exposure occurs, call your poison control center at 1-800-222-1222 for current information on the management of poisoning or over-exposure.
Amoxicillin
In case of amoxicillin overdosage, discontinue medication, treat symptomatically and institute supportive measures as needed. If the overdosage is very recent and there is no contraindication, an attempt at emesis or other means of removal of drug from the stomach may be performed. A prospective study of 51 pediatric patients at a poison-control center suggested that overdosages of less than 250 mg/kg of amoxicillin are not associated with significant clinical symptoms and do not require gastric emptying.2
Interstitial nephritis resulting in oliguric renal failure has been reported in a small number of patients after overdosage with amoxicillin.
Crystalluria, in some cases leading to renal failure, has also been reported after amoxicillin overdosage in adult and pediatric patients. In case of overdosage, adequate fluid intake and diuresis should be maintained to reduce the risk of amoxicillin crystalluria. Renal impairment appears to be reversible with cessation of drug administration. High blood levels may occur more readily in patients with impaired renal function because of decreased renal clearance of amoxicillin. Amoxicillin can be removed from circulation by hemodialysis.
Clarithromycin
Overdosage of clarithromycin can cause gastrointestinal symptoms such as abdominal pain, vomiting, nausea, and diarrhea.
Adverse reactions accompanying overdosage should be treated by the prompt elimination of unabsorbed drug and supportive measures. As with other macrolides, clarithromycin serum concentrations are not expected to be appreciably affected by hemodialysis or peritoneal dialysis.
Prevacid
PREVACID is not removed from the circulation by hemodialysis. In one reported overdose, a patient consumed 600 mg of PREVACID with no adverse reaction. Oral PREVACID doses up to 5000 mg/kg in rats (approximately 650 times the recommended human dose of 60 mg/day based on BSA) and in mice (about 338 times the recommended human dose of 60 mg/day based on BSA) did not produce deaths or any clinical signs.
DOSAGE AND ADMINISTRATION
H. pylori Eradication to Reduce the Risk of Duodenal Ulcer Recurrence
The recommended adult oral dose is 30 mg PREVACID, 1 g amoxicillin, and 500 mg clarithromycin administered together twice daily (morning and evening) for 10 or 14 days (see INDICATIONS AND USAGE).
PREVPAC is not recommended in patients with creatinine clearance less than 30 mL/min.
HOW SUPPLIED
PREVPAC is supplied as an individual daily administration card, each containing:
PREVACID Capsules:
- –
- Two opaque, hard gelatin, black and pink capsules with "TAP" and "PREVACID 30" imprinted on the capsules.
Amoxicillin Capsules, USP:
- –
- Four yellow, opaque, hard gelatin 500 mg capsules imprinted with AMOX 500 on one side and GG 849 on the other side.
BIAXIN Filmtab:
- –
- Two yellow, oval film-coated 500 mg tablets debossed with the "a" logo on one side and the code "KL" on the other side of the tablets.
| NDC 64764-702-01 | Carton containing 14 daily administration cards |
| NDC 64764-702-11 | Daily administration card |
REFERENCES
- CLSI. Methods for Antimicrobial Dilution and Disk Diffusion Susceptibility Testing of Infrequently Isolated or Fastidious Bacteria – 2nd edition. CLSI document M45-A2, 2010.
- Swanson Biearman B, Dean BS, Lopez G, Krenzelok EP. The effects of penicillin and cephalosporin ingestions in children less than six years of age. Vet Hum Toxicol. 1988; 30:66-67.
PREVPAC is distributed by Takeda Pharmaceuticals America, Inc.
PREVACID (lansoprazole) Delayed-Release Capsules
Distributed by Takeda Pharmaceuticals America, Inc.
Deerfield, IL 60015, U.S.A.
Amoxicillin Capsules, USP
Product of Spain. Manufactured in Austria by Sandoz GmbH
for Sandoz Inc., Princeton, NJ 08540, U.S.A.
BIAXIN Filmtab (clarithromycin tablets, USP)
Manufactured by AbbVie Ltd.
Barceloneta, PR 00617
PREVACID and PREVPAC are trademarks registered with the U.S. Patent and Trademark Office and used under license by Takeda Pharmaceuticals America, Inc.
All other trademark names are the property of their respective owners.
© 1997-2018 Takeda Pharmaceuticals America, Inc.
PAC013 R24, June 2018
PRINCIPAL DISPLAY PANEL - Kit Box
NDC 64764-702-01
triple therapy
PREVPAC®
LANSOPRAZOLE
AMOXICILLIN
CLARITHROMYCIN
14 DAILY PATIENT CARDS
EACH DAILY PATIENT CARD CONTAINS:
2 PREVACID® (lansoprazole) Delayed-Release Capsules, 30 mg;
4 AMOXICILLIN CAPSULES, USP, 500 mg; and
2 BIAXIN® Filmtab® (clarithromycin tablets, USP) 500 mg
Takeda
Rx only
| PREVPAC
lansoprazole, amoxicillin and clarithromycin kit |
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
| Labeler - Takeda Pharmaceuticals America, Inc. (830134016) |
| Registrant - Takeda Pharmaceuticals America, Inc. (830134016) |
