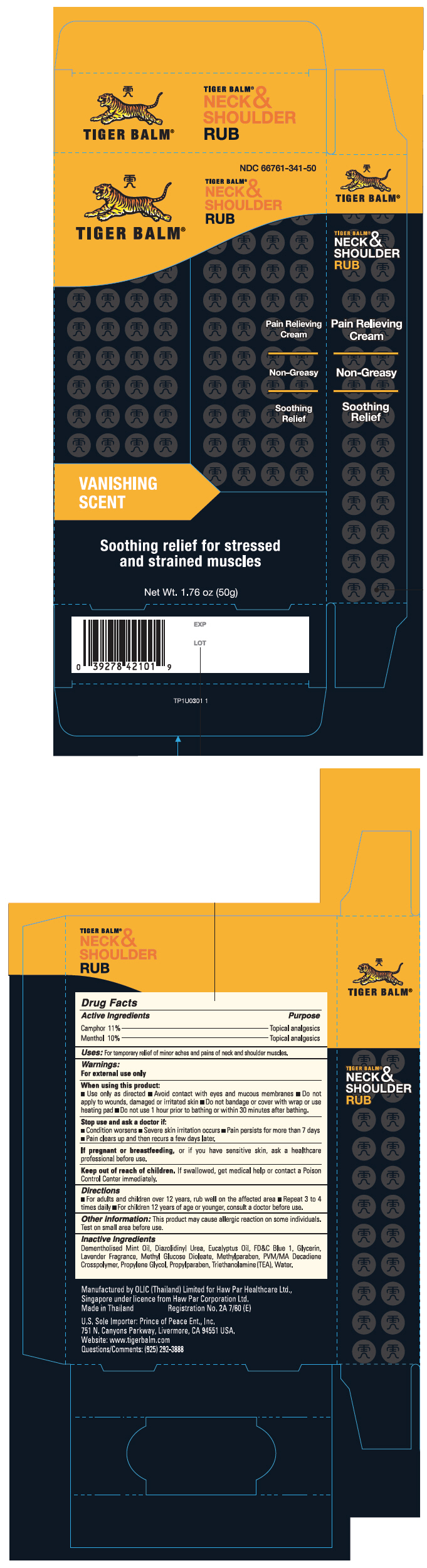
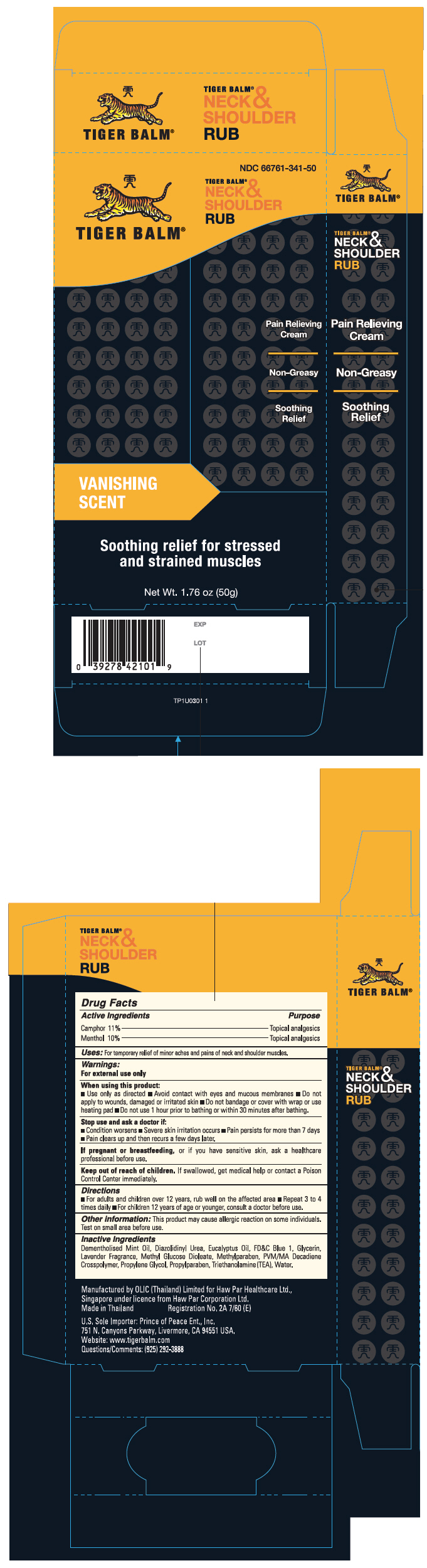
Label: TIGER BALM NECK AND SHOULDER RUB (camphor- synthetic and menthol cream
- NDC Code(s): 66761-341-01, 66761-341-03, 66761-341-50
- Packager: Haw Par Healthcare Ltd.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated January 8, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active Ingredients
- Purpose
- Uses
-
Warnings
For external use only
When using this product:
- Use only as directed
- Avoid contact with eyes and mucous membranes
- Do not apply to wounds, damaged or irritated skin
- Do not bandage or cover with wrap or use heating pad
- Do not use 1 hour prior to bathing or 30 minutes after bathing.
Stop use and ask a doctor if:
- Condition worsens
- Severe skin irritation occurs
- Pain persists for more than 7 days
- Pain clears up and then recurs a few days later.
- Directions
- Other information
- Inactive Ingredients
- SPL UNCLASSIFIED SECTION
- Questions/Comments
- PRINCIPAL DISPLAY PANEL - 50g Tube Carton
-
INGREDIENTS AND APPEARANCE
TIGER BALM NECK AND SHOULDER RUB
camphor (synthetic) and menthol creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:66761-341 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Camphor (Synthetic) (UNII: 5TJD82A1ET) (Camphor (Synthetic) - UNII:5TJD82A1ET) Camphor (Synthetic) 11 mg in 1 g Menthol, Unspecified Form (UNII: L7T10EIP3A) (Menthol, Unspecified Form - UNII:L7T10EIP3A) Menthol, Unspecified Form 10 mg in 1 g Inactive Ingredients Ingredient Name Strength Diazolidinyl Urea (UNII: H5RIZ3MPW4) Eucalyptus oil (UNII: 2R04ONI662) FD&C Blue No. 1 (UNII: H3R47K3TBD) Glycerin (UNII: PDC6A3C0OX) Peg-120 Methyl Glucose Dioleate (UNII: YM0K64F20V) Methylparaben (UNII: A2I8C7HI9T) Propylene Glycol (UNII: 6DC9Q167V3) Propylparaben (UNII: Z8IX2SC1OH) Trolamine (UNII: 9O3K93S3TK) Water (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:66761-341-50 1 in 1 CARTON 10/24/2008 1 50 g in 1 TUBE; Type 0: Not a Combination Product 2 NDC:66761-341-01 1 in 1 CARTON 10/24/2008 2 5 g in 1 TUBE; Type 0: Not a Combination Product 3 NDC:66761-341-03 3 g in 1 PACKET; Type 0: Not a Combination Product 10/24/2008 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M017 10/24/2008 Labeler - Haw Par Healthcare Ltd. (659207039) Establishment Name Address ID/FEI Business Operations OLIC (Thailand) Limited 659673016 MANUFACTURE(66761-341) Establishment Name Address ID/FEI Business Operations Haw Par Healthcare Ltd. 659207039 MANUFACTURE(66761-341)