MOTION SICKNESS ORIGINAL FORMULA- dimenhydrinate tablet
Strategic Sourcing Services LLC
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
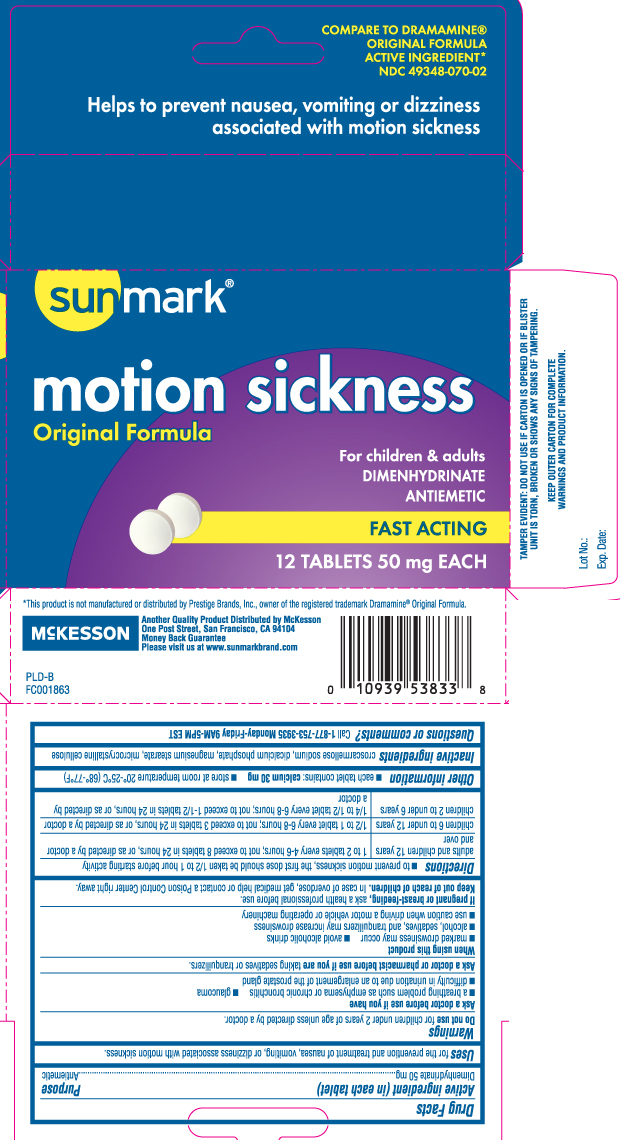
Drug Facts
Uses
for the prevention and treatment of nausea, vomiting, or dizziness associated with motion sickness.
Warnings
Ask a doctor before use if you have
- a breathing problem such as emphysema or chronic bronchitis
- glaucoma
- difficulty in urination due to an enlargement of the prostate gland
Directions
- to prevent motion sickness, the first dose should be taken 1/2 to 1 hour before starting activity
| adults and children 12 years and over | 1 to 2 tablets every 4-6 hours; not to exceed 8 tablets in 24 hours, or as directed by a doctor |
| children 6 to under 12 years | 1/2 to 1 tablet every 6-8 hours; not to exceed 3 tablets in 24 hours, or as directed by a doctor |
| children 2 to under 6 years | 1/4 to 1/2 tablet every 6-8 hours; not to exceed 1-1/2 tablets in 24 hours, or as directed by a doctor |
Inactive ingredients
croscarmellose sodium, dicalcium phosphate, magnesium stearate, microcrystalline cellulose
Principal Display Panel
COMPARE TO DRAMAMINE® ORIGINAL FORMULA ACTIVE INGREDIENT*
Helps to prevent nausea, vomiting or dizziness associated with motion sickness
Motion Sickness
Original Formula
For children & adults
DIMENHYDRINATE
ANTIEMETIC
FAST ACTING
TABLETS 50 mg EACH
*This product is not manufactured or distributed by Prestige Brands, Inc., owner of the registered trademark Dramamine® Original Formula.
Another Quality Product Distributed by McKesson
One Post Street, San Francisco, CA 94104
Please visit us at www.sunmarkbrand.com
TAMPER EVIDENT: DO NOT USE IF CARTON IS OPENED OR IF BLISTER UNIT IS TORN, BROKEN OR SHOWS ANY SIGNS OF TAMPERING.
KEEP OUTER CARTON FOR COMPLETE WARNINGS AND PRODUCT INFORMATION.
| MOTION SICKNESS
ORIGINAL FORMULA
dimenhydrinate tablet |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Strategic Sourcing Services LLC (116956644) |