Label: ACETAMINOPHEN tablet, film coated
-
NDC Code(s):
55301-175-03,
55301-175-06,
55301-175-12,
55301-175-15, view more55301-175-60, 55301-175-95
- Packager: ARMY AND AIR FORCE EXCHANGE SERVICE
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated January 17, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
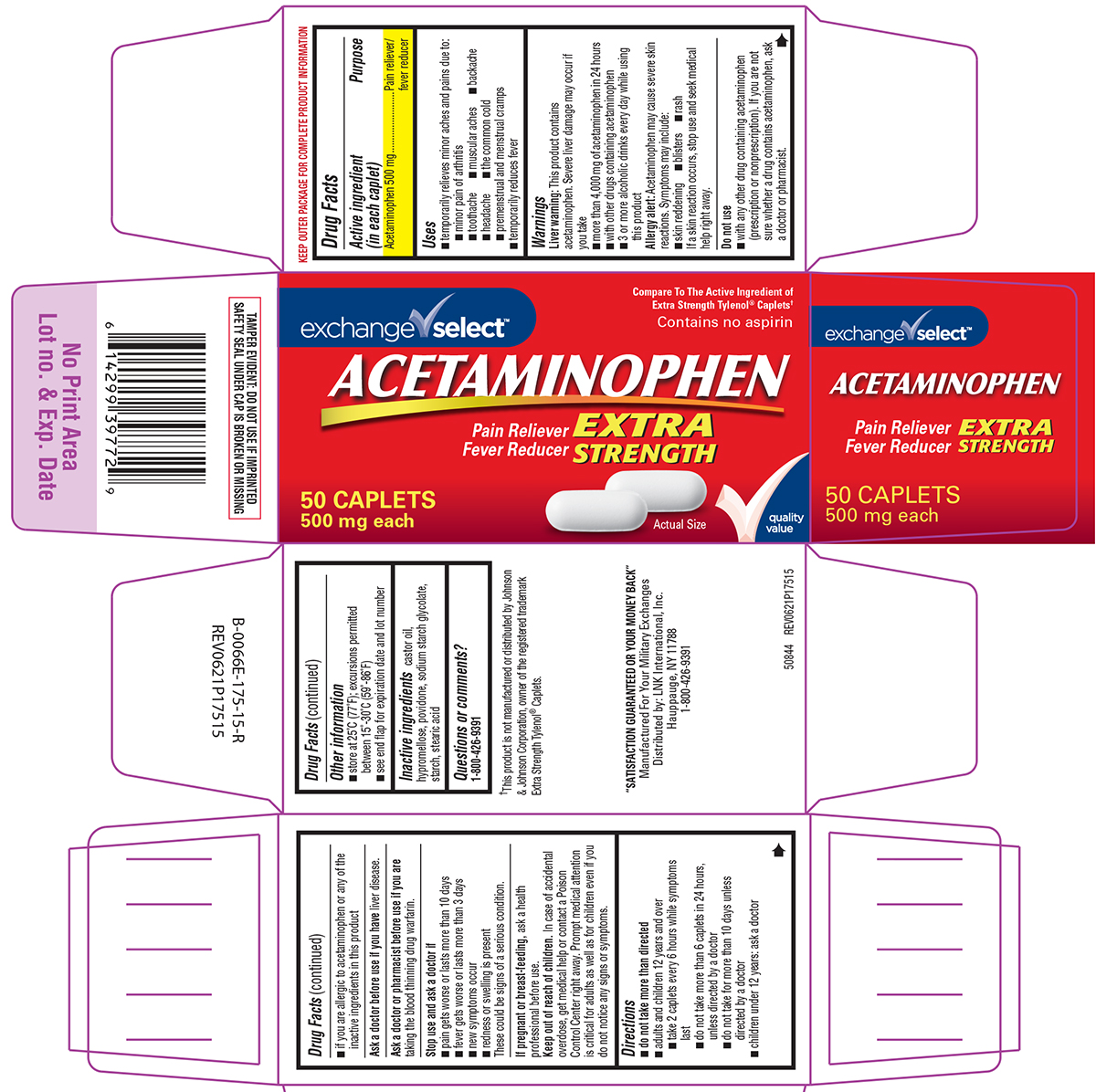
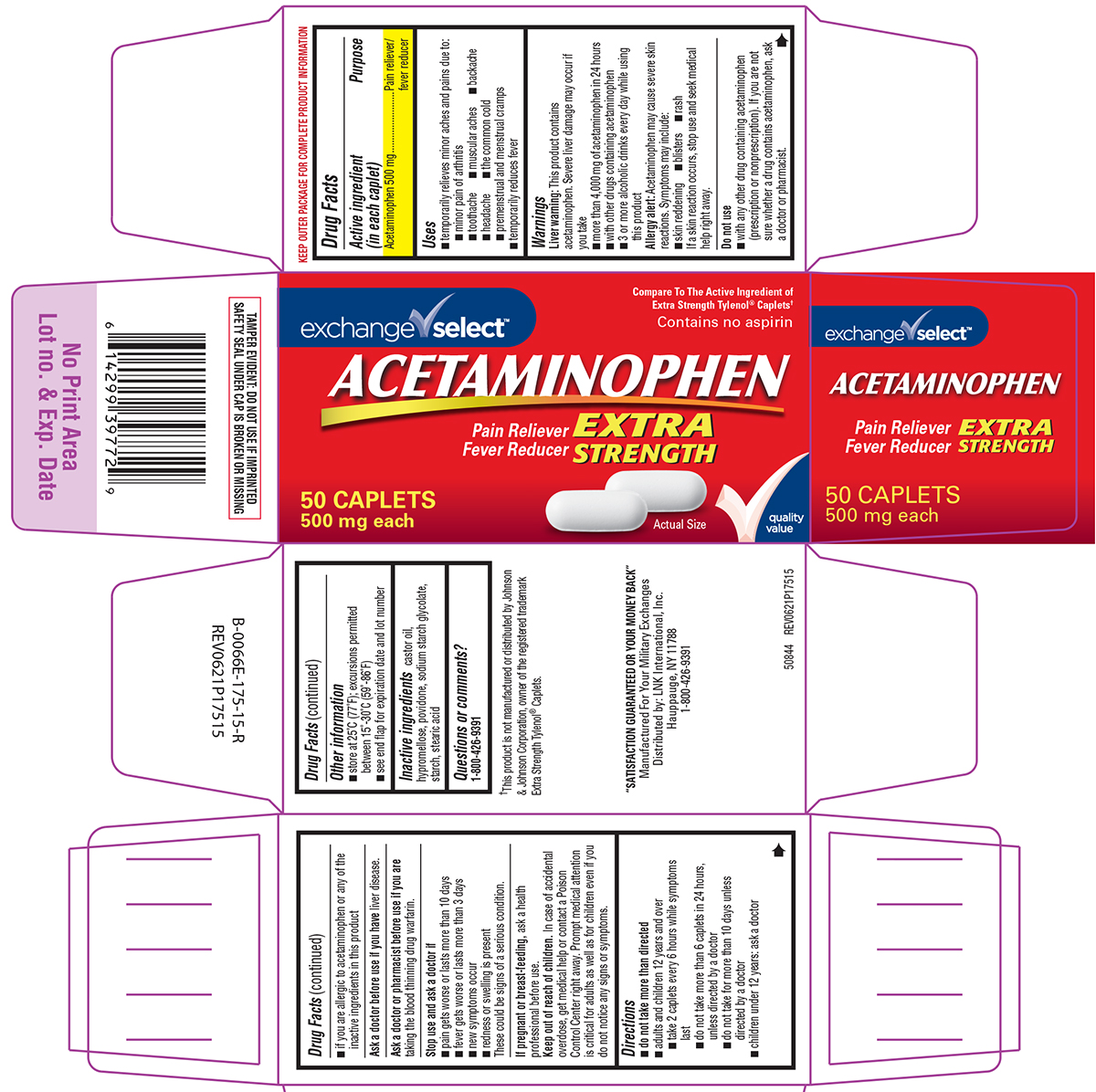
- Active ingredient (in each caplet)
- Purpose
- Uses
-
Warnings
Liver warning: This product contains acetaminophen. Severe liver damage may occur if you take
- more than 4,000 mg of acetaminophen in 24 hours
- with other drugs containing acetaminophen
- 3 or more alcoholic drinks every day while using this product
Allergy alert: Acetaminophen may cause severe skin reactions. Symptoms may include:
- skin reddening
- blisters
- rash
If a skin reaction occurs, stop use and seek medical help right away.
Do not use
- if you are allergic to acetaminophen or any of the inactive ingredients in this product
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
-
Principal Display Panel
exchange√ select™
Compare To The Active Ingredient of
Extra Strength Tylenol® Caplets†Contains no aspirin
ACETAMINOPHEN
EXTRA
STRENGTHPain Reliever
Fever Reducer50 CAPLETS
500 mg eachActual Size
✓quality
valueTAMPER EVIDENT: DO NOT USE IF IMPRINTED
SAFETY SEAL UNDER CAP IS BROKEN OR MISSING†This product is not manufactured or distributed by Johnson
& Johnson Corporation, owner of the registered trademark
Extra Strength Tylenol® Caplets."SATISFACTION GUARANTEED OR YOUR MONEY BACK"
Manufactured For Your Military Exchanges
Distributed by: LNK International, Inc.,
Hauppauge, NY 11788
1-800-426-939150844 REV0621P17515
Exchange Select 44-175
-
INGREDIENTS AND APPEARANCE
ACETAMINOPHEN
acetaminophen tablet, film coatedProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:55301-175 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 500 mg Inactive Ingredients Ingredient Name Strength CASTOR OIL (UNII: D5340Y2I9G) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) STARCH, CORN (UNII: O8232NY3SJ) STEARIC ACID (UNII: 4ELV7Z65AP) Product Characteristics Color white Score no score Shape OVAL Size 17mm Flavor Imprint Code 44;175 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:55301-175-95 10 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 04/02/1993 2 NDC:55301-175-15 1 in 1 CARTON 04/02/1993 2 50 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 3 NDC:55301-175-60 200 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 04/02/1993 4 NDC:55301-175-03 1 in 1 CARTON 04/02/1993 01/17/2024 4 10 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 5 NDC:55301-175-12 1 in 1 CARTON 04/02/1993 01/26/2017 5 100 in 1 BOTTLE; Type 0: Not a Combination Product 6 NDC:55301-175-06 2 in 1 CARTON 04/02/1993 01/26/2017 6 100 in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M013 04/02/1993 Labeler - ARMY AND AIR FORCE EXCHANGE SERVICE (001695568) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 038154464 pack(55301-175) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 832867837 manufacture(55301-175) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 832867894 manufacture(55301-175) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 868734088 manufacture(55301-175) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 967626305 pack(55301-175) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 117597853 pack(55301-175)