MORPHINE SULFATE- morphine sulfate injection
Baxter Healthcare Corporation
Disclaimer: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
----------
CII
Rx only
029600
462-296-00
Morphine
Sulfate Injection, USP
FOR SUBCUTANEOUS, INTRAMUSCULAR OR
SLOW INTRAVENOUS ADMINISTRATION
NOT FOR EPIDURAL OR INTRATHECAL ADMINISTRATION
DESCRIPTION
Chemistry
Morphine Sulfate Injection, USP is a sterile solution for subcutaneous, intramuscular or intravenous injection. Each mL contains morphine sulfate, either 5 mg, 8 mg, 10 mg or 15 mg, monobasic sodium phosphate, monohydrate 10 mg, dibasic sodium phosphate, anhydrous 2.8 mg, sodium formaldehyde sulfoxylate 3 mg and phenol 2.5 mg in Water for Injection; sulfuric acid added, if needed, for pH adjustment. The pH range is 2.5-6.5. Sealed under nitrogen.
Because of the presence and nature of the preservative and antioxidant, the product may have a characteristic odor.
Morphine is a phenanthrene-derivative opiate agonist. It is the principal alkaloid of opium and is considered to be the prototype of the opiate agonists.
Morphine sulfate occurs as white, feathery, silky crystals; cubical masses of crystals; or a white, crystalline powder. The drug contains five molecules of water of hydration and is soluble in water, having an aqueous solubility of approximately 62.5 mg/mL at 25°C, and slightly soluble in alcohol.
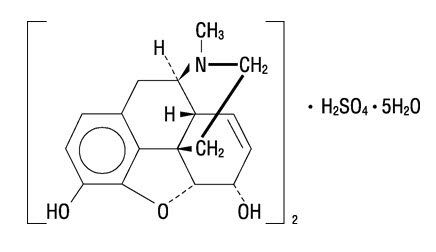
The chemical name of morphine sulfate is 7,8-Didehydro-4,5α-epoxy-17-methylmorphinan-3,6α-diol sulfate (2:1) (salt) pentahydrate, with the following structural formula:
(C17H19NO3)2 • H2SO4 • 5H2O
MW 758.83
CLINICAL PHARMACOLOGY
Like other narcotic analgesics, morphine exerts its principal pharmacological effects on the central nervous system and gastrointestinal tract. Its primary actions of therapeutic value are analgesia and sedation. Opiate analgesia involves at least three anatomical areas of the central nervous system: the periaqueductal-periventricular gray matter, the ventromedial medulla and the spinal cord. A systemically administered opiate may produce analgesia by acting at any, all or some combination of these distinct regions. Morphine interacts predominantly with the µ-receptor. The µ-binding sites of opioids are very discretely distributed in the human brain, with high densities of sites found in the posterior amygdala, hypothalamus, thalamus, nucleus caudatus, putamen and certain cortical areas. They are also found on the terminal axons of primary afferents within laminae I and II (substantia gelatinosa) of the spinal cord and in the spinal nucleus of the trigeminal nerve. Morphine appears to increase the patient's tolerance for pain and to decrease the perception of suffering, although the presence of the pain itself may still be recognized.
In addition to analgesia, alterations in mood, including euphoria and dysphoria, drowsiness and mental clouding commonly occur. Morphine depresses various respiratory centers, depresses the cough reflex and constricts the pupils. Morphine may cause nausea and vomiting by stimulating the chemoreceptor trigger zone (CTZ); however, it also depresses the vomiting center, so that subsequent doses are unlikely to produce vomiting. Nausea and vomiting are significantly more common in ambulatory than in recumbent patients.
Morphine increases the tone and decreases the propulsive contractions of the smooth muscle of the gastrointestinal tract. The resultant prolongation in gastrointestinal transit time is responsible for the constipating effect of morphine. Because morphine may increase biliary tract pressure, some patients with biliary colic may experience worsening rather than relief of pain. Morphine also decreases gastric, pancreatic and biliary secretions.
While morphine generally increases the tone of urinary tract smooth muscle, the net effect tends to be variable, in some cases producing urinary urgency, in others, difficulty in urination. Morphine has been reported to cause antidiuretic hormone (ADH) to be released, thereby reducing urine output.
In therapeutic dosage, morphine does not usually exert major effects on the cardiovascular system. However, some patients exhibit a propensity to develop orthostatic hypotension and fainting. Rapid intravenous injection is more likely to precipitate a fall in blood pressure than are intramuscular or subcutaneous injections.
Narcotic analgesics cause histamine release, which appears to be responsible for wheals or urticaria sometimes seen at the site of injection. Histamine release may also produce dilation of cutaneous blood vessels, with resultant flushing of the face and neck, pruritus and sweating.
Morphine is well absorbed after intramuscular and subcutaneous injection. It is relatively ineffective orally, reportedly because of extensive "first-pass" biotransformation by the intestinal mucosa and the liver.
Morphine is metabolized principally in the liver and undergoes conjugation with glucuronic acid at the 3-hydroxyl group. Secondary conjugation may also occur at the 6-hydroxyl group to form the 3,6-diglucuronide. Morphine is excreted in the urine mainly as morphine-3-glucuronide and smaller amounts of morphine-3,6-diglucuronide and unchanged drug. About 90% of total urinary excretion occurs within 24 hours after the last dose is given. Approximately 7-10% of a dose of morphine is excreted in the feces with a large portion of this excreted via the bile.
The elimination half-life of morphine administered by various parenteral routes has been reported to be in the range of 2.1 to about 2.6 hours.
Onset of analgesia following subcutaneous or intramuscular administration occurs within 10 to 30 minutes.
Peak analgesia occurs within 50 to 90 minutes following subcutaneous injection, 30 to 60 minutes after intramuscular injection and 20 minutes after intravenous injection. The duration of analgesia is usually 3 to 6 hours. Following intramuscular administration of morphine sulfate, maximal respiratory depression occurs within 30 minutes. Maximal respiratory depression following intravenous and subcutaneous injections occurs within 7 minutes and 90 minutes, respectively. Sensitivity of the respiratory center returns to normal within 2 to 3 hours, but respiratory minute volume may remain below normal for 4 to 5 hours.
INDICATIONS AND USAGE
Morphine Sulfate Injection is a potent analgesic used to relieve severe, acute pain or severe, chronic pain (e.g., in terminally ill patients). The drug is also used parenterally for preoperative sedation, as a supplement to anesthesia and for analgesia during labor.
Morphine is used in patients with acute pulmonary edema for its cardiovascular effects and to allay anxiety. Morphine should not be used in the treatment of pulmonary edema resulting from a chemical respiratory irritant. Morphine is the drug of choice in relieving pain of myocardial infarction.
CONTRAINDICATIONS
Morphine is contraindicated in patients with a known hypersensitivity to the drug. Because of morphine's stimulating effect on the spinal cord, morphine should not be used in convulsive states, such as those occurring in status epilepticus, tetanus and strychnine poisoning.
Morphine is also contraindicated in the following conditions: heart failure secondary to chronic lung disease; cardiac arrhythmias; brain tumor; acute alcoholism; delirium tremens; idiosyncracy to the drug. Narcotic analgesics, including morphine, are contraindicated in premature infants or during labor when delivery of a premature infant is anticipated.
NOT FOR EPIDURAL OR INTRATHECAL USE.
WARNINGS
Morphine Sulfate Injection may be habit forming (see DRUG ABUSE AND DEPENDENCE). Overdoses may cause respiratory depression, coma and death (see OVERDOSAGE). Do not use injection if it contains a precipitate.
Head Injury and Increased Intracranial Pressure
The respiratory-depressant effects of morphine and its capacity to elevate cerebrospinal fluid pressure may be markedly exaggerated in the presence of head injury, other intracranial lesions or a preexisting increase in intracranial pressure. Furthermore, narcotics produce adverse reactions which may obscure the clinical course of patients with head injuries. In such patients, morphine must be used with extreme caution and only if its use is deemed essential.
Asthma and Other Respiratory Conditions
Morphine should be used with extreme caution in patients having an acute asthmatic attack, in patients with chronic obstructive pulmonary disease or cor pulmonale, patients having a substantially decreased respiratory reserve, and patients with preexisting respiratory depression, hypoxia or hypercapnia. In such patients, even usual therapeutic doses of narcotics may decrease respiratory drive while simultaneously increasing airway resistance to the point of apnea.
Hypotensive Effect
The administration of morphine may result in severe hypotension in an individual whose ability to maintain blood pressure has already been compromised by a depleted blood volume or concurrent administration of drugs such as the phenothiazines or certain anesthetics.
Morphine may produce orthostatic hypotension in ambulatory patients.
Intravenous Use
If necessary, morphine may be given intravenously, but the injection should be given very slowly. Rapid intravenous injection of narcotic analgesics, including morphine, increases the incidence of adverse reactions. Severe respiratory depression, apnea, hypotension, peripheral circulatory collapse, cardiac arrest, as well as anaphylactoid reactions, have occurred. Morphine should not be administered intravenously unless a narcotic antagonist and the facilities for resuscitation and assisted or controlled respiration are immediately available. When morphine is given parenterally, especially intravenously, the patient should be lying down.
PRECAUTIONS
General
Morphine shares the toxic potentials of the opiate agonists, and the usual precautions of opiate agonist therapy should be observed.
Narcotic analgesics, including morphine, should be administered with caution and the initial dose reduced in patients with acute abdominal conditions, convulsive disorders, significant hepatic or renal impairment, fever, hypothyroidism, Addison's disease, ulcerative colitis, prostatic hypertrophy, in patients with recent gastrointestinal or urinary tract surgery, and in the very young or elderly or debilitated patients. Caution must be used when injecting any opioid subcutaneously or intramuscularly into chilled areas or in patients with hypotension or shock, since impaired perfusion may prevent complete absorption; if repeated injections are administered, an excessive amount may be suddenly absorbed if normal circulation is reestablished.
Morphine sulfate should be used with extreme caution in patients with disorders characterized by hypoxia, since even usual therapeutic doses of narcotics may decrease respiratory drive to the point of apnea while simultaneously increasing airway resistance.
ACUTE ABDOMINAL CONDITIONS-The administration of morphine or other narcotics may obscure the diagnosis or clinical course in patients with acute abdominal conditions.
SUPRAVENTRICULAR TACHYCARDIAS-Because of possible vagolytic action that may produce a significant increase in the ventricular response rate, morphine sulfate should be used with caution in patients with atrial flutter and other supraventricular tachycardias.
CONVULSIONS-Morphine sulfate may aggravate preexisting convulsions in patients with convulsive disorders. If dosage is escalated substantially above recommended levels because of tolerance development, convulsions may occur in individuals without a history of convulsive disorders.
Information for Patients
Morphine may impair the mental and/or physical abilities required for the performance of potentially hazardous tasks, such as driving a car or operating machinery. The concomitant use of alcohol or other central nervous system depressants, including sedatives, hypnotics, other narcotic analgesics, tranquilizers, phenothiazines and antihistamines, may have an additive effect. Morphine, like other narcotic analgesics, may produce orthostatic hypotension in ambulatory patients. The patient should be cautioned accordingly.
Drug Interactions
Morphine should be administered cautiously and in reduced dosage to avoid additive effects when other central nervous system depressants, including other narcotic analgesics, general anesthetics, phenothlazines, tricyclic antidepressants, tranquilizers or alcohol, are given concomitantly.
Virtually all drug interactions involving MAO inhibitors and narcotic analgesics have been reported with meperidine, which is contraindicated in such patients. In patients receiving MAO inhibitors, therefore, before initiating therapy with other narcotic analgesics, including morphine, an initial small test dose is advisable to allow observation of excessive narcotic effects or MAOI interaction.
Drug/Laboratory Test Interactions
Because narcotic analgesics may increase biliary tract pressure, with resultant increases in plasma amylase or lipase levels, determination of these enzyme levels may be unreliable for 24 hours after a narcotic analgesic has been given.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term animal studies have not been performed to assess the carcinogenic potential of morphine, nor are there any other animal or human data available concerning carcinogenesis, mutagenesis or impairment of fertility with this drug.
Pregnancy
Teratogenic Effects
Pregnancy Category C
Morphine has been reported to be teratogenic in mice when administered in single subcutaneous doses over 2000 times the human therapeutic dose. The authors noted that the effects produced resembled hypoxia-induced malformations. In another study, morphine was reported to be teratogenic in golden hamsters at a minimal teratogenic dose of 35 mg/kg, over 230 times the usual therapeutic dose in humans. Morphine sulfate is not teratogenic in rats at 35 mg/kg/day (thirty·five times the usual human dose) but does result in increased pup mortality and growth retardation at doses that narcotize the animal (> 10 mg/kg/day, ten times the usual human dose). There are no adequate and well-controlled studies in pregnant women. Morphine should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Labor and Delivery
Morphine sulfate crosses the placental barrier. The closer to delivery and the larger the dose used, the greater the possibility of respiratory depression in the newborn. Narcotic analgesics should be avoided during labor if delivery of a premature infant is anticipated. If the mother has received narcotic analgesics during labor, newborn infants should be observed closely for signs of respiratory depression. Resuscitation may be required (see OVERDOSAGE).
The effect of morphine, if any, on the later growth, development and functional maturation of the child is unknown.
Clinical studies have failed to demonstrate any effect of morphine on the uterus itself or on the pattern of contractions during labor. However, therapeutic doses of morphine have been reported to somewhat increase the duration of labor. The mechanism involved and the clinical significance of this observation, if any, are unknown.
Nursing Mothers
Caution should be exercised when morphine is administered to a nursing woman. Some studies, but not others, have reported detectable amounts of morphine in breast milk. The levels are probably not clinically significant after usual therapeutic dosage. The possibility of clinically important amounts being excreted in breast milk in individuals abusing morphine should be considered.
Pediatric Use
Safety and efficacy of morphine in neonates have not been established. Opiate agonists, including morphine, should not be used in premature neonates since the drugs reportedly cross the immature blood-brain barrier more readily than they do the mature barrier and thereby produce disproportionate respiratory depression. Morphine should be administered with great caution and in carefully monitored dosages to infants and small children since they may be relatively more sensitive to opiates on a body-weight basis.
ADVERSE REACTIONS
The major hazards of morphine, as with other narcotic analgesics, are respiratory depression, and, to a lesser degree, circulatory depression; respiratory arrest, shock and cardiac arrest have occurred, particularly with overdosage or rapid intravenous administration. Anaphylactoid reactions have been reported when morphine or other phenanthrene alkaloids of opium are administered intravenously.
The most frequently observed adverse reactions include sedation, lightheadedness, dizziness, nausea, vomiting and sweating. These effects seem to be more prominent in ambulatory patients and in those who are not experiencing severe pain. In such individuals, lower doses are advisable. Some adverse reactions in ambulatory patients may be alleviated if the patient lies down.
Central Nervous System
Euphoria, dysphoria, weakness, headache, agitation, tremor, uncoordinated muscle movements, transient hallucinations and disorientation, visual disturbances.
Gastrointestinal
Constipation, biliary tract spasm. Patients with chronic ulcerative colitis may experience increased colonic motility; in patients with acute ulcerative colitis, toxic dilation has been reported.
Cardiovascular
Tachycardia, bradycardia, palpitation, faintness, syncope and orthostatic hypotension.
Allergic
Allergic reactions to opiates occur infrequently; pruritus, urticaria and other skin rashes are common. Anaphylactoid reactions have been reported following intravenous administration.
Other
Opiate-induced histamine release may be responsible for the flushing of the face, sweating and pruritus often seen with these drugs. Wheals and urticaria at the site of injection are probably related to histamine release. Local tissue irritation, pain and induration have been reported following repeated subcutaneous injection.
DRUG ABUSE AND DEPENDENCE
Abuse
Morphine is known to be subject to abuse. Opiates produce relaxation, indifference to pain and stress, lethargy and euphoria. Patients who receive narcotics regularly for more than a few days may exhibit mild symptoms, which may not be recognized as withdrawal, upon discontinuation of therapy. However, the overwhelming majority of patients who receive opiates for medical reasons do not develop drug-seeking behavior or compulsive drug use. Personality characteristics play a major role in determining which patients are likely to abuse drugs. Morphine must be administered only under close supervision to patients with a history of drug abuse or dependence.
Dependence
Psychological dependence, physical dependence and tolerance are known to occur with morphine.
The severity of the abstinence syndrome is related to the degree of dependence, the abruptness of withdrawal and the drug used. If the abstinence syndrome is precipitated by administration of a narcotic antagonist, symptoms appear within a few minutes and are maximal within thirty minutes. Administration of a narcotic antagonist as a means of detecting dependence is not usually recommended. Withdrawal symptoms in patients dependent on morphine begin shortly before the time of the next scheduled dose, reach a peak at 36 to 72 hours after the last dose and then slowly subside over a period of 7 to 10 days. Symptoms include yawning; sweating; lacrimation; rhinorrhea; a restless, tossing sleep; dilated pupils; gooseflesh; irritability; tremor; nausea; vomiting and diarrhea. Treatment of the abstinence syndrome is primarily symptomatic and supportive, including maintenance of proper fluid and electrolyte balance.
OVERDOSAGE
SIGNS AND SYMPTOMS-Serious overdosage of morphine is characterized by respiratory depression (a decrease in respiratory rate and/or tidal volume, Cheyne-Stokes respiration, cyanosis), extreme somnolence progressing to stupor or coma, skeletal-muscle flaccidity, cold and clammy skin, and, sometimes, bradycardia and hypotension. In severe overdosage, particularly by intravenous route, apnea, circulatory collapse, cardiac arrest and death may occur.
It is difficult to determine with opiates what constitutes a standard toxic or lethal dose. Parenteral doses of morphine in excess of 30 mg are likely to produce serious toxic effects in the normal adult. Infants and children are believed to be relatively more sensitive to opiates on a body-weight basis. Elderly patients are also comparatively intolerant to opiates.
TREATMENT-Primary attention should be given to the reestablishment of adequate respiratory exchange through provision of a patent airway and institution of assisted or controlled ventilation. The narcotic antagonist naloxone is a specific antidote against the respiratory depression that may result from overdosage or unusual sensitivity to narcotics. Therefore, an appropriate dose of naloxone should be administered, preferably by the intravenous route, simultaneously with efforts at respiratory resuscitation. Since the duration of action of morphine may exceed that of the antagonist, the patient should be kept under continued surveillance and repeated doses of the antagonist should be administered as needed to maintain adequate respiration.
An antagonist should only be administered in the presence of clinically significant respiratory or cardiovascular depression induced by a narcotic.
Oxygen, intravenous fluids, vasopressors and other supportive measures should be employed as indicated.
NOTE-In an individual physically dependent on narcotics, the administration of the usual dose of a narcotic antagonist will precipitate an acute withdrawal syndrome. The severity of the syndrome will depend on the degree of physical dependence and the dose of antagonist administered. The use of narcotic antagonists in such individuals should be avoided if possible. If a narcotic antagonist must be used to treat serious respiratory depression in the physically dependent patient, the antagonist should be administered with extreme care and only one-tenth to one-fifth the usual initial dose administered.
DOSAGE AND ADMINISTRATION
Morphine Sulfate Injection should be given via the subcutaneous, intramuscular or slow intravenous routes. NOT FOR EPIDURAL OR INTRATHECAL USE.
In patients with severe, chronic pain, dosage should be adjusted according to the severity of the pain and the response and tolerance of the patient. In patients with exceptionally severe, chronic pain or in those who have become tolerant to the analgesic effect of opiate agonists, it may be necessary to exceed the usual dosage.
Reduced dosage is indicated in poor-risk patients, in very young or very old patients and in patients receiving other CNS depressants.
Morphine Sulfate Injection is usually administered subcutaneously; the drug may also be administered by intramuscular or slow intravenous injection. Intramuscular administration is preferred to subcutaneous injection when repeated parenteral doses are necessary, since repeated subcutaneous administration causes local tissue irritation, pain and induration.
Adults
The usual adult subcutaneous or intramuscular dosage of Morphine Sulfate Injection is 10 mg every 4 hours as necessary; dosage may range from 5-20 mg every 4 hours as necessary depending on patient requirements and response.
For intravenous administration, the usual dosage is 4-10 mg administered very slowly over 4-5 minutes. A strength of 2.5-15 mg of morphine sulfate may be diluted in 4-5 mL of sterile water for injection.
Infants and Children
Do not use in premature infants (see CONTRAINDICATIONS). Safety and effectiveness in newborn infants have not been established. Infants and children may receive a subcutaneous dose of 0.1-0.2 mg/kg as necessary; a single pediatric dose should not exceed 15 mg.
PAIN OF MYOCARDIAL INFARCTION-8-15 mg of Morphine Sulfate Injection may be administered parenterally. For very severe pain, additional smaller doses may be given every 3-4 hours.
ANALGESIA DURING LABOR-10 mg of morphine sulfate is usually administered subcutaneously or intramuscularly.
When morphine sulfate is administered intravenously, an opiate antagonist and facilities for administration of oxygen and control of respiration should be available.
Morphine sulfate has been reported to be physically or chemically incompatible with various drug products. Specialized references should be consulted for specific compatibility information.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
HOW SUPPLIED
Morphine Sulfate Injection, USP is available in the following:
5 mg/mL
1 mL DOSETTE vials packaged in 25s (NDC 10019-176-44)
8 mg/mL
1 mL DOSETTE vials packaged in 25s (NDC 10019-177-44)
1 mL DOSETTE ampuls packaged in 25s (NDC 10019-177-68)
10 mg/mL
1 mL DOSETTE vials packaged in 25s (NDC 10019-178-44)
1 mL DOSETTE ampuls packaged in 25s (NDC 10019-178-68)
10 mL Multiple Dose amber vials packaged individually (NDC 10019-178-62)
15 mg/mL
1 mL DOSETTE vials packaged in 25s (NDC 10019-179-44)
1 mL DOSETTE ampuls packaged in 25s (NDC 10019-179-68)
20 mL Multiple Dose amber vials packaged individually (NDC 10019-179-63)
STORAGE
PROTECT FROM LIGHT: Keep covered in carton. Store at 20°-25°C (68°-77°F), excursions permitted to 15°-30°C (59°-86°F)
[see USP Controlled Room Temperature].
Avoid freezing. Do not use if the color is darker than pale yellow, if it is discolored in any other way or if it contains a precipitate.
Baxter and Dosette are trademarks of Baxter International lnc. or its subsidiaries.
Baxter
Manufactured by
Baxter Healthcare Corporation
Deerfield. IL 60015 USA
For Product Inquiry 1 800 ANA DRUG (1-800-262-3784)
Revised December 2003
462-296-00
PACKAGE LABEL - PRINCIPAL DISPLAY PANEL
NDC 10019-176-39
Morphine
Sulfate Inj., USP
5 mg/mL
FOR SC, IM OR
SLOW IV USE
CII
Rx only
1 mL DOSETTE Vial
PROTECT FROM LIGHT
DO NOT USE IF PRECIPITATED
Manufactured by
Baxter Healthcare Corp.
Deerfield, IL 60015 USA
462-278-00
(01)00310019176390
Lot:
Exp.:
Lot:
Exp.:
LIGHT SENSITIVE: Keep covered in carton until time of use.
To open—Cut seal along dotted line.
NDC 10019-176-44
Morphine
Sulfate Injection, USP
CII
5 mg/mL
Rx only
FOR SC, IM OR SLOW IV USE
NOT FOR EPIDURAL OR
INTRATHECAL USE
25 x 1 mL DOSETTE Vials
Baxter
Manufactured by
Baxter Healthcare Corporation
Deerfield, IL 60015 USA
462-279-00
Each mL contains morphine
sulfate 5 mg, monobasic sodium
phosphate, monohydrate 10 mg,
dibasic sodium phosphate,
anhydrous 2.8 mg, sodium
formaldehyde sulfoxylate 3 mg
and phenol 2.5 mg in Water for
Injection. pH 2.5-6.5; sulfuric
acid added, if needed, for pH
adjustment. Sealed under nitrogen.
Usual Dosage: See package insert.
PROTECT FROM LIGHT.
Store at 20˚-25˚C (68˚-77˚F)
[see USP Controlled Room
Temperature]. Avoid freezing.
NOTE: Do not use if color is
darker than pale yellow, if it is
discolored in any other way or if
it contains a precipitate.
N 3 10019 17644 4
NDC: 10019-177-39
NO IMAGE AVAILABLE
NDC: 10019-177-44
NO IMAGE AVAILABLE
NDC: 10019-178-39
NO IMAGE AVAILABLE
NDC: 10019-178-44
NO IMAGE AVAILABLE
NDC: 10019-179-39
NO IMAGE AVAILABLE
Carton Label
Carton Label
NDC: 10019-179-44
NO IMAGE AVAILABLE
| MORPHINE SULFATE
morphine sulfate injection |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| MORPHINE SULFATE
morphine sulfate injection |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| MORPHINE SULFATE
morphine sulfate injection |
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
| MORPHINE SULFATE
morphine sulfate injection |
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
| Labeler - Baxter Healthcare Corporation (005083209) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Baxter Healthcare Corporation | 946499746 | MANUFACTURE(10019-176, 10019-177, 10019-178, 10019-179) | |