CLEARASIL ULTRA RAPID ACTION TREATMENT- salicylic acid lotion
RB Health (US) LLC
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
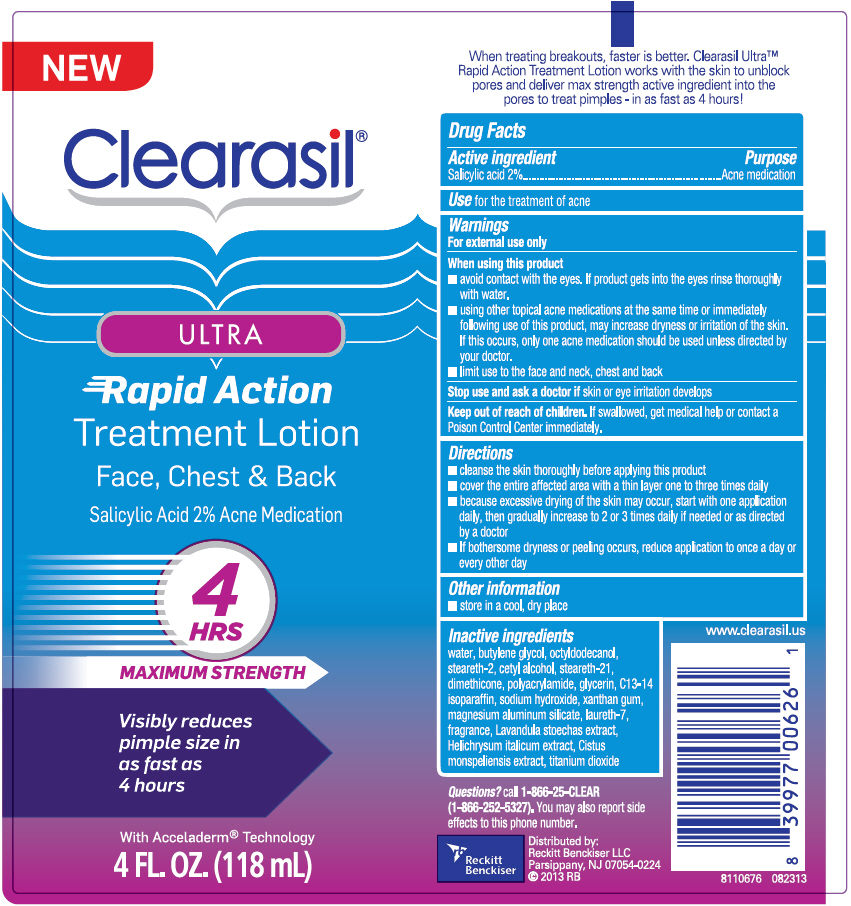
Clearasil
® Ultra
Rapid Action Treatment Lotion
Warnings
For external use only
When using this product
- avoid contact with the eyes. If product gets into the eyes rinse thoroughly with water.
- using other topical acne medications at the same time or immediately following the use of this product, may increase dryness or irritation of the skin. If this occurs, only one acne medication should be used unless directed by your doctor.
- limit use to the face and neck, chest and back
Directions
- cleanse the skin thoroughly before applying this product
- cover the entire affected area with a thin layer one to three times daily
- because excessive drying of the skin may occur, start with one application daily, then gradually increase to 2 or 3 times daily if needed or as directed by a doctor
- if bothersome dryness or peeling occurs, reduce application to once a day or every other day
Inactive ingredients
water, butylene glycol, octyldodecanol, steareth-2, cetyl alcohol, steareth-21, dimethicone, polyacrylamide, glycerin, C13-14 isoparaffin, sodium hydroxide, xanthan gum, magnesium aluminum silicate, laureth-7, fragrance, Lavandula stoechas extract, Helichrysum italicum extract, Cistus monspeliensis extract, titanium dioxide
| CLEARASIL ULTRA
RAPID ACTION TREATMENT
salicylic acid lotion |
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
| Labeler - RB Health (US) LLC (081049410) |
Revised: 5/2023
Document Id: fc899d15-b688-dc78-e053-6394a90aa16b
Set id: 21c9cfb6-b94e-48d9-8d9b-b65f1e847e49
Version: 2
Effective Time: 20230525
RB Health (US) LLC