EXTRA STRENGTH PAIN RELIEVER PM- acetaminophen and diphenhydramine hcl tablet, film coated
SUPERVALU INC.
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Equaline 44-235
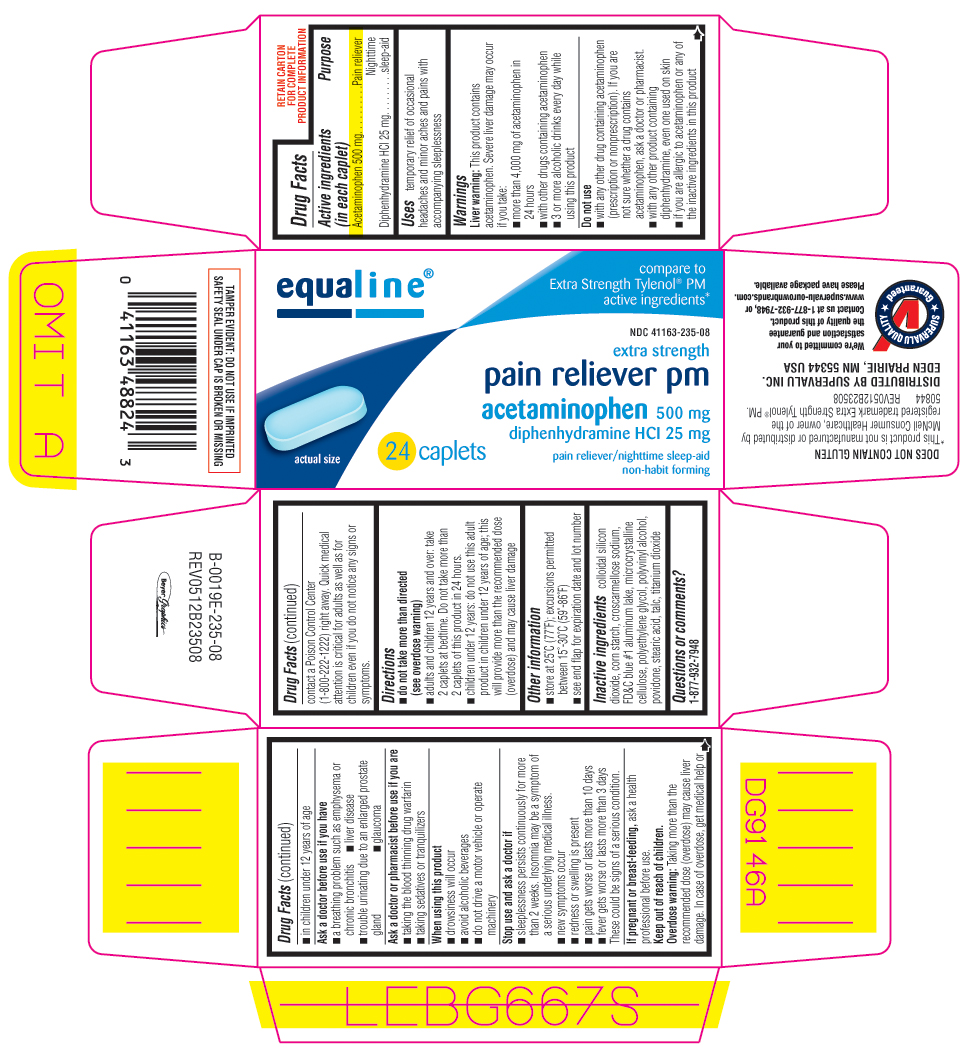
Uses
temporary relief of occasional headaches and minor aches and pains with accompanying sleeplessness
Warnings
Liver warning: This product contains acetaminophen. Severe liver damage may occur if you take:
-
more than 4,000 mg of acetaminophen in 24 hours
-
with other drugs containing acetaminophen
-
3 or more alcoholic drinks every day while using this product
Do not use
-
with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
-
with any other product containing diphenhydramine, even one used on skin
-
if you are allergic to acetaminophen or any of the inactive ingredients in this product
-
in children under 12 years of age
Ask a doctor before use if you have
-
a breathing problem such as emphysema or chronic bronchitis
-
liver disease
-
trouble urinating due to an enlarged prostate gland
-
glaucoma
Ask a doctor or pharmacist before use if you are
-
taking the blood thinning drug warfarin
-
taking sedatives or tranquilizers
When using this product
-
drowsiness will occur
-
avoid alcoholic beverages
-
do not drive a motor vehicle or operate machinery
Stop use and ask a doctor if
-
sleeplessness persists continuously for more than 2 weeks. Insomnia may be a symptom of a serious underlying medical illness.
-
new symptoms occur
-
redness or swelling is present
-
pain gets worse or lasts more than 10 days
-
fever gets worse or lasts more than 3 days
These could be signs of a serious condition.
Keep out of reach of children.
Overdose warning: Taking more than the recommended dose (overdose) may cause liver damage. In case of overdose, get medical help or contact a Poison Control Center (1-800-222-1222) right away. Quick medical attention is critical for adults as well as for children even if you do not notice any signs or symptoms.
Directions
-
do not take more than directed
(see overdose warning) - adults and children 12 years and over: take 2 caplets at bedtime. Do not take more than 2 caplets of this product in 24 hours.
- children under 12 years: Do not use this adult product in children under 12 years of age; this will provide more than the recommended dose (overdose) and may cause liver damage.
Other information
-
store at 25ºC (77ºF); excursions permitted between 15°-30°C (59°-86°F)
-
see end flap for expiration date and lot number
Inactive ingredients
colloidal silicon dioxide, corn starch, croscarmellose sodium, FD&C blue #1 aluminum lake, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol, povidone, stearic acid, talc, titanium dioxide
Principal Display Panel
equaline®
compare to Extra Strength Tylenol® PM active ingredients*
NDC 41163-235-08
extra strength
pain reliever pm
acetaminophen 500 mg
diphenhydramine HCl 25 mg
pain reliever/nighttime sleep-aid
non-habit forming
24 caplets
TAMPER EVIDENT: DO NOT USE IF IMPRINTED SAFETY SEAL UNDER CAP IS BROKEN OR MISSING
DOES NOT CONTAIN GLUTEN
*This product is not manufactured or distributed by McNeil Consumer Healthcare, owner of the registered trademark Extra Strength Tylenol® PM.
50844 REV0512B23508
DISTRIBUTED BY SUPERVALU INC.
EDEN PRARIE, MN 55344 USA
Equaline 44-235
| EXTRA STRENGTH PAIN RELIEVER PM
acetaminophen and diphenhydramine hcl tablet, film coated |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| Labeler - SUPERVALU INC. (006961411) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 038154464 | PACK(41163-235) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 832867894 | MANUFACTURE(41163-235) | |