TRETINOIN- tretinoin cream
Spear Dermatology Products
----------
DESCRIPTION:
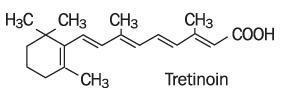
Tretinoin Cream, USP (Emollient) 0.05% contains the active ingredient tretinoin (a retinoid) in an emollient cream base. Tretinoin is a yellow-to-light-orange crystalline powder having a characteristic floral odor. Tretinoin is soluble in dimethylsulfoxide, slightly soluble in polyethylene glycol 400, octanol, and 100% ethanol. It is practically insoluble in water and mineral oil, and it is insoluble in glycerin. The chemical name for tretinoin is (all-E)-3,7-dimethyl-9-(2,6,6-trimethyl-1-cyclonexen-1-yl)-2,4,6,8-nonatetraenoic acid. Tretinoin is also referred to as all-trans-retinoic acid and has a molecular weight of 300.44. The structural formula is represented below.
Tretinoin is available as Tretinoin Cream, USP (Emollient) at a concentration of 0.05% w/w in a water-in-oil emulsion formulation consisting of butylated hydroxytoluene, citric acid monohydrate, dimethicone 50 cs, edetate disodium, fragrance, hydroxyoctacosanyl hydroxystearate, light mineral oil, methoxy PEG-22/dodecyl glycol copolymer, methylparaben, PEG-45/dodecyl glycol copolymer, purified water, quaternium-15, stearoxytrimethylsilane and stearyl alcohol, and sorbitol solution.
CLINICAL PHARMACOLOGY:
Tretinoin is an endogenous retinoid metabolite of Vitamin A that binds to intracellular receptors in the cytosol and nucleus, but cutaneous levels of tretinoin in excess of physiologic concentrations occur following application of a tretinoin-containing topical drug product. Although tretinoin activates three members of the retinoic acid (RAR) nuclear receptors (RARa, RARb, and RARg) which may act to modify gene expression, subsequent protein synthesis, and epithelial cell growth and differentiation, it has not been established whether the clinical effects of tretinoin are mediated through activation of retinoic acid receptors, other mechanisms such as irritation, or both.
The effect of tretinoin on skin with chronic photodamage has not been evaluated in animal studies. When hairless albino mice were treated topically with tretinoin shortly after a period of UVB irradiation, new collagen formation was demonstrated only in photodamaged skin. However, in human skin treated topically, adequate data have not been provided to demonstrate any increase in desmosine, hydroxyproline, or elastin mRNA. Application of 0.1% tretinoin cream to photodamaged human forearm skin was associated with an increase in antibody staining for procollagen I propeptide. No correlation was made between procollagen I propeptide staining with collagen I levels or with observed clinical effects. Thus, the relationships between the increased collagen in rodents, increased procollagen I propeptide in humans, and the clinical effects of tretinoin have not yet been clearly defined.
Tretinoin was shown to enhance UV-stimulated melanogenesis in pigmented mice. Generalized amyloid deposition in the basal layer of tretinoin-treated skin was noted in a two-year mouse study. In a different study, hyalinization at tretinoin-treated skin sites was noted at doses beginning at 0.25 mg/kg in CD-1 mice.
The transdermal absorption of tretinoin from various topical formulations ranged from 1% to 31% of applied dose, depending on whether it was applied to healthy skin or dermatitic skin. When percutaneous absorption of Tretinoin Cream, USP (Emollient) 0.05% was assessed in healthy male subjects (n=14), as well as after repeated daily applications for 28 days, the absorption of tretinoin was less than 2% and endogenous concentrations of tretinoin and its major metabolites were unaltered.
INDICATIONS AND USAGE:
(To understand fully the indication for this product, please read the entire INDICATIONS AND USAGE section of the labeling.)
Tretinoin Cream, USP (Emollient) 0.05% is indicated as an adjunctive agent (see second bullet point below) for use in the mitigation (palliation) of fine wrinkles, mottled hyperpigmentation, and tactile roughness of facial skin in patients who use comprehensive skin care and sunlight avoidance programs (see bullet 3 for populations in which effectiveness has not been established). TRETINOIN CREAM, USP (EMOLLIENT) DOES NOT ELIMINATE WRINKLES, REPAIR SUN-DAMAGED SKIN, REVERSE PHOTOAGING, or RESTORE MORE YOUTHFUL or YOUNGER SKIN. In double-blinded, vehicle-controlled clinical studies, many patients in the vehicle group achieved desired palliative effects on fine wrinkling, mottled hyperpigmentation, and tactile roughness of facial skin with the use of comprehensive skin care and sunlight avoidance programs including sunscreens, protective clothing, and non-prescription emollient creams.
- Tretinoin Cream, USP (Emollient) 0.05% has NOT DEMONSTRATED A MITIGATING EFFECT on significant signs of chronic sun exposure such as coarse or deep wrinkling, skin yellowing, lentigines, telangiectasia, skin laxity, keratinocytic atypia, melanocytic atypia, or dermal elastosis.
- Tretinoin Cream, USP (Emollient) 0.05% should be used under medical supervision as an adjunct to a comprehensive skin care and sunlight avoidance program that includes the use of effective sunscreens (minimum SPF of 15) and protective clothing when desired results on fine wrinkles, mottled hyperpigmentation, and roughness of facial skin have not been achieved with a comprehensive skin care and sunlight avoidance program alone.
- The effectiveness of Tretinoin Cream, USP (Emollient) 0.05% in the mitigation of fine wrinkles, mottled hyperpigmentation, and tactile roughness of facial skin has not been established in people greater than 50 years of age OR in people with moderately to heavily pigmented skin. In addition, patients with visible actinic keratoses and patients with a history of skin cancer were excluded from clinical trials of Tretinoin Cream, USP (Emollient) 0.05%. Thus the effectiveness and safety of Tretinoin Cream, USP (Emollient) 0.05% in these populations are not known at this time.
- Neither the safety nor the effectiveness of Tretinoin Cream, USP (Emollient) for the prevention or treatment of actinic keratoses or skin neoplasms has been established.
- Neither the safety nor the efficacy of using Tretinoin Cream, USP (Emollient) 0.05% daily for greater than 48 weeks has been established, and daily use beyond 48 weeks has not been systematically and histologically investigated in adequate and well-controlled trials. (see WARNINGS section.)
CLINICAL TRIALS DATA:
Two adequate and well-controlled trials were conducted involving a total of 161 evaluable patients (under 50 years of age) treated with Tretinoin Cream, USP (Emollient) 0.05% and 154 evaluable patients treated with the vehicle cream on the face for 24 weeks as an adjunct to a comprehensive skin care and sun-avoidance program, to assess the effects on fine wrinkling, mottled hyperpigmentation, and tactile skin roughness. Patients were evaluated at baseline on a 10-point scale, and changes from that baseline rating were categorized as follows:
No Improvement: No change or an increase of 1 unit or more
Minimal Improvement: Reduction of 1 unit
Moderate Improvement: Reduction of 2 units or more
In these trials, the fine wrinkles, mottled hyperpigmentation, and tactile roughness of the facial skin were thought to be caused by multiple factors which included intrinsic aging or environmental factors, such as chronic sunlight exposure.
The results of these assessments are as follows:
| *CSP= Comprehensive skin protection and sun-avoidance programs included use of sunscreens, protective clothing, and non-prescription emollient creams. | |||
| FINE WRINKLING | |||
| NO IMPROVEMENT | MINIMAL IMPROVEMENT | MODERATE IMPROVEMENT |
|
| Tretinoin Cream, USP (Emollient) 0.05% + CSP* | 36% | 40% | 24% |
| Vehicle + CSP | 62% | 30% | 8% |
| MOTTLED HYPERPIGMENTATION | |||
| NO IMPROVEMENT | MINIMAL IMPROVEMENT | MODERATE IMPROVEMENT |
|
| Tretinoin Cream, USP (Emollient) 0.05% + CSP* | 35% | 27% | 38% |
| Vehicle + CSP | 53% | 21% | 27% |
| TACTILE SKIN ROUGHNESS | |||
| NO IMPROVEMENT | MINIMAL IMPROVEMENT | MODERATE IMPROVEMENT |
|
| Tretinoin Cream, USP (Emollient) 0.05% + CSP* | 49% | 35% | 16% |
| Vehicle + CSP | 67% | 23% | 10% |
Most of the improvement in these signs was noted during the first 24 weeks of therapy. Thereafter, therapy primarily maintained the improvement realized during the first 24 weeks.
A majority of patients will lose most mitigating effects of Tretinoin Cream, USP (Emollient) 0.05% on fine wrinkles, mottled hyperpigmentation, and tactile roughness of facial skin with discontinuation of a comprehensive skin care and sunlight avoidance program including Tretinoin Cream, USP (Emollient) 0.05%; however, the safety and effectiveness of using Tretinoin Cream, USP (Emollient) 0.05% daily for greater than 48 weeks have not been established.
CONTRAINDICATIONS:
This drug is contraindicated in individuals with a history of sensitivity reactions to any of its components. It should be discontinued if hypersensitivity to any of its ingredients is noted.
WARNINGS:
- Tretinoin Cream, USP (Emollient) is a dermal irritant, and the results of continued irritation of the skin for greater than 48 weeks in chronic long-term use are not known. There is evidence of atypical changes in melanocytes and keratinocytes, and of increased dermal elastosis in some patients treated with Tretinoin Cream, USP (Emollient) for longer than 48 weeks. The significance of these findings is unknown.
- Safety and effectiveness of Tretinoin Cream, USP (Emollient) 0.05% in individuals with moderately or heavily pigmented skin have not been established.
- Tretinoin Cream, USP (Emollient) should not be administered if the patient is also taking drugs known to be photosensitizers (e.g., thiazides, tetracyclines, fluoroquinolones, phenothiazines, sulfonamides) because of the possibility of augmented phototoxicity.
Exposure to sunlight (including sunlamps) should be avoided or minimized during use of Tretinoin Cream, USP (Emollient), because of heightened sunburn susceptibility. Patients should be warned to use sunscreens (minimum SPF of 15) and protective clothing when using Tretinoin Cream, USP (Emollient). Patients with sunburn should be advised not to use Tretinoin Cream, USP (Emollient) until fully recovered. Patients who may have considerable sun exposure, e.g., due to their occupation and those patients with inherent sensitivity to sunlight, should exercise caution when using Tretinoin Cream, USP (Emollient) and follow the precautions outlined in the Patient Package Insert.
Tretinoin Cream, USP (Emollient) should be kept out of the eyes, mouth, angles of the nose, and mucous membranes. Topical use may cause severe local erythema, pruritus, burning, stinging, and peeling at the site of application. If the degree of local irritation warrants, patients should be directed to use less medication, decrease the frequency of application, discontinue use temporarily, or discontinue use altogether and consider additional appropriate therapy.
Tretinoin has been reported to cause severe irritation on eczematous skin and should be used only with caution in patients with this condition.
Application of larger amounts of medication than recommended has not been shown to lead to more rapid or better results, and marked redness, peeling, or discomfort may occur.
PRECAUTIONS:
General:
Tretinoin Cream, USP (Emollient) should only be used as an adjunct to a comprehensive skin care and sun avoidance program. (See INDICATIONS AND USAGE section.)
If a drug sensitivity, chemical irritation, or a systemic adverse reaction develops, use of Tretinoin Cream, USP (Emollient) should be discontinued.
Weather extremes, such as wind or cold, may be more irritating to patients using tretinoin-containing products.
Information for Patients:
Tretinoin Cream, USP (Emollient) 0.05% is to be used as described below unless otherwise directed by your physician:
- It is for use on the face only.
- Avoid contact with the eyes, ears, nostrils, angles of the nose, and mouth. Tretinoin Cream, USP (Emollient) may cause severe redness, itching, burning, stinging, and peeling if used on these areas.
- In the evening, gently wash your face with a mild soap. Pat skin dry and wait 20-30 minutes before applying Tretinoin Cream, USP (Emollient). Apply only a pea-sized amount of Tretinoin Cream, USP (Emollient) to your face at one time. This should be enough to cover the entire affected area lightly.
- Do not wash your face for at least one hour after applying Tretinoin Cream, USP (Emollient).
- For best results, you are advised not to apply another skin care product or cosmetic for at least one hour after applying Tretinoin Cream, USP (Emollient).
- In the morning, apply a moisturizing sunscreen, SPF 15 or greater.
- Tretinoin Cream, USP (Emollient) is a serious medication. Do not use Tretinoin Cream, USP (Emollient) if you are pregnant or attempting to become pregnant. If you become pregnant while using Tretinoin Cream, USP (Emollient), please contact your physician immediately.
- Avoid sunlight and other medicines that may increase your sensitivity to sunlight.
- Tretinoin Cream, USP (Emollient) does not remove wrinkles or repair sun-damaged skin.
Please refer to the Patient Package Insert for additional patient information.
Drug Interactions:
Concomitant topical medications, medicated or abrasive soaps, shampoos, cleansers, cosmetics with a strong drying effect, products with high concentrations of alcohol, astringents, spices or lime, permanent wave solutions, electrolysis, hair depilatories or waxes, and products that may irritate the skin should be used with caution in patients being treated with Tretinoin Cream, USP (Emollient) because they may increase irritation with Tretinoin Cream, USP (Emollient).
Tretinoin Cream, USP (Emollient) should not be administered if the patient is also taking drugs known to be photosensitizers (e.g., thiazides, tetracyclines, fluoroquinolones, phenothiazines, sulfonamides) because of the possibility of augmented phototoxicity.
Carcinogenesis, Mutagenesis, Impairment of Fertility:
In a 91-week dermal study in which CD-1 mice were administered 0.017% and 0.035% formulations of tretinoin, cutaneous squamous cell carcinomas and papillomas were observed in some female mice. These concentrations are lower than the concentration of tretinoin in this clinical formulation (0.05%). A dose-related incidence of liver tumors in male mice was observed at those same doses. The maximum systemic doses associated with the 0.017% and 0.035% formulations are 0.5 and 1.0 mg/kg/day. These doses are 4 and 8 times the maximum human systemic dose, when adjusted for total body surface area. The biological significance of the neoplastic findings is not clear because they occurred at doses that exceeded the maximally tolerated dermal dose of tretinoin and because they were within the background natural occurrence rate for these tumors in this strain of mice. There was no evidence of carcinogenic potential when 0.025 mg/kg/day of tretinoin was administered topically to mice (1/5th of times the maximum human systemic dose, adjusted for total body surface area). For purposes of comparisons of the systemic animal exposure to the systemic human exposure, the maximum human systemic dose is 1 gram of Tretinoin Cream, USP (Emollient) 0.05% applied daily to a 50 kg person (0.01 mg tretinoin/kg body weight, or 0.37 mg/m2 total body surface area).
Studies in hairless albino mice suggest that concurrent exposure to tretinoin may enhance the tumorigenic potential of carcinogenic doses of UVB and UVA light from a solar simulator. This effect has been confirmed in a later study in pigmented mice, and dark pigmentation did not overcome the enhancement of photocarcinogenesis by 0.05% tretinoin. Although the significance of these studies in humans is not clear, patients should minimize exposure to sunlight or artificial ultraviolet irradiation sources.
The mutagenic potential of tretinoin was evaluated in the Ames assay and in the in vivo mouse micronucleus assay, both of which were negative.
In dermal Segment I fertility studies in rats, slight (not statistically significant) decreases in sperm count and motility were seen at 0.5 mg/kg/day (8 times the maximum human systemic dose adjusted for total body surface area), and slight (not statistically significant) increases in the number and percent of nonviable embryos in females treated with 0.25 mg/kg/day (4 times the maximum human systemic dose adjusted for total body surface area) and above were observed. A dermal Segment III study with Tretinoin Cream, USP (Emollient) has not been performed in any species. In oral Segment I and Segment III studies in rats with tretinoin, decreased survival of neonates and growth retardation were observed at doses in excess of 2 mg/kg/day (>33 times the maximum human systemic dose adjusted for total body surface area).
Pregnancy:
Teratogenic effects: Pregnancy Category C.
ORAL tretinoin has been shown to be teratogenic in rats, mice, rabbits, hamsters, and subhuman primates. It was teratogenic and fetotoxic in Wistar rats when given orally or topically in doses greater than 1 mg/kg/day (17 times the maximum human systemic dose normalized for total body surface area). However, variations in teratogenic doses among various strains of rats have been reported. In the cynomolgus monkey, which, metabolically, is closer to humans for tretinoin than the other species examined, fetal malformations were reported at doses of 10 mg/kg/day or greater, but none were observed at 5 mg/kg/day (167 times the maximum human systemic dose adjusted for total body surface area), although increased skeletal variations were observed at all doses. A dose-related increase in embryolethality and abortion was reported. Similar results have also been reported in pigtail macaques.
TOPICAL tretinoin in animal teratogenicity tests has generated equivocal results. There is evidence for teratogenicity (shortened or kinked tail) of topical tretinoin in Wistar rats at doses greater than 1 mg/kg/day (17 times the maximum human systemic dose adjusted for total body surface area). Anomalies (humerus: short 13%, bent 6%, os parietal incompletely ossified 14%) have also been reported when 10 mg/kg/day was dermally applied.
There are other reports in New Zealand White rabbits administered doses of greater than 0.2 mg/kg/day (7 times the maximum human systemic dose adjusted for total body surface area) of an increased incidence of domed head and hydrocephaly, typical of retinoid-induced fetal malformations in this species.
In contrast, several well-controlled animal studies have shown that dermally applied tretinoin may be fetotoxic, but not overtly teratogenic, in rats and rabbits at doses of 1.0 and 0.5 mg/kg/day, respectively (17 times the maximum human systemic dose adjusted for total body surface area in both species).
With widespread use of any drug, a small number of birth defect reports associated temporally with the administration of the drug would be expected by chance alone. Thirty human cases of temporally-associated congenital malformations have been reported during twenty-five years of clinical use of another formulation of topical tretinoin (Retin-A). Although no definite pattern of teratogenicity and no causal association has been established from these cases, 5 of the reports describe the rare birth defect category holoprosencephaly (defects associated with incomplete midline development of the forebrain). The significance of these spontaneous reports in terms of risk to the fetus is not known.
Non-teratogenic effects:
Dermal tretinoin has been shown to be fetotoxic in rabbits when administered 0.5 mg/kg/day (17 times the human topical dose normalized for total body surface area). Oral tretinoin has been shown to be fetotoxic, resulting in skeletal variations and increased intrauterine death, in rats when administered 2.5 mg/kg/day (42 times the maximum human systemic dose adjusted for total body surface area).
There are, however, no adequate and well-controlled studies in pregnant women. Tretinoin Cream, USP (Emollient) should not be used during pregnancy.
Nursing Mothers:
It is not known whether this drug is excreted in human milk. Since many drugs are excreted in human milk, mitigation of fine wrinkles, mottled hyperpigmentation, and tactile roughness on the face with Tretinoin Cream, USP (Emollient) may be postponed in nursing mothers until after completion of the nursing period.
Pediatric Use:
Safety and effectiveness in patients less than 18 years of age have not been established.
Geriatric Use:
Clinical studies of Tretinoin Cream, USP (Emollient) 0.05% did not include a sufficient number of subjects aged 65 and over to determine whether they respond differently than younger subjects. Other reported clinical experience has not identified differences in responses between elderly and younger patients.
ADVERSE REACTIONS:
(See WARNINGS and PRECAUTIONS sections.)
In double-blind, vehicle-controlled studies involving 179 patients who applied Tretinoin Cream, USP (Emollient) 0.05% to their faces, adverse reactions associated with the use of Tretinoin Cream, USP (Emollient) 0.05% were limited primarily to the skin. During these trials, 4% of patients had to discontinue use of Tretinoin Cream, USP (Emollient) 0.05% because of adverse reactions. These discontinuations were due to skin irritation or related cutaneous adverse reactions.
Local reactions such as peeling, dry skin, burning, stinging, erythema, and pruritus were reported by almost all subjects during therapy with Tretinoin Cream, USP (Emollient) 0.05%. These signs and symptoms were usually of mild to moderate severity and generally occurred early in therapy. In most patients the dryness, peeling, and redness recurred after an initial (24 week) decline.
In spontaneous post-marketing adverse event reporting for Tretinoin Cream, USP (Emollient) 0.05%, approximately 2% of those were for skin hypo- or hyperpigmentation. Other spontaneously reported adverse events predominantly appear to be local reactions similar to those seen in clinical trials.
OVERDOSAGE:
Application of larger amounts of medication than recommended has not been shown to lead to more rapid or better results, and marked redness, peeling, or discomfort may occur. Oral ingestion of the drug may lead to the same side effects as those associated with excessive oral intake of Vitamin A.
DOSAGE AND ADMINISTRATION:
- Do NOT use Tretinoin Cream, USP (Emollient) if the patient is pregnant or is attempting to become pregnant or is at high risk of pregnancy
- Do NOT use Tretinoin Cream, USP (Emollient) if the patient is sunburned or if the patient has eczema or other chronic skin conditions of the face
- Do NOT use Tretinoin Cream, USP (Emollient) if the patient is inherently sensitive to sunlight
- Do NOT use Tretinoin Cream, USP (Emollient) if the patient is also taking drug(s) known to be photosensitizers (e.g., thiazides, tetracyclines, fluoroquinolones, phenothiazines, sulfonamides) because of the possibility of augmented phototoxicity.
Patients require detailed instruction to obtain maximal benefits and to understand all the precautions necessary to use this product with greatest safety. The physician should review the Patient Package Insert.
Tretinoin Cream, USP (Emollient) should be applied to the face once a day in the evening, using only enough to cover the entire affected area lightly. Patients should gently wash their faces with a mild soap, pat the skin dry, and wait 20 to 30 minutes before applying Tretinoin Cream, USP (Emollient). The patient should apply a pea-sized amount of cream to cover the entire affected face lightly. Caution should be taken when applying the cream to avoid the eyes, ears, nostrils, and mouth.
Application of Tretinoin Cream, USP (Emollient) may cause a transitory feeling of warmth or slight stinging.
Mitigation (palliation) of facial fine wrinkling, mottled hyperpigmentation, and tactile roughness may occur gradually over the course of therapy. Up to six months of therapy may be required before the effects are seen. Most of the improvement noted with Tretinoin Cream, USP (Emollient) 0.05% is seen during the first 24 weeks of therapy. Thereafter, therapy primarily maintains the improvement realized during the first 24 weeks.
With discontinuation of Tretinoin Cream, USP (Emollient) 0.05% therapy, a majority of patients will lose most mitigating effects of Tretinoin Cream, USP (Emollient) 0.05% on fine wrinkles, mottled hyperpigmentation, and tactile roughness of facial skin; however, the safety and effectiveness of using Tretinoin Cream, USP (Emollient) 0.05% daily for greater than 48 weeks have not been established.
Application of larger amounts of medication than recommended may not lead to more rapid results or better results, and marked redness, peeling, or discomfort may occur.
Patients treated with Tretinoin Cream, USP (Emollient) 0.05% may use cosmetics, but the areas to be treated should be cleansed thoroughly before the medication is applied. (See PRECAUTIONS section.)
HOW SUPPLIED
Tretinoin Cream, USP (Emollient) 0.05% is available in these sizes:
NDC 66530-247-40 40 gram tube
NDC 66530-247-60 60 gram tube
Storage: Store at 20-25°C (68-77°F) [see USP Controlled Room Temperature]. DO NOT FREEZE.
Rx only.
128278
Printed in USA
Manufactured by DPT Laboratories, San Antonio, TX 78215
Distributed by Spear Dermatology Products, Randolph, NJ 07869
SUPPLEMENTAL PATIENT MATERIAL
USE ONLY ON THE FACE
Read this leaflet carefully before you start to use your medicine. Read the information you get every time you get more medicine. There may be new information about the drug. This leaflet does not take the place of talks with your doctor. It is important for you to talk with your doctor about how to use Tretinoin Cream, USP (Emollient) for the best results and how to reduce side effects.
What is the Most Important Information about Tretinoin Cream, USP (Emollient)?
Tretinoin Cream, USP (Emollient) is a serious medicine. Do not use Tretinoin Cream, USP (Emollient) if you are pregnant or attempting to become pregnant. If you become pregnant while using Tretinoin Cream, USP (Emollient), please contact your doctor immediately.
Avoid sunlight and other medicines that may increase your sensitivity to sunlight (See “Who should not use Tretinoin Cream, USP (Emollient)?”).
Tretinoin Cream, USP (Emollient) does not remove wrinkles or repair sun-damaged skin. (See “What is Tretinoin Cream, USP (Emollient)?” for more details.) Tretinoin Cream, USP (Emollient) 0.05% has not been tested in people over age 50 or in people with medium or dark skin color. (See “Who should not use Tretinoin Cream, USP (Emollient)?”).
What is TRETINOIN CREAM, USP (EMOLLIENT)?
Tretinoin Cream, USP (Emollient) 0.05% is a prescription medicine that may reduce fine facial wrinkles, facial mottled hyperpigmentation, and facial skin roughness. It is for patients who are also using a skin care and sunlight avoidance program. Tretinoin Cream, USP (Emollient) does not remove wrinkles or repair sun-damaged skin. Tretinoin Cream, USP (Emollient) does not work for everyone who uses it. It may work better for some patients than for others.
Tretinoin Cream, USP (Emollient) should be used only under the guidance of your doctor as part of a skin care and sunlight avoidance program. This program should include avoiding sunlight as much as possible, using clothing to protect you from sunlight, using sunscreens with a minimum SPF of 15, and using face creams that add moisture to the skin.
When you use Tretinoin Cream, USP (Emollient), you will not see improvement right away. Generally, you may notice some effects in 3 to 4 months. If Tretinoin Cream, USP (Emollient) treatment is stopped, the improvement may gradually disappear.
The use of Tretinoin Cream, USP (Emollient) 0.05% in patients for more than 48 weeks has not been studied. Therefore, it is not known if Tretinoin Cream, USP (Emollient) 0.05% is safe or works if used longer than 48 weeks. Tretinoin Cream, USP (Emollient) 0.05% has not been shown to work or be safe in people over age 50, in people with medium to dark skin color, or in people with visible actinic keratoses or in people with a history of skin cancer.
Who Should Not Use Tretinoin Cream, USP (Emollient)?
Do not use Tretinoin Cream, USP (Emollient) if:
- you are pregnant or plan to become pregnant. If you become pregnant while using Tretinoin Cream, USP (Emollient), please contact your doctor immediately.
- you are sunburned or your skin is irritated.
- you are highly sensitive to sunlight.
- you are allergic to any of the ingredients in Tretinoin Cream, USP (Emollient). The active ingredient is tretinoin. Ask your doctor or pharmacist about the inactive ingredients.
Tretinoin Cream, USP (Emollient) can cause increased skin irritation and increased chance of sunburn.
Tell your doctor if you have any skin condition. Tretinoin Cream, USP (Emollient) may not be right for you.
Because Tretinoin Cream, USP (Emollient) may make your skin more likely to burn from sunlight, tell your doctor if you are using other medicines that increase sensitivity to sunlight. You should not use Tretinoin Cream, USP (Emollient) with such medicines. These include, but are not limited to:
- thiazides (to treat high blood pressure)
- tetracyclines, fluoroquinolones, sulfonamides (to treat infection)
- phenothiazines (to treat serious emotional problems)
If you are taking any prescription or non-prescription medicines, check with your doctor to make sure you can use Tretinoin Cream, USP (Emollient) with them.
We do not know if Tretinoin Cream, USP (Emollient) is passed to infants through breast milk. Therefore, tell your doctor if you are breast feeding.
How should I use Tretinoin Cream, USP (Emollient)?
Use Tretinoin Cream, USP (Emollient) as part of a total skin care and sunlight avoidance program. Follow your doctor's instructions on how to use Tretinoin Cream, USP (Emollient). Tretinoin Cream, USP (Emollient) is usually applied to the face once a day in the evening, following the 3 steps listed below:
- Gently wash your face with a mild soap.
- Pat the skin dry and wait 20-30 minutes before applying Tretinoin Cream, USP (Emollient).
- Apply only a pea-sized amount of Tretinoin Cream, USP (Emollient) to the face at one time. It should be enough to cover the affected area lightly.
Be especially careful when applying Tretinoin Cream, USP (Emollient) to avoid your eyes, ears, nostrils, angles of the nose, and mouth. Tretinoin Cream, USP (Emollient) may cause severe redness, itching, burning, stinging, and peeling if used on these areas.
Using too much Tretinoin Cream, USP (Emollient) may increase discomfort and skin redness and peeling.
You may use cosmetics one hour after applying Tretinoin Cream, USP (Emollient). If you do, be sure to clean your face before applying Tretinoin Cream, USP (Emollient) again. Skin moisturizers should be used at least every morning to protect the treated areas from dryness.
Use sunscreen and wear protective clothing to protect the treated areas from sunlight. If you sunburn easily, or if you spend a lot of time exposed to sunlight, be especially careful to protect your skin.
What should I avoid while using Tretinoin Cream, USP (Emollient)?
Tretinoin Cream, USP (Emollient) can make your treated skin more sensitive to sunlight. Therefore, keep out of the sunlight as much as possible and do not use sunlamps. Avoid as much as possible products that can increase skin irritation, such as:
- other skin medicines
- medicated or abrasive (rough) soaps
- permanent wave solutions
- chemical hair removers or waxes
- electrolysis
- products with alcohol, spices, astringents, or lime
- cleansers, shampoos, or cosmetics with a strong drying effect
- other products that may irritate your skin
What are the possible side effects of Tretinoin Cream, USP (Emollient)?
You may feel brief warmth or stinging on your skin after you use Tretinoin Cream, USP (Emollient). Most patients report peeling, dry skin, burning, stinging, itching, and redness. These are usually mild to moderate and occur early in treatment. Contact your doctor if the side effects are a problem.
General advice about prescription medicines
Medicines are sometimes prescribed for conditions that are not mentioned in patient information leaflets. Only use Tretinoin Cream, USP (Emollient) to treat the condition that your doctor has prescribed it for. Do not give Tretinoin Cream, USP (Emollient) to other people. It may harm them.
This leaflet summarizes the most important information about Tretinoin Cream, USP (Emollient). If you would like more information, talk with your doctor. You can ask your pharmacist or doctor for information about Tretinoin Cream, USP (Emollient) that is written for health professionals.
|
TRETINOIN
tretinoin cream |
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
| Labeler - Spear Dermatology Products (066551941) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| DPT Laboratories | 832224526 | MANUFACTURE(66530-247) | |