Label: SATOGESIC HOT- capsaicin patch
- NDC Code(s): 49873-608-01
- Packager: Sato Pharmaceutical Co., Ltd.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated November 10, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
-
WARNINGS
Warnings
For external use only
Do not use
■ on wounds, or on damaged, or irritated skin
■ on children under 12 years with arthritis-like conditions unless directed by a doctor
When using this product
■ avoid contact with eyes or mucous membranes ■ do not cover with any additional wrap - OTHER SAFETY INFORMATION
-
DOSAGE & ADMINISTRATION
Directions
■ Adults and children 2 years of age and over:
■ strip off the polyethylene film and place adhesive pad over affected area
■ pad may be cut with scissors for small areas such as fingers
■ change pad 1 or 2 times a day
■ apply to affected area not more than 3 or 4 times daily
■ Children under 2 years of age: do not use, consult a doctor
-
INACTIVE INGREDIENT
Inactive ingredients
Butylparaben, carbomer, carboxymethylcellulose sodium, dried aluminum hydroxide gel, edetate disodium, glycerin, malic acid, methacrylic acid and n-butyl acrylate copolymer, methylparaben, partially neutralized polyacrylate, polyethylene glycol 3000 hydrogenated castor oil, polyvinyl alcohol, purified water, sorbitol, titanium dioxide
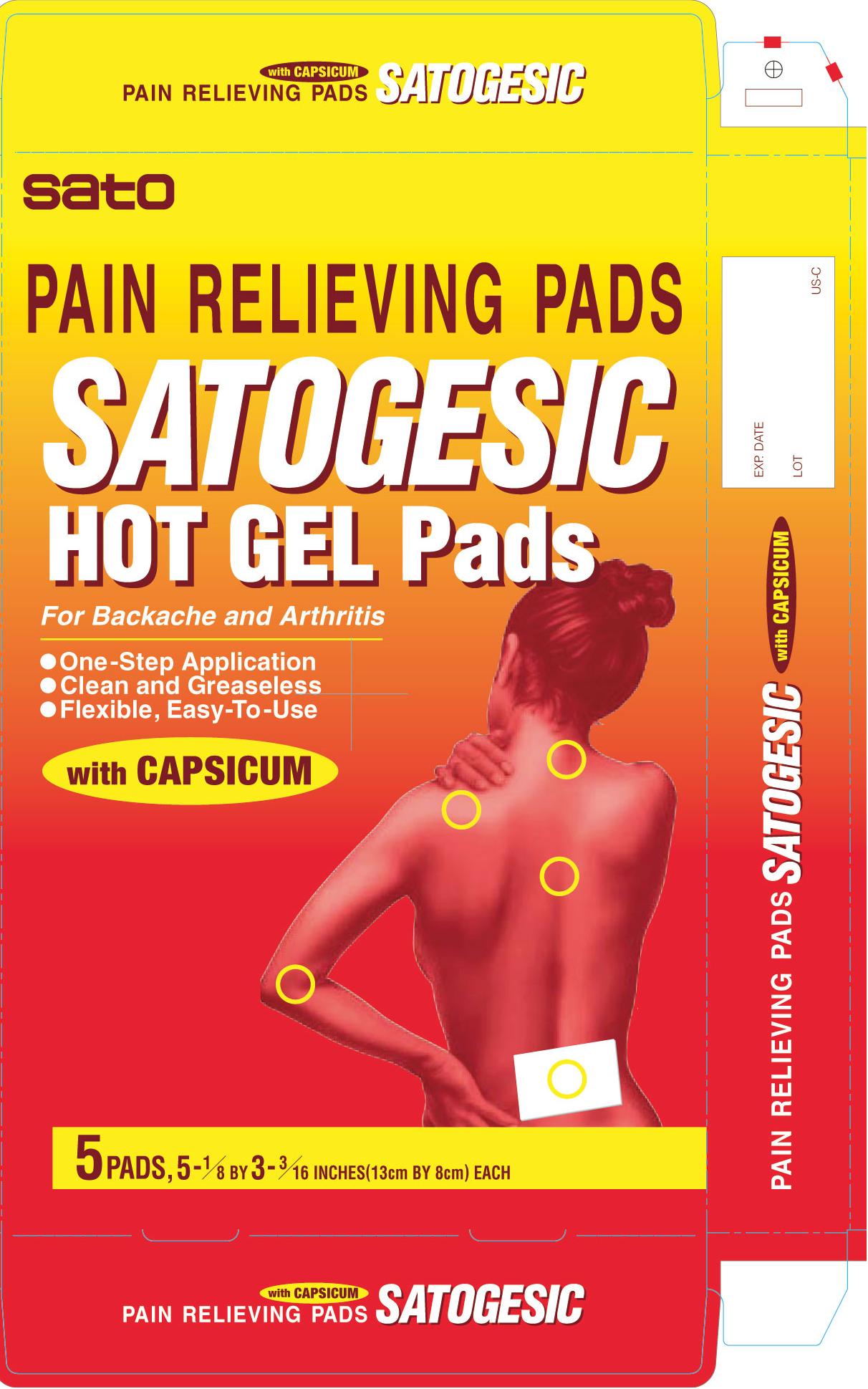
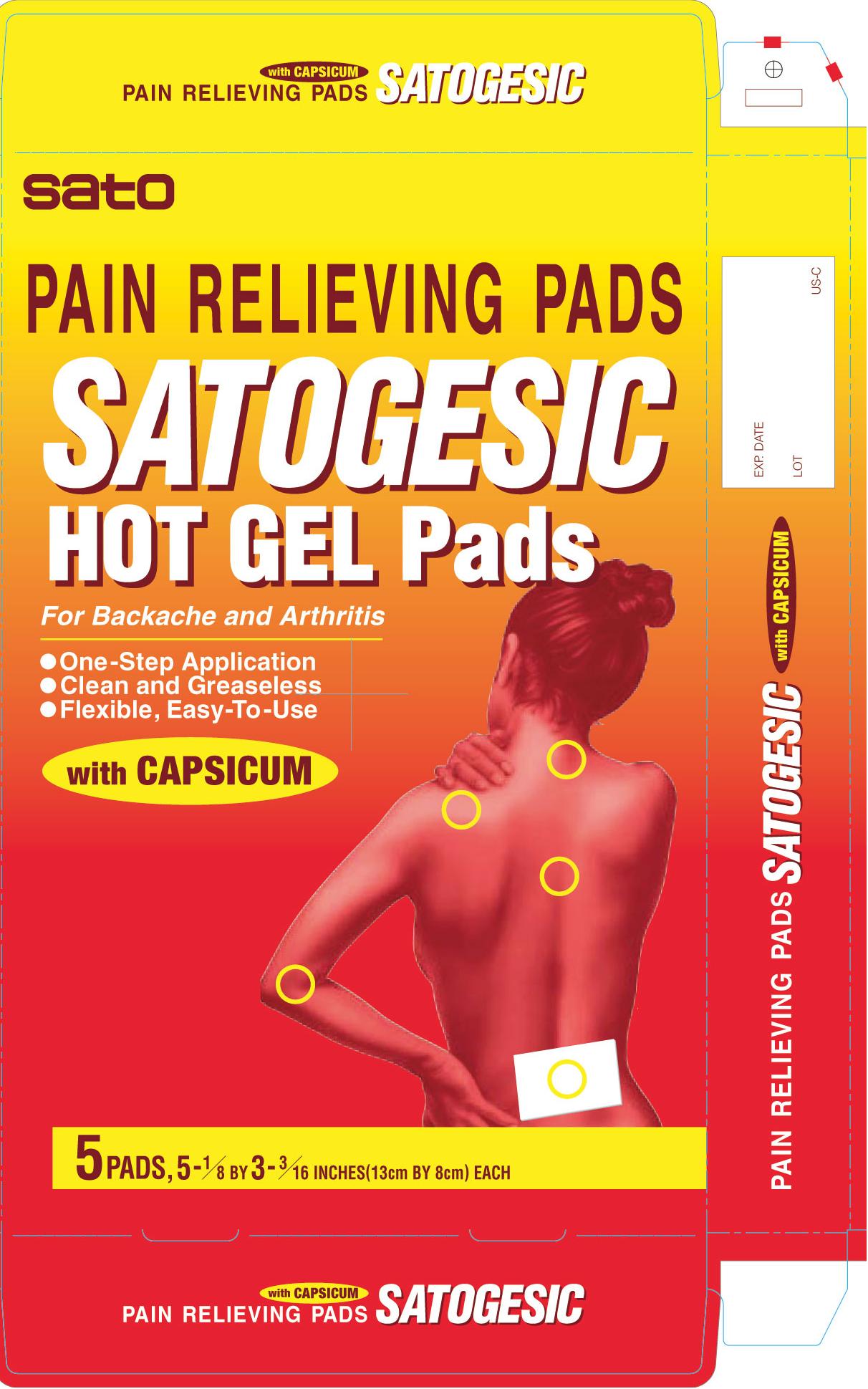
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
SATOGESIC HOT
capsaicin patchProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:49873-608 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CAPSAICIN (UNII: S07O44R1ZM) (CAPSAICIN - UNII:S07O44R1ZM) CAPSAICIN 0.25 mg Inactive Ingredients Ingredient Name Strength BUTYL ACRYLATE/METHACRYLIC ACID COPOLYMER (2% METHACRYLIC ACID) (UNII: Q984S31CYH) CARBOXYMETHYLCELLULOSE SODIUM (UNII: K679OBS311) POLYVINYL ALCOHOL (UNII: 532B59J990) SORBITOL (UNII: 506T60A25R) GLYCERIN (UNII: PDC6A3C0OX) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) EDETATE DISODIUM (UNII: 7FLD91C86K) ALGELDRATE (UNII: 03J11K103C) MALIC ACID (UNII: 817L1N4CKP) WATER (UNII: 059QF0KO0R) BUTYLPARABEN (UNII: 3QPI1U3FV8) METHYLPARABEN (UNII: A2I8C7HI9T) POLYOXYL 60 HYDROGENATED CASTOR OIL (UNII: 02NG325BQG) CARBOMER INTERPOLYMER TYPE B (ALLYL PENTAERYTHRITOL CROSSLINKED) (UNII: 132584PQMO) ACRYLIC ACID/SODIUM ACRYLATE COPOLYMER (1:1; 600 MPA.S AT 0.2%) (UNII: M4PPW69Y4H) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:49873-608-01 1 in 1 CARTON 05/17/2012 1 5 in 1 POUCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M017 11/22/2006 Labeler - Sato Pharmaceutical Co., Ltd. (690575642) Establishment Name Address ID/FEI Business Operations Sato Pharmaceutical Co., Ltd. 715699133 manufacture(49873-608) , label(49873-608) , pack(49873-608)