ACTIMMUNE - interferon gamma-1b injection, solution
InterMune, Inc.
----------
ACTIMMUNE® (Interferon gamma-1b)
DESCRIPTION
ACTIMMUNE® (Interferon gamma-1b), a biologic response modifier, is a single-chain polypeptide containing 140 amino acids. Production of ACTIMMUNE is achieved by fermentation of a genetically engineered Escherichia coli bacterium containing the DNA which encodes for the human protein. Purification of the product is achieved by conventional column chromatography. ACTIMMUNE is a highly purified sterile solution consisting of non-covalent dimers of two identical 16,465 dalton monomers; with a specific activity of 20 million International Units (IU)/mg (2x106 IU per 0.5 mL) which is equivalent to 30 million units/mg.
ACTIMMUNE is a sterile, clear, colorless solution filled in a single-use vial for subcutaneous injection. Each 0.5 mL of ACTIMMUNE contains: 100 mcg (2 million IU) of Interferon gamma-1b formulated in 20 mg mannitol, 0.36 mg sodium succinate, 0.05 mg polysorbate 20 and Sterile Water for Injection. Note that the above activity is expressed in International Units (1 million IU/50mcg). This is equivalent to what was previously expressed as units (1.5 million U/50mcg).
CLINICAL PHARMACOLOGY
General
Interferons bind to specific cell surface receptors and initiate a sequence of intracellular events that lead to the transcription of interferon-stimulated genes. The three major groups of interferons (alpha, beta, gamma) have partially overlapping biological activities that include immunoregulation such as increased resistance to microbial pathogens and inhibition of cell proliferation. Type 1 interferons (alpha and beta) bind to the alpha/beta receptor. Interferon-gamma binds to a different cell surface receptor and is classified as Type 2 interferon. Specific effects of interferon-gamma include the enhancement of the oxidative metabolism of macrophages, antibody dependent cellular cytotoxicity (ADCC), activation of natural killer (NK) cells, and the expression of Fc receptors and major histocompatibility antigens.
Chronic Granulomatous Disease (CGD) is an inherited disorder of leukocyte function caused by defects in the enzyme complex responsible for phagocyte superoxide generation. ACTIMMUNE does not increase phagocyte superoxide production even in treatment responders.1
In severe, malignant osteopetrosis (an inherited disorder characterized by an osteoclast defect, leading to bone overgrowth, and by deficient phagocyte oxidative metabolism), a treatment-related enhancement of superoxide production by phagocytes was observed. ACTIMMUNE was found to enhance osteoclast function in vivo.2-4
In both disorders, the exact mechanism(s) by which ACTIMMUNE has a treatment effect has not been established. Changes in superoxide levels during ACTIMMUNE therapy do not predict efficacy and should not be used to assess patient response to therapy.
Pharmacokinetics
The intravenous, intramuscular, and subcutaneous pharmacokinetics of ACTIMMUNE have been investigated in 24 healthy male subjects following single-dose administration of 100 mcg/m2. ACTIMMUNE is rapidly cleared after intravenous administration (1.4 liters/minute) and slowly absorbed after intramuscular or subcutaneous injection. After intramuscular or subcutaneous injection, the apparent fraction of dose absorbed was greater than 89%. The mean elimination half-life after intravenous administration of 100 mcg/m2 in healthy male subjects was 38 minutes. The mean elimination half-lives for intramuscular and subcutaneous dosing with 100 mcg/m2 were 2.9 and 5.9 hours, respectively. Peak plasma concentrations, determined by ELISA, occurred approximately 4 hours (1.5 ng/mL) after intramuscular dosing and 7 hours (0.6 ng/mL) after subcutaneous dosing. Multiple dose subcutaneous pharmacokinetic studies were conducted in 38 healthy male subjects. There was no accumulation of ACTIMMUNE after 12 consecutive daily injections of 100 mcg/m2. Pharmacokinetic studies in patients with Chronic Granulomatous Disease have not been performed.
Trace amounts of interferon-gamma were detected in the urine of squirrel monkeys following intravenous administration of 500 mcg/kg. Interferon-gamma was not detected in the urine of healthy human volunteers following administration of 100 mcg/m2 of ACTIMMUNE by the intravenous, intramuscular and subcutaneous routes. In vitro perfusion studies utilizing rabbit livers and kidneys demonstrate that these organs are capable of clearing interferon-gamma from perfusate. Studies of the administration of interferon-gamma to nephrectomized mice and squirrel monkeys demonstrate a reduction in clearance of interferon-gamma from blood; however, prior nephrectomy did not prevent elimination.
Effects in Chronic Granulomatous Disease
A randomized, double-blind, placebo-controlled study of ACTIMMUNE (Interferon gamma-1b) in patients with Chronic Granulomatous Disease (CGD), was performed to determine whether ACTIMMUNE administered subcutaneously on a three times weekly schedule could decrease the incidence of serious infectious episodes and improve existing infectious and inflammatory conditions in patients with Chronic Granulomatous Disease. One hundred twenty-eight eligible patients were enrolled on this study including patients with different patterns of inheritance. Most patients received prophylactic antibiotics. Patients ranged in age from 1 to 44 years with the mean age being 14.6 years. The study was terminated early following demonstration of a highly statistically significant benefit of ACTIMMUNE therapy compared to placebo with respect to time to serious infection (p=0.0036), the primary endpoint of the investigation. Serious infection was defined as a clinical event requiring hospitalization and the use of parenteral antibiotics. The final analysis provided further support for the primary endpoint (p=0.0006). There was a 67 percent reduction in relative risk of serious infection in patients receiving ACTIMMUNE (n=63) compared to placebo (n=65). Additional supportive evidence in the number of primary serious infections in the ACTIMMUNE group (30 on placebo versus 14 on ACTIMMUNE, p=0.002) and the total number and rate of serious infections including recurrent events (56 on placebo versus 20 on ACTIMMUNE, p=<0.0001). Moreover, the length of hospitalization for the treatment of all clinical events provided evidence highly supportive of an ACTIMMUNE treatment benefit. Placebo patients required three times as many inpatient hospitalization days for treatment of clinical events compared to patients receiving ACTIMMUNE (1493 versus 497 total days, p=0.02). An ACTIMMUNE treatment benefit with respect to time to serious infection was consistently demonstrated in all subgroup analyses according to stratification factors, including pattern of inheritance, use of prophylactic antibiotics, as well as age. There was a 67 percent reduction in relative risk of serious infection in patients receiving ACTIMMUNE compared to placebo across all groups. The beneficial effect of ACTIMMUNE therapy was observed throughout the entire study, in which the mean duration of ACTIMMUNE administration was 8.9 months/patient.
Effects in Osteopetrosis
A controlled, randomized study in patients with severe, malignant osteopetrosis was conducted with ACTIMMUNE administered subcutaneously three times weekly. Sixteen patients were randomized to receive either ACTIMMUNE plus calcitriol (n=11), or calcitriol alone (n=5). Patients ranged in age from 1 month to 8 years, mean 1.5 years. Treatment failure was considered to be disease progression as defined by 1) death, 2) significant reduction in hemoglobin or platelet counts, 3) a serious bacterial infection requiring antibiotics, or 4) a 50 dB decrease in hearing or progressive optic atrophy. The median time to disease progression was significantly delayed in the ACTIMMUNE plus calcitriol arm versus calcitriol alone. In the treatment arm, the median was not reached. Based on the observed data, however, the median time to progression in this arm was at least 165 days versus a median of 65 days in the calcitriol alone arm. In an analysis which combined data from a second study, 19 of 24 patients treated with ACTIMMUNE plus or minus calcitriol for at least 6 months had reduced trabecular bone volume compared to baseline.
INDICATIONS AND USAGE
ACTIMMUNE is indicated for reducing the frequency and severity of serious infections associated with Chronic Granulomatous Disease.
ACTIMMUNE is indicated for delaying time to disease progression in patients with severe, malignant osteopetrosis.
CONTRAINDICATIONS
ACTIMMUNE is contraindicated in patients who develop or have known hypersensitivity to interferon-gamma, E. coli derived products, or any component of the product.
WARNINGS
Cardiovascular Disorders
Acute and transient "flu-like" symptoms such as fever and chills induced by ACTIMMUNE at doses of 250 mcg/m2/day (greater than 10 times the weekly recommended dose) or higher may exacerbate pre-existing cardiac conditions. ACTIMMUNE should be used with caution in patients with pre-existing cardiac conditions, including ischemia, congestive heart failure or arrhythmia.
Neurologic Disorders
Decreased mental status, gait disturbance and dizziness have been observed, particularly in patients receiving ACTIMMUNE doses greater than 250 mcg/m2/day (greater than 10 times the weekly recommended dose). Most of these abnormalities were mild and reversible within a few days upon dose reduction or discontinuation of therapy. Caution should be exercised when administering ACTIMMUNE to patients with seizure disorders or compromised central nervous system function.
Bone Marrow Toxicity
Reversible neutropenia and thrombocytopenia that can be severe and may be dose related have been observed during ACTIMMUNE therapy. Caution should be exercised when administering ACTIMMUNE to patients with myelosuppression.
Hepatic Toxicity
Elevations of AST and/or ALT (up to 25-fold) have been observed during ACTIMMUNE therapy. The incidence appeared to be higher in patients less than 1 year of age compared to older children. The transaminase elevations were reversible with reduction in dosage or interruption of ACTIMMUNE treatment. Patients begun on ACTIMMUNE before age one year should receive monthly assessments of liver function. If severe hepatic enzyme elevations develop, ACTIMMUNE dosage should be modified (see DOSAGE AND ADMINISTRATION: Dose Modification).
PRECAUTIONS
General
Isolated cases of acute serious hypersensitivity reactions have been observed in patients receiving ACTIMMUNE. If such an acute reaction develops the drug should be discontinued immediately and appropriate medical therapy instituted. Transient cutaneous rashes have occurred in some patients following injection but have rarely necessitated treatment interruption.
Information for Patients
Patients being treated with ACTIMMUNE and/or their parents should be informed regarding the potential benefits and risks associated with treatment. If home use is determined to be desirable by the physician, instructions on appropriate use should be given, including review of the contents of the Patient Information Insert. This information is intended to aid in the safe and effective use of the medication. It is not a disclosure of all possible adverse or intended effects.
If home use is prescribed, a puncture resistant container for the disposal of used syringes and needles should be supplied to the patient. Patients should be thoroughly instructed in the importance of proper disposal and cautioned against any reuse of needles and syringes. The full container should be disposed of according to the directions provided by the physician (see Patient Information Insert).
The most common adverse experiences occurring with ACTIMMUNE therapy are "flu-like" or constitutional symptoms such as fever, headache, chills, myalgia or fatigue (see ADVERSE REACTIONS) which may decrease in severity as treatment continues. Some of the "flu-like" symptoms may be minimized by bedtime administration. Acetaminophen may be used to prevent or partially alleviate the fever and headache.
Laboratory Tests
In addition to those tests normally required for monitoring patients with Chronic Granulomatous Disease and osteopetrosis, the following laboratory tests are recommended for all patients on ACTIMMUNE (Interferon gamma-1b) therapy prior to the beginning of and at three month intervals during treatment (see WARNINGS: Bone Marrow and Hepatic Toxicity).
- Hematologic tests - including complete blood counts, differential and platelet counts
- Blood chemistries - including renal and liver function tests. In patients less than 1 year of age, liver function tests should be measured monthly (see ADVERSE REACTIONS: Post-Marketing Experience).
- Urinalysis
Drug Interactions
Interactions between ACTIMMUNE and other drugs have not been fully evaluated. Caution should be exercised when administering ACTIMMUNE in combination with other potentially myelosuppressive agents (see WARNINGS).
Preclinical studies in rodents using species-specific interferon-gamma have demonstrated a decrease in hepatic microsomal cytochrome P-450 concentrations. This could potentially lead to a depression of the hepatic metabolism of certain drugs that utilize this degradative pathway.
Carcinogenesis, Mutagenesis and Impairment of Fertility
Carcinogenesis: ACTIMMUNE has not been tested for its carcinogenic potential.
Mutagenesis: Ames tests using five different tester strains of bacteria with and without metabolic activation revealed no evidence of mutagenic potential. ACTIMMUNE was tested in a micronucleus assay for its ability to induce chromosomal damage in bone marrow cells of mice following two intravenous doses of 20 mg/kg. No evidence of chromosomal damage was noted.
Impairment of Fertility: Female cynomolgus monkeys treated with daily subcutaneous doses of 30 or 150 mcg/kg ACTIMMUNE (approximately 20 and 100 times the human dose) exhibited irregular menstrual cycles or absence of cyclicity during treatment. Similar findings were not observed in animals treated with 3 mcg/kg ACTIMMUNE.
Female mice receiving recombinant murine IFN-gamma (rmulFN-gamma) at 32 times the maximum recommended clinical dose of ACTIMMUNE for 4 weeks via intramuscular injection exhibited an increased incidence of atretic ovarian follicles.
Male cynomolgus monkeys treated intravenously for 4 weeks with 8 times the maximum recommended clinical dose of ACTIMMUNE exhibited decreased spermatogenesis. The impact of this finding on fertility is not known. Male mice receiving rmulFN-gamma at 32 times the maximum recommended clinical dose of ACTIMMUNE for 4 weeks via intramuscular injection exhibited decreased spermatogenesis.
Male mice treated subcutaneously with rmuIFN-gamma from shortly after birth through puberty, with 280 times the maximum recommended clinical dose of ACTIMMUNE exhibited profound yet reversible decreases in sperm counts and fertility, and an increase in the number of abnormal sperm.
The clinical significance of these findings observed following treatment of mice with rmulFN-gamma is uncertain.
Pregnancy
Teratogenic Effects: Pregnancy Category C. ACTIMMUNE has shown an increased incidence of abortions in primates when given in doses approximately 100 times the human dose. A study in pregnant primates treated with subcutaneous doses 2-100 times the human dose failed to demonstrate teratogenic activity for ACTIMMUNE.
Female mice treated subcutaneously with rmulFN-gamma at 280 times the maximum recommended clinical dose of ACTIMMUNE from shortly after birth through puberty but not during pregnancy had offspring which exhibited decreased body weight during the lactation period. The clinical significance of this finding observed following treatment of mice with rmulFN-gamma is uncertain.
There are no adequate and well-controlled studies in pregnant women. ACTIMMUNE should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Nursing Mothers
It is not known whether ACTIMMUNE is excreted in human milk. Because many drugs are excreted in human milk and because of the potential for serious adverse reactions in nursing infants from ACTIMMUNE, a decision should be made whether to discontinue nursing or to discontinue the drug, dependent upon the importance of the drug to the mother.
ADVERSE REACTIONS
The following data on adverse reactions are based on the subcutaneous administration of ACTIMMUNE at a dose of 50 mcg/m2, three times weekly, in patients with Chronic Granulomatous Disease (CGD) during an investigational trial in the United States and Europe.
The most common adverse events observed in patients with CGD are shown in the following table:
| Percent of Patients | ||||||
|---|---|---|---|---|---|---|
| Clinical Toxicity | ACTIMMUNE CGD (n=63) | Placebo CGD (n=65) | ||||
| Fever | 52 | 28 | ||||
| Headache | 33 | 9 | ||||
| Rash | 17 | 6 | ||||
| Chills | 14 | 0 | ||||
| Injection site erythema or tenderness | 14 | 2 | ||||
| Fatigue | 14 | 11 | ||||
| Diarrhea | 14 | 12 | ||||
| Vomiting | 13 | 5 | ||||
| Nausea | 10 | 2 | ||||
| Myalgia | 6 | 0 | ||||
| Arthralgia | 2 | 0 | ||||
| Injection site pain | 0 | 2 | ||||
Miscellaneous adverse events which occurred infrequently in patients with CGD and may have been related to underlying disease included back pain (2 percent versus 0 percent), abdominal pain (8 percent versus 3 percent) and depression (3 percent versus 0 percent) for ACTIMMUNE and placebo treated patients, respectively.
Similar safety data were observed in 34 patients with severe malignant osteopetrosis.
ACTIMMUNE has also been evaluated in additional disease states in studies in which patients have generally received higher doses (>100 mcg/m2/three times weekly) administered by intramuscular or subcutaneous injection, or intravenous infusion. All of the previously described adverse reactions which occurred in patients with Chronic Granulomatous Disease have also been observed in patients receiving higher doses. Adverse reactions not observed in patients with Chronic Granulomatous Disease but reported in patients receiving ACTIMMUNE (Interferon gamma-1b) in other studies include: Cardiovascular—hypotension, syncope, tachyarrhythmia, heart block, heart failure, and myocardial infarction. Central Nervous System—confusion, disorientation, gait disturbance, Parkinsonian symptoms, seizure, hallucinations, and transient ischemic attacks. Gastrointestinal—dyspepsia, hepatic insufficiency, gastrointestinal bleeding, and pancreatitis, including pancreatitis with fatal outcome. General Disorders and Administration Site Conditions—malaise, injection site hemorrhage. Hematologic—deep venous thrombosis and pulmonary embolism. Immunological—increased autoantibodies, lupus-like syndrome. Metabolic—hyponatremia, hyperglycemia and hypertriglyceridemia. Musculoskeletal—clubbing, muscle spasms. Pulmonary—tachypnea, bronchospasm, and interstitial pneumonitis. Renal—reversible renal insufficiency. Other—chest discomfort, exacerbation of dermatomyositis.
Abnormal Laboratory Test Values: Elevations of ALT and AST, neutropenia, thrombocytopenia, and proteinuria have been observed (see WARNINGS and PRECAUTIONS: Laboratory Tests).
No neutralizing antibodies to ACTIMMUNE have been detected in any Chronic Granulomatous Disease patients receiving ACTIMMUNE.
Post-Marketing Experience
Children with CGD less than 3 years of age:
Data on the safety and activity of ACTIMMUNE in 37 patients under the age of 3 years was pooled from four uncontrolled post-marketing studies. The rate of serious infections per patient-year in this uncontrolled group was similar to the rate observed in the ACTIMMUNE treatment groups in controlled trials. Developmental parameters (height, weight and endocrine maturation) for this uncontrolled group conformed to national normative scales before and during ACTIMMUNE therapy.
In 6 of the 10 patients receiving ACTIMMUNE therapy before age one year 2-fold to 25-fold elevations from baseline of AST and/or ALT were observed. These elevations occurred as early as 7 days after starting treatment. Treatment with ACTIMMUNE was interrupted in all 6 of these patients and was restarted at a reduced dosage in 4. Liver transaminase values returned to baseline in all patients and transaminase elevation recurred in one patient upon ACTIMMUNE rechallenge. An 11-fold alkaline phosphatase elevation and hypokalemia in one patient and neutropenia (ANC=525 cells/mm3) in another patient resolved with interruption of ACTIMMUNE treatment and did not recur with rechallenge.
In the post-marketing safety database clinically significant adverse events observed during ACTIMMUNE therapy in children under the age of three years (n=14) included: two cases of hepatomegaly, and one case each of Stevens-Johnson syndrome, granulomatous colitis, urticaria, and atopic dermatitis.
OVERDOSAGE
Central nervous system adverse reactions including decreased mental status, gait disturbance and dizziness have been observed, particularly in cancer patients receiving doses greater than 100 mcg/m2/day by intravenous or intramuscular administration. These abnormalities were reversible within a few days upon dose reduction or discontinuation of therapy. Reversible neutropenia, elevation of hepatic enzymes and of triglycerides, and thrombocytopenia have also been observed.
DOSAGE AND ADMINISTRATION
The recommended dosage of ACTIMMUNE for the treatment of patients with Chronic Granulomatous Disease and severe, malignant osteopetrosis is 50 mcg/m2 (1 million IU/m2) for patients whose body surface area is greater than 0.5 m2 and 1.5 mcg/kg/dose for patients whose body surface area is equal to or less than 0.5 m2. Note that the above activity is expressed in International Units (1 million IU/50mcg). This is equivalent to what was previously expressed as units (1.5 million U/50mcg). Injections should be administered subcutaneously three times weekly (for example, Monday, Wednesday, Friday). The optimum sites of injection are the right and left deltoid and anterior thigh. ACTIMMUNE can be administered by a physician, nurse, family member or patient when trained in the administration of subcutaneous injections. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
The formulation does not contain a preservative. A vial of ACTIMMUNE is suitable for a single use only. The unused portion of any vial should be discarded. Higher doses are not recommended. Safety and efficacy has not been established for ACTIMMUNE given in doses greater or less than the recommended dose of 50 mcg/m2. The minimum effective dose of ACTIMMUNE has not been established.
ACTIMMUNE should not be mixed with other drugs in the same syringe.
HOW SUPPLIED
ACTIMMUNE (Interferon gamma-1b) is a sterile, clear, colorless solution filled in a single-use vial for subcutaneous injection. Each 0.5 mL of ACTIMMUNE contains: 100 mcg (2 million IU) of Interferon gamma-1b, formulated in 20 mg mannitol, 0.36 mg sodium succinate, 0.05 mg polysorbate 20 and Sterile Water for Injection.
Single vial (NDC 64116-011-01)
Cartons of 12 (NDC 64116-011-12)
Stability and Storage
Vials of ACTIMMUNE must be placed in a 2-8°C (36-46°F) refrigerator immediately upon receipt to ensure optimal retention of physical and biochemical integrity. DO NOT FREEZE. Avoid excessive or vigorous agitation. DO NOT SHAKE. An unentered vial of ACTIMMUNE should not be left at room temperature for a total time exceeding 12 hours prior to use. Vials exceeding this time period should not be returned to the refrigerator; such vials should be discarded.
Do not use beyond the expiration date stamped on the vial.
REFERENCES
- The International Chronic Granulomatous Disease Cooperative Study Group. A controlled trial of interferon gamma to prevent infection in chronic granulomatous disease. N Engl J Med 324 : 509-516, 1991.
- Beard CJ, Key L, Newburger PE, Ezekowitz RAB, et al. Neutrophil defect associated with malignant infantile osteopetrosis. J Lab Clin Med 108 : 498-505, 1986.
- Shankar L, Gerritsen EJA, and Key LL. Osteopetrosis: pathogenesis and rationale for the use of interferon-γ-1b. Biodrugs 7 : 23-29, 1997.
- Key LL, Rodriguiz RM, Willi SM. Long-term treatment of osteopetrosis with recombinant human interferon gamma. N Engl J Med 24 : 1594-1599, 1995.
Manufactured by:
InterMune, Inc.
Brisbane, CA 94005
U.S. License No. 1626
Revised January 2009
©2009 InterMune, Inc. PH01037.04
Information for the Patient/Caregiver
ACTIMMUNE® (Interferon gamma-1b)
DO NOT ADMINISTER ACTIMMUNE UNTIL YOUR PHYSICIAN HAS THOROUGHLY TRAINED YOU IN THE PROPER TECHNIQUES.
ACTIMMUNE is supplied in single-use vials. The unused portion of each vial should be disposed of as instructed by your physician. DO NOT SHAKE.
Your physician will tell you what size needle and syringe to use and also give you instructions on sterile technique. Keep all used syringes and needles out of the reach of children. Follow your physician's instructions on the safe disposal of used syringes and needles.
MEASURING THE DOSE
In measuring the correct dose for injection, be sure to check that the ACTIMMUNE® solution is clear. If the solution is cloudy or hazy, do not inject it, but return the ACTIMMUNE vial to your pharmacist or prescribing physician.
1. Wash your hands thoroughly with soap and water before preparing the medication. This helps prevent infection.
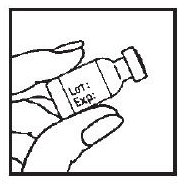
2. Check the date on the ACTIMMUNE vial to be sure the drug has not expired.
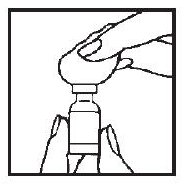
3. Remove the protective plastic cap and wipe the rubber stopper located on top of the ACTIMMUNE® vial with an alcohol swab.
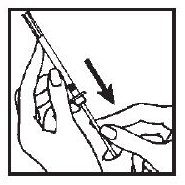
4. Draw air into the syringe by pulling back on the plunger. The amount of air should be equal to the ACTIMMUNE dose.
5. Remove and save the needle guard. Slowly insert the needle straight through the center of the rubber stopper into the ACTIMMUNE vial.
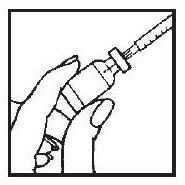
6. Gently push the plunger to discharge the air into the vial.
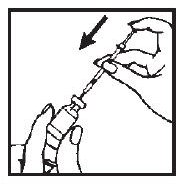
7. Turn the vial upside down with the syringe needle still in it and hold it in one hand. Be sure the tip of the needle is in the solution. Using your other hand slowly pull back on the plunger in a continuous motion until the correct amount of ACTIMMUNE® solution is in the syringe.
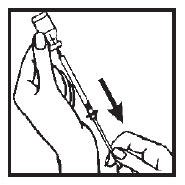
8. Remove the needle from the ACTIMMUNE vial and replace the needle guard until time of administration or injection. Administration should be as soon after filling the syringe as possible; do not store ACTIMMUNE in the syringe.
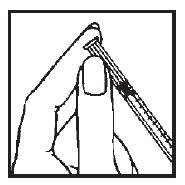
SELECTING THE INJECTION SITE
Your doctor or nurse will teach you how to locate appropriate injection sites. It is very important that you rotate the site of an injection each time you give the medication. Even if you or your child develop a preference for one site—as often happens—you still should rotate the injection site.
Following are the injection sites most often recommended:
■ Upper Arm
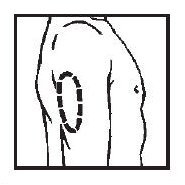
■ Abdomen
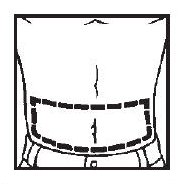
■ Thigh
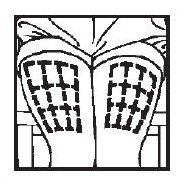
GIVING THE MEDICATION
Your doctor or nurse will provide you with hands-on training on how to give an injection. Needles and syringes should be used only once to ensure sterility of both the needle and the syringe. The following is a review of the steps involved in giving the medication:
1. Cleanse the injection site with an alcohol-saturated cotton ball or cotton swab.
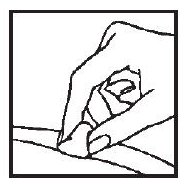
2. Remove the needle guard from the syringe filled with the proper dose of solution and hold the syringe the way you would hold a pencil. Double check that the correct amount of ACTIMMUNE® solution is in the syringe.
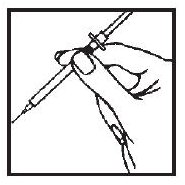
3. Squeeze the skin between your fingers before and during the injection. Insert the needle into the skin at a 45° angle with a quick, firm motion. This hurts less than pushing the needle in slowly.
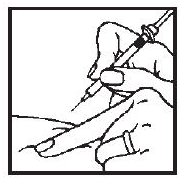
4. After the needle is in, pull back very slightly with one hand on the plunger to see if blood comes into the syringe. This is to be sure that the needle has not entered a blood vessel. If blood does come into the syringe, do not inject the ACTIMMUNE® solution. Withdraw the needle and insert at another location.
5. If blood does not come into the syringe, slowly (within a few seconds) inject the solution by gently pushing the plunger until the syringe is empty.
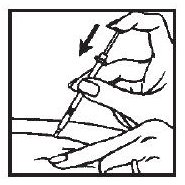
6. Withdraw the needle quickly, pulling it straight out, and apply pressure over the injection site with a dry gauze pad or cotton ball. A drop of blood may appear. Put a Band-Aid® on the injection site if desired.
7. To prevent injury, safely dispose of all used needles and syringes after a single use as instructed by your physician by following these simple steps:
- ■
- Place all used needles and syringes in a hard, plastic container with a screw-on cap, or a metal container with a plastic lid, such as a coffee can properly labeled as to content. If a metal container is used, cut a small hole in the plastic lid and tape the lid to the metal container. When the metal container is full, cover the hole with tape and throw it away. If a hard, plastic container is used, always screw the cap on tightly after each use. When the plastic container is full, tape around the cap and throw it away.
- ■
- Do not use glass or clear, plastic containers, or any container that will be recycled or returned to a store.
- ■
- Always store the container out of the reach of children.
- ■
- Please check with your doctor, nurse or pharmacist for other suggestions. There may be special state and local laws that they will discuss with you.
8. Occasionally a problem may develop at the injection site. If you notice any of the following signs or symptoms, contact your doctor or nurse:
- ■
- A lump or swelling that doesn't go away
- ■
- Bruising that doesn't go away
- ■
- Any signs of infection or inflammation at an injection site (pus, persistent redness, surrounding skin that is hot to the touch, persistent pain after the injection)
STORAGE
ACTIMMUNE® (Interferon gamma-1b) must be refrigerated immediately. Refrigerate at 36° to 46° Fahrenheit (2° to 8° Centigrade). DO NOT FREEZE.
ACTIMMUNE is supplied in single-use vials. The unused portion of each vial should be disposed of according to state and local regulations as instructed by your physician. If you have any questions, contact your physician.
InterMune, Inc.
Brisbane, CA 94005
PA01022.01
November 2006
PACKAGE LABEL – PRINCIPAL DISPLAY PANEL – VIAL
NDC 64116-011-01
ACTIMMUNE®
(Interferon gamma-1b)
100 mcg (2 million IU)/0.5 mL
0.5 mL Single-use vial
US License No.: 1626
Manufactured by:
Intermune, Inc.
Brisbane, CA 94005
DOSAGE AND ADMINISTRATION:
See package insert.
DO NOT SHAKE.
STORAGE:
Refrigerate at
2° to 8°C/
36° to 46°F.
DO NOT FREEZE.
Rx ONLY
LB0161.6

PACKAGE LABEL – PRINCIPAL DISPLAY PANEL – CARTON
KEEP REFRIGERATED
NDC 64116-011-12
ACTIMMUNE®
(Interferon gamma-1b)
How supplied: Each carton contains 12 single-use
vials of Interferon gamma-1b, ACTIMMUNE.
Contents: Each 0.5 mL vial of ACTIMMUNE
contains 100 mcg (2 million IU) Interferon
gamma-1b formulated in 20 mg mannitol, 0.36 mg
sodium succinate, 0.05 mg polysorbate 20 and
Sterile Water for Injection for subcutaneous
injection. ACTIMMUNE contains no preservatives.
Dosage and administration: See
package insert for full prescribing
information. ACTIMMUNE is
suitable for single-use only.
DO NOT SHAKE.
Storage: Refrigerate at
2° to 8°C/36° to 46°F.
DO NOT FREEZE.
ACTIMMUNE®
(Interferon gamma-1b)
Manufactured by:
Intermune, Inc.
Brisbane, CA 94005
US License No.: 1626
INTERMUNE®
KEEP REFRIGERATED
ACTIMMUNE®
(Interferon gamma-1b)
Dispensing pharmacist:
1. Refrigerate
(2° to 8°C/36° to 46°F).
2. Be sure patient information
is enclosed.
3. Remove physician insert
before dispensing.
Rx ONLY
KEEP REFRIGERATED
INTERMUNE®

| ACTIMMUNE
interferon gamma-1b injection, solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - InterMune, Inc. (035491294) |
| Registrant - InterMune, Inc. (035491294) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Boehringer Ingelheim Pharma GmbH & Co KG | 340700520 | MANUFACTURE(64116-011) | |