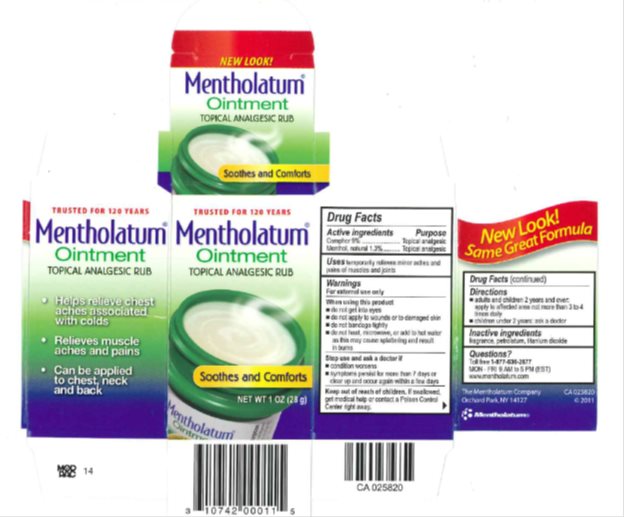
MENTHOLATUM- camphor, menthol ointment
The Mentholatum Company
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Drug Facts
Warnings
For external use only
When using this product
- •
- do not heat
- •
- do not microwave
- •
- do not add to hot water or any container where heating water. May cause splattering and result in burns.
- •
- do not get into eyes
- •
- do not apply to wounds or to damaged skin
- •
- do not bandage tightly
| MENTHOLATUM
camphor, menthol ointment |
||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||
| Labeler - The Mentholatum Company (002105757) |
Revised: 12/2016
Document Id: 83a1534a-79e1-45db-b6f4-e4921183156c
Set id: 0c633ac9-7cc4-477d-8296-2a891ed4a867
Version: 5
Effective Time: 20161206
The Mentholatum Company