BACK AND BODY EXTRA STRENGTH- aspirin, caffeine tablet, film coated
GREENBRIER INTERNATIONAL, INC.
----------
Assured 44-413 Delisted
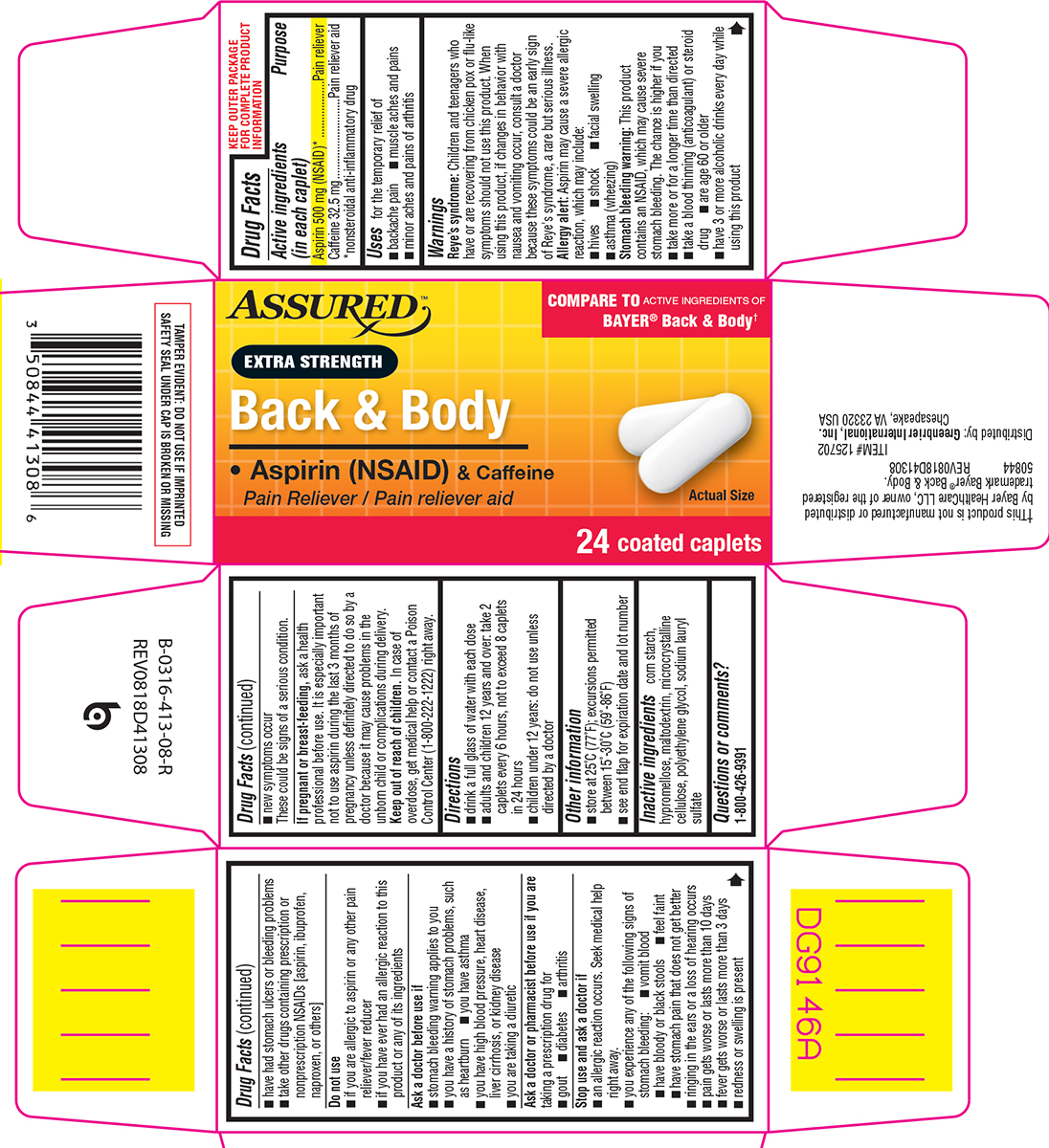
Active ingredients (in each caplet)
Aspirin 500 mg (NSAID)*
Caffeine 32.5 mg
*nonsteroidal anti-inflammatory drug
Uses
for the temporary relief of
- backache pain
- muscle aches and pains
- minor aches and pains of arthritis
Warnings
Reye's syndrome: Children and teenagers who have or are recovering from chicken pox or flu-like symptoms should not use this product. When using this product, if changes in behavior with nausea and vomiting occur, consult a doctor because these symptoms could be an early sign of Reye's syndrome, a rare but serious illness.
Allergy alert: Aspirin may cause a severe allergic reaction, which may include:
- hives
- facial swelling
- shock
- asthma (wheezing)
Stomach bleeding warning: This product contains an NSAID, which may cause severe stomach bleeding. The chance is higher if you
- are age 60 or older
- take more or for a longer time than directed
- have had stomach ulcers or bleeding problems
- take a blood thinning (anticoagulant) or steroid drug
- take other drugs containing prescription or nonprescription NSAIDs [aspirin, ibuprofen, naproxen, or others]
- have 3 or more alcoholic drinks every day while using this product
Do not use
- if you are allergic to aspirin or any other pain reliever/fever reducer
- if you have ever had an allergic reaction to this product or any of its ingredients
Ask a doctor before use if
- stomach bleeding warning applies to you
- you have a history of stomach problems, such as heartburn
- you have asthma
- you have high blood pressure, heart disease, liver cirrhosis, or kidney disease
- you are taking a diuretic
Ask a doctor or pharmacist before use if you are
taking a prescription drug for
- gout
- diabetes
- arthritis
Stop use and ask a doctor if
- an allergic reaction occurs. Seek medical help right away.
- you experience any of the following signs of stomach bleeding:
- vomit blood
- have bloody or black stools
- feel faint
- have stomach pain that does not get better
- ringing in the ears or a loss of hearing occurs
- pain gets worse or lasts more than 10 days
- fever gets worse or lasts more than 3 days
- redness or swelling is present
- new symptoms occur
These could be signs of a serious condition.
Directions
- drink a full glass of water with each dose
- adults and children 12 years and over: take 2 caplets every 6 hours, not to exceed 8 caplets in 24 hours
- children under 12 years: do not use unless directed by a doctor
Other information
- store at 25ºC (77ºF); excursions permitted between 15º-30ºC (59º-86ºF)
- see end flap for expiration date and lot number
Inactive ingredients
corn starch, hypromellose, maltodextrin, microcrystalline cellulose, polyethylene glycol, sodium lauryl sulfate
Principal Display Panel
ASSURED™
COMPARE TO ACTIVE INGREDIENTS OF
BAYER® Back & Body†
EXTRA STRENGTH
Back & Body
• Aspirin (NSAID) & Caffeine
Pain Reliever / Pain reliever aid
Actual Size
24 coated caplets
TAMPER EVIDENT: DO NOT USE IF IMPRINTED
SAFETY SEAL UNDER CAP IS BROKEN OR MISSING
†This product is not manufactured or distributed by Bayer
HealthCare LLC, owner of the registered trademark Bayer®
Back & Body. 50844 REV0818D41308
ITEM# 125702
Distributed by: Greenbrier International, Inc.
Chesapeake, VA 23320 USA
44-413
| BACK AND BODY
EXTRA STRENGTH
aspirin, caffeine tablet, film coated |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Labeler - GREENBRIER INTERNATIONAL, INC. (610322518) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 038154464 | pack(33992-0413) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 832867837 | manufacture(33992-0413) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 832867894 | manufacture(33992-0413) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 967626305 | pack(33992-0413) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 117025878 | manufacture(33992-0413) | |