Label: EYE WASH- water solution
-
Contains inactivated NDC Code(s)
NDC Code(s): 67510-0662-4 - Packager: Kareway Product, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated February 22, 2013
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- Purpose
- Uses
- Warnings
-
Directions
Remove contact lenses before using.
- Do not touch tip of container to any surface to avoid contamination.
- Replace cap after use.
------------------------------------------------------------------------------
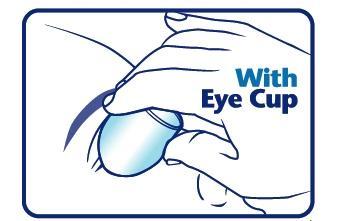
When using an eye cup
- rinse the cup with Pure-aid Eye Wash immediately before each use
- avoid contamination of the rim and inside surfaces of the cup
- fill the cup half full with Pure-aid Eye Wash Solution and apply the cup to the affected eye(s), pressing tightly to prevent spillage
- tilt the head backward. Open eyelids wide and rotate eyeball to thoroughly wash the eye
- rinse cup with clean water after each use
- Other information
- Inactive ingredients
- Package label
-
INGREDIENTS AND APPEARANCE
EYE WASH
water solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:67510-0662 Route of Administration OPHTHALMIC Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength WATER (UNII: 059QF0KO0R) (WATER - UNII:059QF0KO0R) WATER 99.05 mL in 100 mL Inactive Ingredients Ingredient Name Strength BORIC ACID (UNII: R57ZHV85D4) SODIUM BORATE (UNII: 91MBZ8H3QO) SODIUM CHLORIDE (UNII: 451W47IQ8X) HYDROCHLORIC ACID (UNII: QTT17582CB) EDETATE DISODIUM (UNII: 7FLD91C86K) POLYAMINOPROPYL BIGUANIDE (UNII: 322U039GMF) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:67510-0662-4 1 in 1 BOX 1 118 mL in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part349 09/08/2011 Labeler - Kareway Product, Inc. (121840057)


