Label: HUSKY INSTANT HAND SANITIZER solution
-
Contains inactivated NDC Code(s)
NDC Code(s): 63779-056-19, 63779-056-27, 63779-056-77, 63779-056-87 - Packager: Canberra Corporation
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated February 21, 2014
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
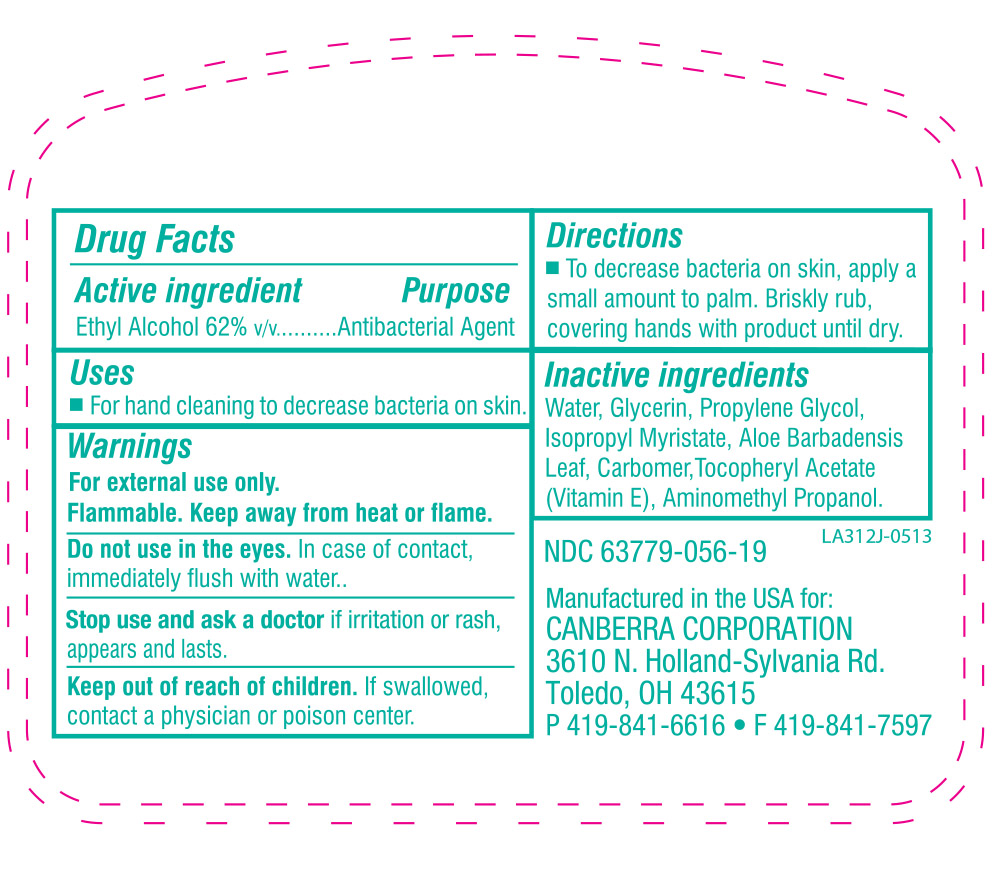
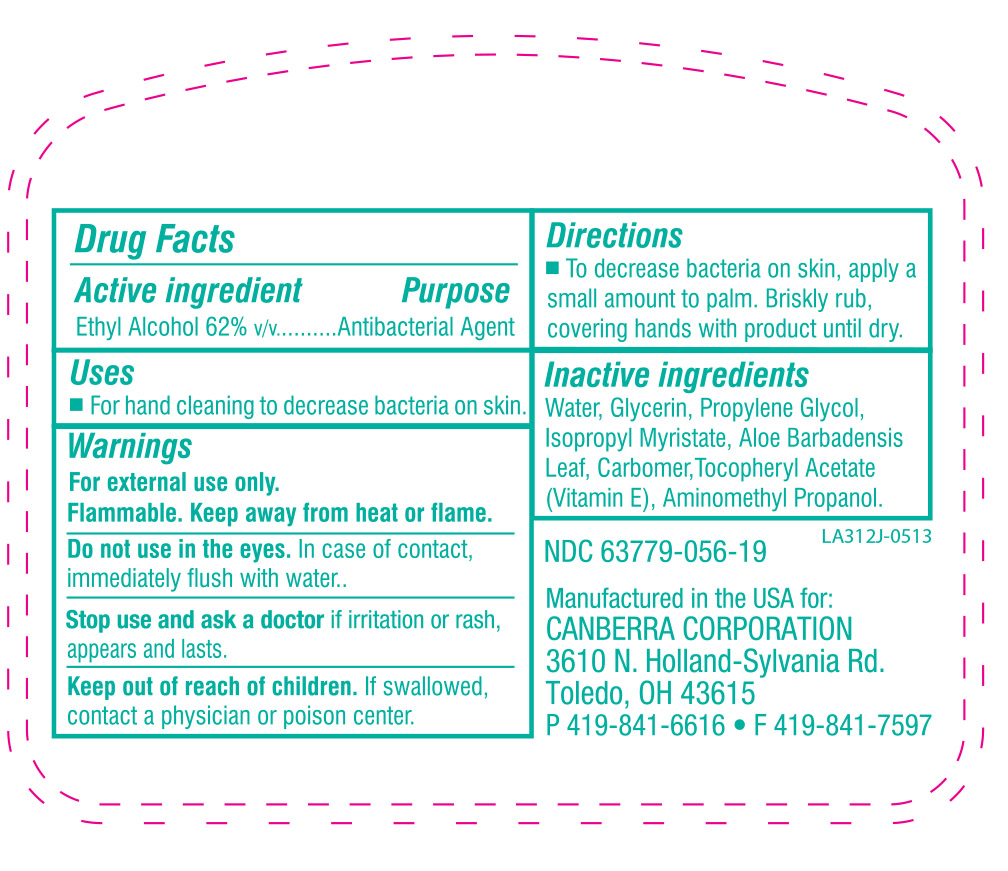
- WARNINGS
- ACTIVE INGREDIENT
- KEEP OUT OF REACH OF CHILDREN
- INACTIVE INGREDIENT
-
DOSAGE & ADMINISTRATION
63779-056-77 & 63779-056-27:
To decrease bacteria on skin, apply a small amount to palm. Briskly rub, covering hands with product until dry.
Children under 6 years of age should be supervised when using this product.
63779-056-87 & 63779-056-19:
To decrease bacteria on skin, apply a small amount to palm. Briskly rub, covering hands with product until dry.
- PURPOSE
-
INDICATIONS & USAGE
For hand cleaning to decrease bacteria on skin that could cause disease. Recommended for repeated use.
Do not use in the eyes. In case of contact, immediately flush with water.
Stop use and ask a doctor if irritation or rash appears and lasts.
Keep out of reach of children. If swallowed, contact a physician or poison center.
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
HUSKY INSTANT HAND SANITIZER
husky instant hand sanitizer solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:63779-056 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 62 mL in 100 mL Inactive Ingredients Ingredient Name Strength POLYACRYLIC ACID (250000 MW) (UNII: 9G2MAD7J6W) WATER (UNII: 059QF0KO0R) GLYCERIN (UNII: PDC6A3C0OX) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) ISOPROPYL MYRISTATE (UNII: 0RE8K4LNJS) AMINOMETHYLPROPANOL (UNII: LU49E6626Q) ALOE VERA LEAF (UNII: ZY81Z83H0X) .ALPHA.-TOCOPHEROL ACETATE, DL- (UNII: WR1WPI7EW8) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:63779-056-19 237 mL in 1 BOTTLE 2 NDC:63779-056-87 29 mL in 1 BOTTLE 3 NDC:63779-056-27 800 mL in 1 BAG 4 NDC:63779-056-77 1100 mL in 1 BOTTLE, PLASTIC Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333A 05/22/2013 Labeler - Canberra Corporation (068080621) Registrant - Kutol Products Company, Inc. (004236139) Establishment Name Address ID/FEI Business Operations Kutol Products Company, Inc. 004236139 manufacture(63779-056) , analysis(63779-056) , pack(63779-056)




 63779-056-19.jpg
63779-056-19.jpg 63779-056-27.jpg
63779-056-27.jpg 63779-056-77.jpg
63779-056-77.jpg 63779-056-87.jpg
63779-056-87.jpg