NODOLOR- isometheptene mucate, dichloralphenazone, and acetaminophen capsule
Macoven Pharmaceuticals
Disclaimer: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
----------
Nodolor™ Capsules
CIV
Liver Warning
This product contains acetaminophen. Severe liver damage may occur if a person takes:
- More than 4,000 mg of acetaminophen in 24 hours
- With other drugs containing acetaminophen
- With 3 or more alcoholic drinks everyday while using this product.
DESCRIPTION
| Each red capsule contains: | |
| Isometheptene Mucate, USP | 65 mg |
| Dichloralphenazone, USP | 100 mg |
| Acetaminophen, USP | 325 mg |
Isometheptene Mucate is a white crystalline powder having a characteristic aromatic odor and bitter taste. It is an unsaturated aliphatic amine with sympathomimetic properties. Its molecular weight is 492.65 and it has the following structural formula:
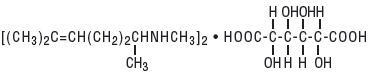
Dichloralphenazone is a white, microcrystalline powder, with slight odor and tastes saline at first, becoming acrid. It is a mild sedative. Its molecular weight is 519.03 and it has the following structural formula:
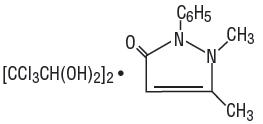
Acetaminophen, a non-salicylate, occurs as a white, odorless, crystalline powder, possessing a slightly bitter taste. Its molecular weight is 151.16 and it has the following structural formula:
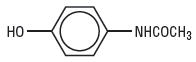
Acetaminophen raises the threshold of painful stimuli, thus exerting an analgesic affect against certain types of headaches.
The inactive ingredients are: Colloidal Silicon Dioxide, FD&C Blue #1, FD&C Red #40, Gelatin, Magnesium Stearate, Microcrystalline Cellulose and Titanium Dioxide.
CLINICAL PHARMACOLOGY
Isometheptene Mucate, a sympathomimetic amine, acts by constricting dilated cranial and cerebral arterioles, thus reducing the stimuli that lead to vascular headaches. Dichloralphenazone, a mild sedative, reduces the patient's emotional reaction to the pain of both vascular and tension headaches. Acetaminophen raises the threshold to painful stimuli, thus exerting an analgesic effect against all types of headaches.
INDICATIONS AND USAGE
For relief of tension and vascular headaches.
| Based on a review of this drug (isometheptene mucate) by the National Academy of Sciences-National Research Council and/or other information, FDA has classified the other indication as "possibly" effective in the treatment of migraine headache. Final classification of the less-than-effective indication requires further investigation. |
CONTRAINDICATIONS
Nodolor™ Capsules is contraindicated in:
- Hypersensitivity or intolerance to any component of this product
- Cardiovascular or cerebrovascular insufficiency, including recent myocardial infarction or stroke
- Glaucoma
- Severe cases of renal disease
- Hypertension
- Organic heart disease
- Peripheral vascular disease
- Hepatic disease
- Those patients who are on monoamine-oxidase (mao) inhibitor therapy
PRECAUTIONS
Caution should be observed in hypertension, peripheral vascular disease and after recent cardiovascular attacks.
This drug may make you dizzy or drowsy. Do not drive, use machinery, or do any activity that requires alertness until you are sure you can perform such activities safely. Avoid alcoholic beverages. Remember that alcohol may be a cause of headaches.
DRUG INTERACTIONS
This drug should not be used with the following medications because very serious interactions may occur: sodium oxybate.
Avoid MAO inhibitors (isocarboxazid, linezolid, methylene blue, moclobemide, phenelzine, procarbazine, rasagiline, selegiline, or tranylcypromine) within 2 weeks before, during, and after treatment with this medication. In some cases a serious (possibly fatal) drug interaction may occur.
Consider drugs that cause drowsiness such as: certain antihistamines (e.g., diphenhydramine), anti-seizure drugs (e.g., carbamazepine, phenytoin), medicine for sleep or anxiety (e.g., alprazolam, diazepam, zolpidem), muscle relaxants (e.g., cyclobenzaprine), narcotic pain relievers (e.g., codeine), psychiatric medicines (e.g., chlorpromazine, risperidone, trazodone).
This medication may interfere with certain laboratory tests (including certain urine glucose tests, urine catecholamine levels, urine 5-HIAA levels), possibly causing false test results.
PREGNANCY
FDA has not assigned a pregnancy category for Nodolor™ Capsules. Acetaminophen is routinely used for short-term pain relief and fever in all stages of pregnancy.
Acetaminophen crosses the placenta and is believed to be safe in pregnancy when used intermittently for short durations. No controlled studies have been done to establish the safety of dichloralphenazone or isometheptene or of their combination with acetaminophen. Nodolor™ Capsules should only be given during pregnancy when need has been clearly established.
NURSING MOTHERS
Acetaminophen is excreted into human milk in small concentrations. Metabolites of dichloralphenazone are excreted into human milk. There are no data on the excretion of isometheptene into human milk. Problems in humans have not been documented.
PEDIATRIC USE
Studies with this medicine have been done only in adult patients, and there is no specific information about its use in children.
GERIATRIC
There is no specific published information comparing use of this combination medicine in the elderly with use in other age groups. Geriatric patients are more likely to have peripheral vascular disease, and are therefore more likely to be adversely affected by peripheral vasoconstriction, than are younger adults. However, isometheptene may be safer for elderly patients than the ergot derivatives used to abort acute vascular headaches. Also, elderly patients are more likely to have age related renal function impairment, which may require caution in patients receiving acetaminophen and isometheptene.
ADVERSE REACTIONS
There is a potential for allergic reactions (e.g., swelling of the face, mouth, and throat, difficulty breathing, itching, or rash). A very serious allergic reaction, called anaphylaxis, to this drug is unlikely, but if it occurs, discontinue the drug and seek immediate medical attention.
Transient dizziness, drowsiness, and nausea may occur. This can usually be eliminated by reducing the dose. If any of these effects persist or worsen, notify your doctor or pharmacist promptly.
This is not a complete list of possible side effects. If you notice other effects not listed above, contact your doctor or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-877-622-6836.
To report SUSPECTED ADVERSE REACTIONS, contact Macoven Pharmaceuticals at 1-800-793-2145.
OVERDOSAGE
If overdose is suspected, contact your local poison control center or call 1-800-222-1222. Symptoms of overdose may include: restlessness, severe drowsiness/dizziness, low body temperature, fast/slow breathing, fast/slow/irregular heartbeat, unresponsiveness.
DOSAGE AND ADMINISTRATION
Do not exceed the recommended dosage because severe liver damage may occur.
Tell your doctor about all usage of pain relievers.
HOW SUPPLIED
Red capsules imprinted with 617 in bottles of 100ct, NDC 44183-442-01.
Store at 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F) [See USP Controlled Room Temperature].
Dispense in a tight, light-resistant container (USP/NF) with a child-resistant closure.
Keep out of reach of children.
All prescription substitutions using this product shall be pursuant to state statutes as applicable. This is not an Orange Book product.
| NODOLOR
isometheptene mucate, dichloralphenazone, and acetaminophen capsule |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Labeler - Macoven Pharmaceuticals (832591965) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| ECI Pharmaceuticals LLC | 962476029 | MANUFACTURE(44183-442) | |
