Label: CVS ADULT ENEMA- docusate sodium w/benzocaine liquid
- NDC Code(s): 17433-9991-1, 17433-9991-5
- Packager: Summit Pharmaceuticals
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated August 20, 2021
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
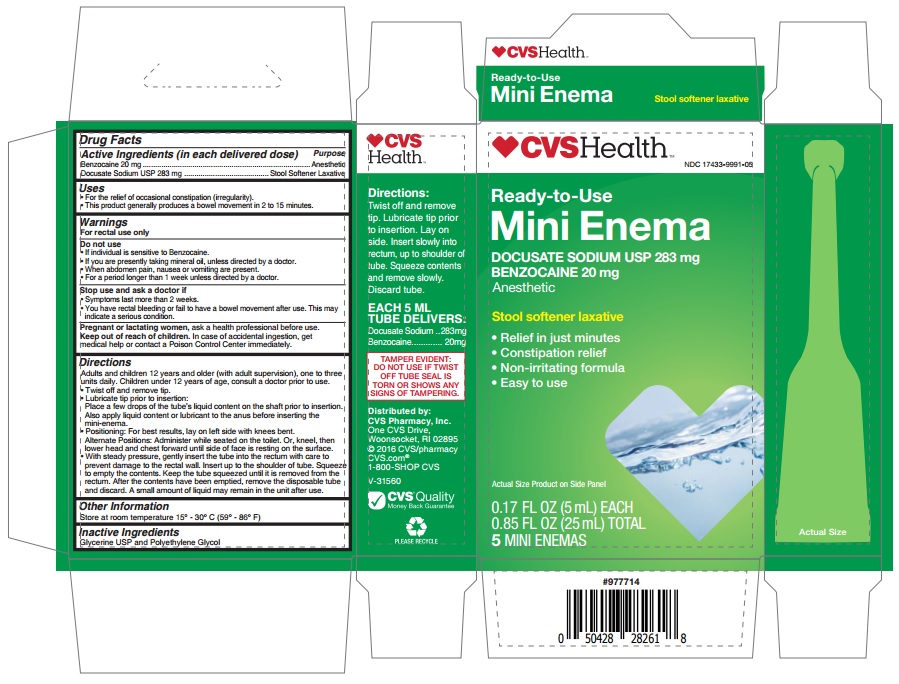
- Drug Facts
- Uses
-
Warnings
For rectal use only
Do not use
- If individual is sensitive to Benzocaine.
- If you are presently taking mineral oil, unless directed by a doctor.
- When abdomen pain, nausea or vomiting are present.
- For a period longer than 1 week unless directed by a doctor.
-
Directions
Adults and children 12 years and older (with adult supervision), one to three units daily. Children under 12 years of age, consult a doctor prior to use.
- Twist off and remove tip.
- Lubricate tip prior to insertion: Place a few drops of the tube's liquid content on the shaft prior to insertion. Also apply liquid content or lubricant to the anus before inserting the mini-enema.
- Positioning: For best results, lay on left side with knees bent. Alternate Positions: Administer while seated on the toilet. Or, kneel, then lower head and chest forward until side of face is resting on the surface.
- With steady pressure, gently insert the tube into the rectum with care to prevent damage to the rectal wall. Insert up to the shoulder of tube. Squeeze to empty the contents. Keep the tube squeezed until it is removed from the rectum. After the contents have been emptied, remove the disposable tube and discard. A small amount of liquid may remain in the unit after use.
- Other Information
- Inactive Ingredients
- QUESTIONS
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
CVS ADULT ENEMA
docusate sodium w/benzocaine liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:17433-9991 Route of Administration RECTAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DOCUSATE SODIUM (UNII: F05Q2T2JA0) (DOCUSATE - UNII:M7P27195AG) DOCUSATE SODIUM 283 mg in 4.6 g BENZOCAINE (UNII: U3RSY48JW5) (BENZOCAINE - UNII:U3RSY48JW5) BENZOCAINE 20 mg in 4.6 g Inactive Ingredients Ingredient Name Strength GLYCERIN (UNII: PDC6A3C0OX) POLYETHYLENE GLYCOL 300 (UNII: 5655G9Y8AQ) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:17433-9991-5 5 in 1 BOX 09/01/2014 06/30/2024 1 NDC:17433-9991-1 4.6 g in 1 TUBE; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part334 09/01/2014 06/30/2024 Labeler - Summit Pharmaceuticals (010717819)