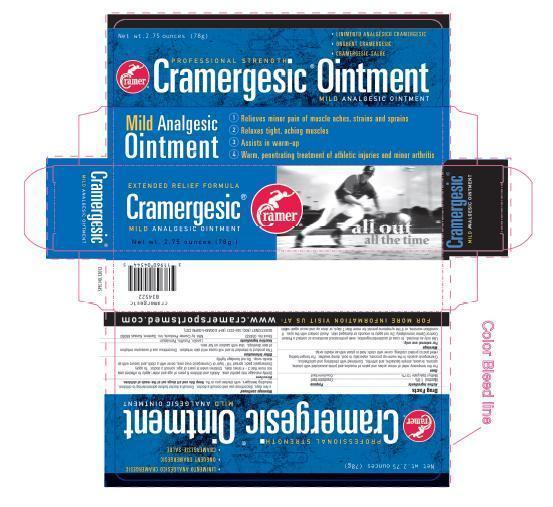
Label: CRAMERGESIC- menthol, methyl salicylate ointment
-
Contains inactivated NDC Code(s)
NDC Code(s): 11960-0045-1, 11960-0045-2, 11960-0045-3, 11960-0045-4 - Packager: Cramer Products Inc
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated April 19, 2013
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
-
Uses
For the temporary relief of minor aches and pains of muscles and joints associated with strain, sprains, bruises, simple backache, and arthritis. Combined with stretching and calisthenics, Cramergesic assists in the warm-up process, especially in cool, damp weather. For longer-lasting relief and to protect clothing, cover with cloth; hold in place with an elastic wrap.
-
Warnings
For external use only.
Use only as directed. In case of accidental ingestion, seek professional assistance or contact a Poison Control Center immediately. Do not apply to wounds or damaged skin. Avoid contact with the eyes. If condition worsens, or if the symptoms persist for more than 7 days, or clear up and occur again within a few days, discontinue use and consult a doctor. Consult a doctor before administering to children, including teenagers, with chicken pox or flu.
-
DOSAGE & ADMINISTRATION
Directions
Gently massage into painful area. Adults and children 6 years of age and older: apply to affected area not more than 3 - 4 times daily. Children under 6 years of age: consult a doctor. To apply Cramergesic pack: spread 1/8 " layer of Cramergesic over area, cover with a cloth, and secure with an elastic wrap. Do not bandage tightly.
Other information
This product is intended to and will cause mild skin irritation. Discontinue use if excessive irritation of skin develops. Use with caution on fair skin.
- INACTIVE INGREDIENT
- QUESTIONS
- PRINCIPAL DISPLAY PANEL
- PURPOSE
- KEEP OUT OF REACH OF CHILDREN
-
INGREDIENTS AND APPEARANCE
CRAMERGESIC
menthol, methyl salicylate ointmentProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:11960-0045 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength menthol (UNII: L7T10EIP3A) (menthol - UNII:L7T10EIP3A) menthol 0.019 g in 1 g methyl salicylate (UNII: LAV5U5022Y) (salicylic acid - UNII:O414PZ4LPZ) methyl salicylate 0.137 g in 1 g Inactive Ingredients Ingredient Name Strength lanolin (UNII: 7EV65EAW6H) paraffin (UNII: I9O0E3H2ZE) petrolatum (UNII: 4T6H12BN9U) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:11960-0045-2 1 in 1 CARTON 1 NDC:11960-0045-1 78 g in 1 TUBE 2 NDC:11960-0045-3 454 g in 1 JAR 3 NDC:11960-0045-4 2268 g in 1 JAR Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part348 07/15/1992 Labeler - Cramer Products Inc (007138183) Establishment Name Address ID/FEI Business Operations Cramer Products Inc 007138183 manufacture(11960-0045) , pack(11960-0045)