Label: BICILLIN L-A- penicillin g benzathine injection, suspension
-
NDC Code(s):
60793-700-01,
60793-700-10,
60793-701-02,
60793-701-10, view more60793-702-04, 60793-702-10
- Packager: Pfizer Laboratories Div Pfizer Inc
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated October 13, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
-
BOXED WARNING
(What is this?)
WARNING
NOT FOR INTRAVENOUS USE. DO NOT INJECT INTRAVENOUSLY OR ADMIX WITH OTHER INTRAVENOUS SOLUTIONS. THERE HAVE BEEN REPORTS OF INADVERTENT INTRAVENOUS ADMINISTRATION OF PENICILLIN G BENZATHINE WHICH HAS BEEN ASSOCIATED WITH CARDIORESPIRATORY ARREST AND DEATH. Prior to administration of this drug, carefully read the WARNINGS, ADVERSE REACTIONS, and DOSAGE AND ADMINISTRATION sections of the labeling.
- SPL UNCLASSIFIED SECTION
-
Description
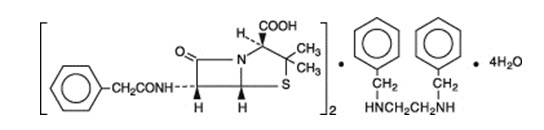
Bicillin L-A (penicillin G benzathine injectable suspension) is available for deep intramuscular injection. Penicillin G benzathine is prepared by the reaction of dibenzylethylene diamine with two molecules of penicillin G. It is chemically designated as (2S, 5R, 6R)-3,3-Dimethyl-7-oxo-6-(2-phenylacetamido)-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid compound with N,N' -dibenzylethylenediamine (2:1), tetrahydrate. It occurs as a white, crystalline powder and is very slightly soluble in water and sparingly soluble in alcohol. Its chemical structure is as follows:
Bicillin L-A contains penicillin G benzathine in aqueous suspension with sodium citrate buffer and, as w/v, approximately 0.65% sodium citrate, 0.59% povidone, 0.54% carboxymethylcellulose sodium, 0.53% lecithin, 0.12% methylparaben, and 0.013% propylparaben.
Bicillin L-A contains approximately 0.11 mEq of sodium per 600,000 units of penicillin G (approximately 2.59 mg of sodium per 600,000 units of penicillin G).
Bicillin L-A suspension in the disposable-syringe formulation is viscous and opaque. It is available in a 1 mL, 2 mL, and 4 mL sizes containing the equivalent of 600,000 (actual volume of 1.17 mL contains 620,100), 1,200,000 (actual volume of 2.34 mL contains 1,240,200), and 2,400,000 (actual volume of 4.67 mL contains 2,475,100) units respectively of penicillin G as the benzathine salt. Read CONTRAINDICATIONS, WARNINGS, PRECAUTIONS, and DOSAGE AND ADMINISTRATION sections prior to use.
-
CLINICAL PHARMACOLOGY
General
Penicillin G benzathine has an extremely low solubility and, thus, the drug is slowly released from intramuscular injection sites. The drug is hydrolyzed to penicillin G. This combination of hydrolysis and slow absorption results in blood serum levels much lower but much more prolonged than other parenteral penicillins.
Intramuscular administration of 300,000 units of penicillin G benzathine in adults results in blood levels of 0.03 to 0.05 units per mL, which are maintained for 4 to 5 days. Similar blood levels may persist for 10 days following administration of 600,000 units and for 14 days following administration of 1,200,000 units. Blood concentrations of 0.003 units per mL may still be detectable 4 weeks following administration of 1,200,000 units.
Approximately 60% of penicillin G is bound to serum protein. The drug is distributed throughout the body tissues in widely varying amounts. Highest levels are found in the kidneys with lesser amounts in the liver, skin, and intestines. Penicillin G penetrates into all other tissues and the spinal fluid to a lesser degree. With normal kidney function, the drug is excreted rapidly by tubular excretion. In neonates and young infants and in individuals with impaired kidney function, excretion is considerably delayed.
Microbiology
Mechanism of Action
Penicillin G exerts a bactericidal action against penicillin-susceptible microorganisms during the stage of active multiplication. It acts through the inhibition of biosynthesis of cell-wall peptidoglycan, rendering the cell wall osmotically unstable.
Resistance
Penicillin is not active against penicillinase-producing bacteria or against organisms resistant to beta-lactams because of alterations in the penicillin-binding proteins. Resistance to penicillin G has not been reported in Streptococcus pyogenes.
Antimicrobial Activity
Penicillin has been shown to be active against most isolates of the following bacteria, both in vitro and in clinical infections as described in the INDICATIONS AND USAGE section.
Gram-positive bacteria
Beta-hemolytic streptococci (groups A, B, C, G, H, L and M).
Other microorganisms
Treponema pallidum
Treponema carateumSusceptibility Testing
For specific information regarding susceptibility test interpretive criteria and associated test methods and quality control standards recognized by FDA for this drug, please see: https://www.fda.gov/STIC.
-
INDICATIONS AND USAGE
To reduce the development of drug-resistant bacteria and maintain the effectiveness of Bicillin L-A and other antibacterial drugs, Bicillin L-A should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
Intramuscular penicillin G benzathine is indicated in the treatment of infections due to penicillin-G-sensitive microorganisms that are susceptible to the low and very prolonged serum levels common to this particular dosage form. Therapy should be guided by bacteriological studies (including sensitivity tests) and by clinical response.
The following infections will usually respond to adequate dosage of intramuscular penicillin G benzathine:
Mild-to-moderate infections of the upper-respiratory tract due to susceptible streptococci.
Venereal infections—Syphilis, yaws, bejel, and pinta.
Medical Conditions in which Penicillin G Benzathine Therapy is indicated as Prophylaxis:
Rheumatic fever and/or chorea—Prophylaxis with penicillin G benzathine has proven effective in preventing recurrence of these conditions. It has also been used as follow-up prophylactic therapy for rheumatic heart disease and acute glomerulonephritis.
- CONTRAINDICATIONS
-
WARNINGS
WARNING
NOT FOR INTRAVENOUS USE. DO NOT INJECT INTRAVENOUSLY OR ADMIX WITH OTHER INTRAVENOUS SOLUTIONS. THERE HAVE BEEN REPORTS OF INADVERTENT INTRAVENOUS ADMINISTRATION OF PENICILLIN G BENZATHINE WHICH HAS BEEN ASSOCIATED WITH CARDIORESPIRATORY ARREST AND DEATH. Prior to administration of this drug, carefully read the WARNINGS, ADVERSE REACTIONS, and DOSAGE AND ADMINISTRATION sections of the labeling.
Anaphylaxis
SERIOUS AND OCCASIONALLY FATAL HYPERSENSITIVITY (ANAPHYLACTIC) REACTIONS HAVE BEEN REPORTED IN PATIENTS ON PENICILLIN THERAPY. THESE REACTIONS ARE MORE LIKELY TO OCCUR IN INDIVIDUALS WITH A HISTORY OF PENICILLIN HYPERSENSITIVITY AND/OR A HISTORY OF SENSITIVITY TO MULTIPLE ALLERGENS. THERE HAVE BEEN REPORTS OF INDIVIDUALS WITH A HISTORY OF PENICILLIN HYPERSENSITIVITY WHO HAVE EXPERIENCED SEVERE REACTIONS WHEN TREATED WITH CEPHALOSPORINS. BEFORE INITIATING THERAPY WITH BICILLIN L-A, CAREFUL INQUIRY SHOULD BE MADE CONCERNING PREVIOUS HYPERSENSITIVITY REACTIONS TO PENICILLINS, CEPHALOSPORINS, OR OTHER ALLERGENS. IF AN ALLERGIC REACTION OCCURS, BICILLIN L-A SHOULD BE DISCONTINUED AND APPROPRIATE THERAPY INSTITUTED. SERIOUS ANAPHYLACTIC REACTIONS REQUIRE IMMEDIATE EMERGENCY TREATMENT WITH EPINEPHRINE. OXYGEN, INTRAVENOUS STEROIDS AND AIRWAY MANAGEMENT, INCLUDING INTUBATION, SHOULD ALSO BE ADMINISTERED AS INDICATED.
Severe cutaneous adverse reactions
Severe cutaneous adverse reactions (SCAR), such as Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), drug reaction with eosinophilia and systemic symptoms (DRESS), and acute generalized exanthematous pustulosis (AGEP) have been reported in patients taking penicillin G (the active moiety in Bicillin L-A). When SCAR is suspected, Bicillin L-A should be discontinued immediately and an alternative treatment should be considered.
Clostridioides difficile associated diarrhea
Clostridioides difficile associated-diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including Bicillin L-A, and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibacterial use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibiotic use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibiotic treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
Method of Administration
Do not inject into or near an artery or nerve. See administration instructions below.
Injection into or near a nerve may result in permanent neurological damage.
Inadvertent intravascular administration, including inadvertent direct intra-arterial injection or injection immediately adjacent to arteries, of Bicillin L-A and other penicillin preparations has resulted in severe neurovascular damage, including transverse myelitis with permanent paralysis, gangrene requiring amputation of digits and more proximal portions of extremities, and necrosis and sloughing at and surrounding the injection site consistent with the diagnosis of Nicolau syndrome. Such severe effects have been reported following injections into the buttock, thigh, and deltoid areas. Other serious complications of suspected intravascular administration which have been reported include immediate pallor, mottling, or cyanosis of the extremity both distal and proximal to the injection site, followed by bleb formation; severe edema requiring anterior and/or posterior compartment fasciotomy in the lower extremity. The above-described severe effects and complications have most often occurred in infants and small children. Prompt consultation with an appropriate specialist is indicated if any evidence of compromise of the blood supply occurs at, proximal to, or distal to the site of injection.1–9 (See PRECAUTIONS, and DOSAGE AND ADMINISTRATION sections.)
FOR DEEP INTRAMUSCULAR INJECTION ONLY. There have been reports of inadvertent intravenous administration of penicillin G benzathine which has been associated with cardiorespiratory arrest and death. Therefore, do not inject intravenously or admix with other intravenous solutions. (See DOSAGE AND ADMINISTRATION section.)
Administer by DEEP INTRAMUSCULAR INJECTION ONLY in the upper, outer quadrant of the buttock (dorsogluteal) or the ventrogluteal site. Quadriceps femoris fibrosis and atrophy have been reported following repeated intramuscular injections of penicillin preparations into the anterolateral thigh. Because of these adverse effects and the vascularity of this region, administration in the anterolateral thigh is not recommended.
-
PRECAUTIONS
General
Prescribing Bicillin L-A in the absence of a proven or strongly suspected bacterial infection or a prophylactic indication is unlikely to provide benefit to the patient and increases the risk of a development of drug-resistant bacteria.
Penicillin should be used with caution in individuals with histories of significant allergies and/or asthma.
Care should be taken to avoid intravenous or intra-arterial administration, or injection into or near major peripheral nerves or blood vessels, since such injection may produce neurovascular damage. (See WARNINGS, and DOSAGE AND ADMINISTRATION sections.)
Prolonged use of antibiotics may promote the overgrowth of nonsusceptible organisms, including fungi. Should superinfection occur, appropriate measures should be taken.
Bicillin L-A contains approximately 0.11 mEq of sodium per 600,000 units of penicillin G (approximately 2.59 mg of sodium per 600,000 units of penicillin G).
Information for Patients
Diarrhea is a common problem caused by antibiotics which usually ends when the antibiotic is discontinued. Sometimes after starting treatment with antibiotics, patients can develop watery and bloody stools (with or without stomach cramps and fever) even as late as two or more months after having taken the last dose of the antibiotic. If this occurs, patients should contact their physician as soon as possible.
Patients should be counseled that antibacterial drugs including Bicillin L-A should only be used to treat bacterial infections. They do not treat viral infections (e.g., the common cold). When Bicillin L-A is prescribed to treat a bacterial infection, patients should be told that although it is common to feel better early in the course of therapy, the medication should be taken exactly as directed. Skipping doses or not completing the full course of therapy may (1) decrease the effectiveness of the immediate treatment and (2) increase the likelihood that bacteria will develop resistance and will not be treatable by Bicillin L-A or other antibacterial drugs in the future.
Laboratory Tests
In streptococcal infections, therapy must be sufficient to eliminate the organism; otherwise, the sequelae of streptococcal disease may occur. Cultures should be taken following completion of treatment to determine whether streptococci have been eradicated.
Drug Interactions
Tetracycline, a bacteriostatic antibiotic, may antagonize the bactericidal effect of penicillin, and concurrent use of these drugs should be avoided.
Concurrent administration of penicillin and probenecid increases and prolongs serum penicillin levels by decreasing the apparent volume of distribution and slowing the rate of excretion by competitively inhibiting renal tubular secretion of penicillin.
Pregnancy
Teratogenic effects
Pregnancy Reproduction studies performed in the mouse, rat, and rabbit have revealed no evidence of impaired fertility or harm to the fetus due to penicillin G. Human experience with the penicillins during pregnancy has not shown any positive evidence of adverse effects on the fetus. There are, however, no adequate and well-controlled studies in pregnant women showing conclusively that harmful effects of these drugs on the fetus can be excluded. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
Nursing Mothers
Soluble penicillin G (the hydrolysate of penicillin G benzathine) is excreted in breast milk. Caution should be exercised when penicillin G benzathine is administered to a nursing woman.
Carcinogenesis, Mutagenesis, Impairment of Fertility
No long-term animal studies have been conducted with this drug.
Geriatric Use
Clinical studies of penicillin G benzathine did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy. This drug is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. (See CLINICAL PHARMACOLOGY.) Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.
-
ADVERSE REACTIONS
As with other penicillins, untoward reactions of the sensitivity phenomena are likely to occur, particularly in individuals who have previously demonstrated hypersensitivity to penicillins or in those with a history of allergy, asthma, hay fever, or urticaria.
As with other treatments for syphilis, the Jarisch-Herxheimer reaction has been reported.
The following adverse reactions have been reported with Bicillin L-A during post-marketing experience:
Skin and Appendages: Stevens-Johnson syndrome (SJS) and drug reaction with eosinophilia and systemic symptoms (DRESS). (See WARNINGS.)
Immune System Disorders: Acute myocardial ischemia with or without myocardial infarction may occur as part of an allergic reaction (Kounis syndrome).
The following have been reported with parenteral penicillin G (the active moiety in Bicillin L-A):
General: Hypersensitivity reactions including the following: skin eruptions (maculopapular to exfoliative dermatitis), urticaria, laryngeal edema, fever, eosinophilia; other serum sickness-like reactions (including chills, fever, edema, arthralgia, and prostration); and anaphylaxis including shock and death: severe cutaneous adverse reactions (SCAR), such as toxic epidermal necrolysis (TEN) and acute generalized exanthematous pustulosis (AGEP). (See WARNINGS.) Note: Urticaria, other skin rashes, and serum sickness-like reactions may be controlled with antihistamines and, if necessary, systemic corticosteroids. Whenever such reactions occur, penicillin G should be discontinued unless, in the opinion of the physician, the condition being treated is life-threatening and amenable only to therapy with penicillin G. Serious anaphylactic reactions require immediate emergency treatment with epinephrine. Oxygen, intravenous steroids, and airway management, including intubation, should also be administered as indicated.
Gastrointestinal: Pseudomembranous colitis. Onset of pseudomembranous colitis symptoms may occur during or after antibacterial treatment. (See WARNINGS section.)
Hematologic: Hemolytic anemia, leukopenia, thrombocytopenia.
Neurologic: Neuropathy.
Urogenital: Nephropathy.
The following adverse events have been temporally associated with parenteral administration of penicillin G benzathine (a component of Bicillin L-A):
Body as a Whole: Hypersensitivity reactions including allergic vasculitis, pruritus, fatigue, asthenia, and pain; aggravation of existing disorder; headache, Nicolau syndrome.
Cardiovascular: Cardiac arrest; hypotension; tachycardia; palpitations; pulmonary hypertension; pulmonary embolism; vasodilation; vasovagal reaction; cerebrovascular accident; syncope.
Gastrointestinal: Nausea, vomiting; blood in stool; intestinal necrosis.
Hemic and Lymphatic: Lymphadenopathy.
Injection Site: Injection site reactions including pain, inflammation, lump, abscess, necrosis, edema, hemorrhage, cellulitis, hypersensitivity, atrophy, ecchymosis, and skin ulcer. Neurovascular reactions including warmth, vasospasm, pallor, mottling, gangrene, numbness of the extremities, cyanosis of the extremities, and neurovascular damage.
Metabolic: Elevated BUN, creatinine, and SGOT.
Musculoskeletal: Joint disorder; periostitis; exacerbation of arthritis; myoglobinuria; rhabdomyolysis.
Nervous System: Nervousness; tremors; dizziness; somnolence; confusion; anxiety; euphoria; transverse myelitis; seizures; coma. A syndrome manifested by a variety of CNS symptoms such as severe agitation with confusion, visual and auditory hallucinations, and a fear of impending death (Hoigne's syndrome), has been reported after administration of penicillin G procaine and, less commonly, after injection of the combination of penicillin G benzathine and penicillin G procaine. Other symptoms associated with this syndrome, such as psychosis, seizures, dizziness, tinnitus, cyanosis, palpitations, tachycardia, and/or abnormal perception in taste, also may occur.
Respiratory: Hypoxia; apnea; dyspnea.
Skin: Diaphoresis.
Special Senses: Blurred vision; blindness.
Urogenital: Neurogenic bladder; hematuria; proteinuria; renal failure; impotence; priapism.
- OVERDOSAGE
-
DOSAGE AND ADMINISTRATION
Streptococcal (Group A) Upper Respiratory Infections (for example, pharyngitis)
Adults—a single injection of 1,200,000 units; older pediatric patients—a single injection of 900,000 units; infants and pediatric patients under 60 lbs.—300,000 to 600,000 units.
Syphilis
Primary, secondary, and latent—2,400,000 units (1 dose). Late (tertiary and neurosyphilis)—2,400,000 units at 7-day intervals for three doses.
Congenital—under 2 years of age: 50,000 units/kg/body weight; ages 2 to 12 years: adjust dosage based on adult dosage schedule.
Yaws, Bejel, and Pinta—1,200,000 units (1 injection).
Prophylaxis—for rheumatic fever and glomerulonephritis.
Following an acute attack, penicillin G benzathine (parenteral) may be given in doses of 1,200,000 units once a month or 600,000 units every 2 weeks.
METHOD OF ADMINISTRATION
BICILLIN L-A IS INTENDED FOR INTRAMUSCULAR INJECTION ONLY. DO NOT INJECT INTO OR NEAR AN ARTERY OR NERVE, OR INTRAVENOUSLY OR ADMIX WITH OTHER INTRAVENOUS SOLUTIONS. (SEE WARNINGS SECTION.)
Administer by DEEP INTRAMUSCULAR INJECTION in the upper, outer quadrant of the buttock (dorsogluteal) or the ventrogluteal site. In neonates, infants and small children, the midlateral aspect of the thigh may be preferable. Administration in the anterolateral thigh is not recommended due to the adverse effects observed (see WARNINGS section), and vascularity of this region. When doses are repeated, vary the injection site.
Because of the high concentration of suspended material in this product, the needle may be blocked if the injection is not made at a slow, steady rate.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever solution and container permit.
-
HOW SUPPLIED
Bicillin L-A (penicillin G benzathine injectable suspension) is supplied in packages of 10 disposable syringes as follows:
1 mL size, containing 600,000 units per syringe, (21 gauge, thin-wall 1-inch needle for pediatric use), with 0.11 mEq of sodium per 600,000 units of penicillin G (2.59 mg of sodium per 600,000 units of penicillin G), NDC 60793-700-10.
2 mL size, containing 1,200,000 units per syringe, (21 gauge, thin-wall 1-1/2-inch needle), with 0.22 mEq of sodium per 1,200,000 units of penicillin G (5.17 mg of sodium per 1,200,000 units of penicillin G), NDC 60793-701-10.
4 mL size, containing 2,400,000 units per syringe (18 gauge, × 1–1/2-inch needle), with 0.45 mEq of sodium per 2,400,000 units of penicillin G (10.32 mg of sodium per 2,400,000 units of penicillin G), NDC 60793-702-10.
-
REFERENCES
- 1.
- SHAW, E.: Transverse myelitis from injection of penicillin. Am. J. Dis. Child., 111:548, 1966.
- 2.
- KNOWLES, J.: Accidental intra-arterial injection of penicillin. Am. J. Dis. Child., 111:552, 1966.
- 3.
- DARBY, C. et al: Ischemia following an intragluteal injection of benzathine-procaine penicillin G mixture in a one-year-old boy. Clin. Pediatrics, 12:485, 1973.
- 4.
- BROWN, L. & NELSON, A.: Postinfectious intravascular thrombosis with gangrene. Arch. Surg., 94:652, 1967.
- 5.
- BORENSTINE, J.: Transverse myelitis and penicillin (Correspondence). Am. J. Dis. Child., 112:166, 1966.
- 6.
- ATKINSON, J.: Transverse myelopathy secondary to penicillin injection. J. Pediatrics, 75:867, 1969.
- 7.
- TALBERT, J. et al: Gangrene of the foot following intramuscular injection in the lateral thigh: A case report with recommendations for prevention. J. Pediatrics, 70:110, 1967.
- 8.
- FISHER, T.: Medicolegal affairs. Canad. Med. Assoc. J., 112:395, 1975.
- 9.
- SCHANZER, H. et al: Accidental intra-arterial injection of penicillin G. JAMA, 242:1289, 1979.
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 1 mL Syringe Label
-
PRINCIPAL DISPLAY PANEL - 1 mL Syringe Package
NDC 60793-700-10
Contains 10 of NDC 60793-700-01Ten Syringes (1 mL size)
Bicillin® L-A
(penicillin G benzathine injectable suspension)600,000 units per 1 mL
FOR PEDIATRIC USE
FOR DEEP INTRAMUSCULAR INJECTION ONLY
WARNING: FATAL IF GIVEN BY OTHER ROUTES
BEFORE INJECTING, SEE PACKAGE INSERT FOR ADMINISTRATION INSTRUCTIONS.Pfizer Injectables
Rx only
- PRINCIPAL DISPLAY PANEL - 2 mL Syringe Label
-
PRINCIPAL DISPLAY PANEL - 2 mL Syringe Package
NDC 60793-701-10
Contains 10 of NDC 60793-701-02Ten Syringes (2 mL size)
Bicillin® L-A
(penicillin G benzathine injectable suspension)1,200,000 units per 2 mL
FOR DEEP INTRAMUSCULAR INJECTION ONLY
WARNING: FATAL IF GIVEN BY OTHER ROUTES
BEFORE INJECTING, SEE PACKAGE INSERT FOR ADMINISTRATION INSTRUCTIONS.Pfizer Injectables
Rx only
- PRINCIPAL DISPLAY PANEL - 4 mL Syringe Label
-
PRINCIPAL DISPLAY PANEL - 4 mL Syringe Package
NDC 60793-702-10
Contains 10 of NDC 60793-702-04Ten Syringes (4 mL size)
Bicillin® L-A
(penicillin G benzathine injectable suspension)2,400,000 units per 4 mL
disposable syringe
FOR DEEP INTRAMUSCULAR INJECTION ONLY
WARNING: FATAL IF GIVEN BY OTHER ROUTES
BEFORE INJECTING, SEE PACKAGE INSERT FOR ADMINISTRATION INSTRUCTIONS.Pfizer Injectables
Rx only
-
INGREDIENTS AND APPEARANCE
BICILLIN L-A
penicillin g benzathine injection, suspensionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:60793-700 Route of Administration INTRAMUSCULAR Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PENICILLIN G BENZATHINE (UNII: RIT82F58GK) (PENICILLIN G - UNII:Q42T66VG0C) PENICILLIN G 600000 [iU] in 1 mL Inactive Ingredients Ingredient Name Strength LECITHIN, SOYBEAN (UNII: 1DI56QDM62) CARBOXYMETHYLCELLULOSE SODIUM, UNSPECIFIED (UNII: K679OBS311) POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) METHYLPARABEN (UNII: A2I8C7HI9T) PROPYLPARABEN (UNII: Z8IX2SC1OH) SODIUM CITRATE, UNSPECIFIED FORM (UNII: 1Q73Q2JULR) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:60793-700-10 10 in 1 PACKAGE 06/27/1952 1 NDC:60793-700-01 1 mL in 1 SYRINGE; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA050141 06/27/1952 BICILLIN L-A
penicillin g benzathine injection, suspensionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:60793-701 Route of Administration INTRAMUSCULAR Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PENICILLIN G BENZATHINE (UNII: RIT82F58GK) (PENICILLIN G - UNII:Q42T66VG0C) PENICILLIN G 1200000 [iU] in 2 mL Inactive Ingredients Ingredient Name Strength LECITHIN, SOYBEAN (UNII: 1DI56QDM62) CARBOXYMETHYLCELLULOSE SODIUM, UNSPECIFIED (UNII: K679OBS311) POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) METHYLPARABEN (UNII: A2I8C7HI9T) PROPYLPARABEN (UNII: Z8IX2SC1OH) SODIUM CITRATE, UNSPECIFIED FORM (UNII: 1Q73Q2JULR) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:60793-701-10 10 in 1 PACKAGE 06/27/1952 1 NDC:60793-701-02 2 mL in 1 SYRINGE; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA050141 06/27/1952 BICILLIN L-A
penicillin g benzathine injection, suspensionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:60793-702 Route of Administration INTRAMUSCULAR Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PENICILLIN G BENZATHINE (UNII: RIT82F58GK) (PENICILLIN G - UNII:Q42T66VG0C) PENICILLIN G 2400000 [iU] in 4 mL Inactive Ingredients Ingredient Name Strength LECITHIN, SOYBEAN (UNII: 1DI56QDM62) CARBOXYMETHYLCELLULOSE SODIUM, UNSPECIFIED (UNII: K679OBS311) POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) METHYLPARABEN (UNII: A2I8C7HI9T) PROPYLPARABEN (UNII: Z8IX2SC1OH) SODIUM CITRATE, UNSPECIFIED FORM (UNII: 1Q73Q2JULR) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:60793-702-10 10 in 1 PACKAGE 06/27/1952 1 NDC:60793-702-04 4 mL in 1 SYRINGE; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA050141 06/27/1952 Labeler - Pfizer Laboratories Div Pfizer Inc (134489525) Establishment Name Address ID/FEI Business Operations King Pharmaceuticals LLC 962691478 ANALYSIS(60793-700, 60793-701, 60793-702) , MANUFACTURE(60793-700, 60793-701, 60793-702) , PACK(60793-700, 60793-701, 60793-702) , LABEL(60793-700, 60793-701, 60793-702)