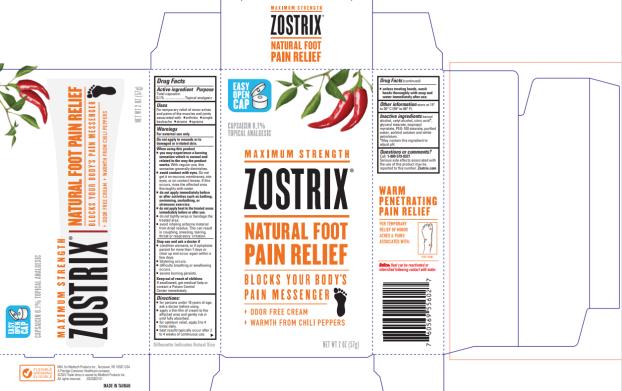
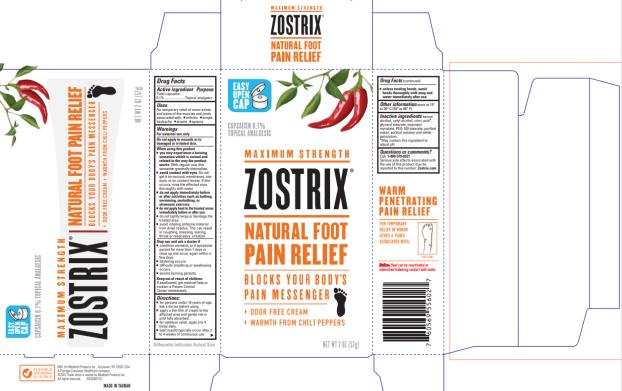
Label: ZOSTRIX HIGH POTENCY FOOT PAIN RELIEF- capsaicin cream
- NDC Code(s): 61787-556-02
- Packager: MEDTECH PRODUCTS INC
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated March 3, 2022
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredient
- Purpose
- Uses
-
Warnings
For external use only.
Do not apply to wounds or to damaged or irritated skin.
When using this product
-
you may experience a burning sensation which is normal and related to the way the product works. With regular use, this sensation generally diminishes.
-
avoid contact with eyes. Do not get it on mucous membranes, into eyes, or on contact lenses. If this occurs, rinse the affected area thoroughly with water.
-
do not apply immediately before or after activities such as bathing, swimming, sun bathing, or strenuous exercise.
-
do not apply heat to the treated areas immediately before or after use.
- do not tightly wrap or bandage the treated area.
- avoid inhaling airborne material from dried residue. This can result in coughing, sneezing, tearing, throat or respiratory irritation.
-
you may experience a burning sensation which is normal and related to the way the product works. With regular use, this sensation generally diminishes.
-
Directions
- for persons under 18 years of age, ask a doctor before using.
- apply a thin film of cream to the affected area and gently rub in until fully absorbed.
- for optimum relief, apply 3 to 4 times daily
- best results typically occur after 2 to 4 weeks of continuous use.
-
wash hands thoroughly with soap and water immediately after use.
- see package insert for more information.
- for persons under 18 years of age, ask a doctor before using.
- Other information
- Inactive ingredients
- Questions or Comments?
- Package/Label Principal Display Panel
-
INGREDIENTS AND APPEARANCE
ZOSTRIX HIGH POTENCY FOOT PAIN RELIEF
capsaicin creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:61787-556 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CAPSAICIN (UNII: S07O44R1ZM) (CAPSAICIN - UNII:S07O44R1ZM) CAPSAICIN 0.75 mg in 1 g Inactive Ingredients Ingredient Name Strength ISOPROPYL MYRISTATE (UNII: 0RE8K4LNJS) WATER (UNII: 059QF0KO0R) SORBITOL (UNII: 506T60A25R) CETYL ALCOHOL (UNII: 936JST6JCN) PETROLATUM (UNII: 4T6H12BN9U) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) PEG-100 STEARATE (UNII: YD01N1999R) BENZYL ALCOHOL (UNII: LKG8494WBH) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:61787-556-02 1 in 1 CARTON 10/22/2013 1 56.6 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part348 10/22/2013 Labeler - MEDTECH PRODUCTS INC (114707784) Establishment Name Address ID/FEI Business Operations Denison Pharmaceuticals, LLC 001207208 manufacture(61787-556)