BARRIER WATERLESS SURGICAL PREP
-
povidone-iodine,
alcohol and
isopropyl alcohol solution
Aurora Pharmaceutical LLC
Disclaimer: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
----------
INDICATIONS
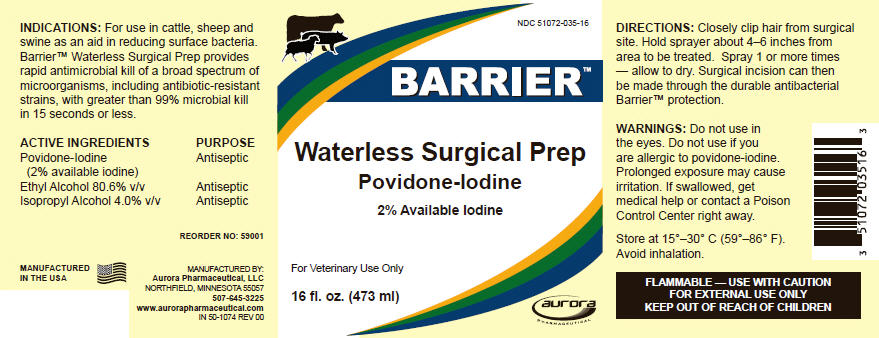
For use in cattle, sheep and swine as an aid in reducing surface bacteria. Barrier™ Waterless Surgical Prep provides rapid antimicrobial kill of a broad spectrum of microorganisms, including antibiotic-resistant strains, with greater than 99% microbial kill in 15 seconds or less.
| ACTIVE INGREDIENTS | PURPOSE |
|---|---|
| Povidone-Iodine (2% available iodine) | Antiseptic |
| Ethyl Alcohol 80.6% v/v | Antiseptic |
| Isopropyl Alcohol 4.0% v/v | Antiseptic |
DIRECTIONS
Closely clip hair from surgical site. Hold sprayer about 4–6 inches from area to be treated. Spray 1 or more times — allow to dry. Surgical incision can then be made through the durable antibacterial Barrier™ protection.
WARNINGS
Do not use in the eyes. Do not use if you are allergic to povidone-iodine. Prolonged exposure may cause irritation. If swallowed, get medical help or contact a Poison Control Center right away.
FLAMMABLE — USE WITH CAUTION
FOR EXTERNAL USE ONLY
KEEP OUT OF REACH OF CHILDREN
REORDER NO: 59001
MANUFACTURED
IN THE USA
MANUFACTURED BY:
Aurora Pharmaceutical, LLC
NORTHFIELD, MINNESOTA 55057
507-645-3225
www.aurorapharmaceutical.com
IN 50-1074 REV 00
PRINCIPAL DISPLAY PANEL - 473 ml Bottle Label
NDC 51072-035-16
BARRIER™
Waterless Surgical Prep
Povidone-Iodine
2% Available Iodine
For Veterinary Use Only
16 fl. oz. (473 ml)
aurora
PHARMACEUTICAL
| BARRIER
WATERLESS SURGICAL PREP
povidone-iodine, alcohol, isopropyl alcohol solution |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| unapproved drug other | 08/20/2010 | 11/30/2012 | |
| Labeler - Aurora Pharmaceutical LLC (832848639) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| Aurora Pharmaceutical LLC | 832848639 | manufacture | |
Revised: 01/2013 Aurora Pharmaceutical LLC