DONEPEZIL HYDROCHLORIDE - donepezil hydrochloride tablet, film coated
Macleods Pharmaceuticals Limited
----------
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use Donepezil Hydrochloride Tablets USP safely and effectively. See full prescribing information for Donepezil Hydrochloride Tablets USP.
Donepezil Hydrochloride Tablets USP Initial U.S. Approval: 1996 RECENT MAJOR CHANGESNone
INDICATIONS AND USAGEDonepezil hydrochloride USP is an acetylcholinesterase inhibitor indicated for the treatment of dementia of the Alzheimer's type. Efficacy has been demonstrated in patients with mild, moderate, and severe Alzheimer's disease (1.0).
DOSAGE AND ADMINISTRATIONDOSAGE FORMS AND STRENGTHS
CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
ADVERSE REACTIONSThe most common adverse reactions in clinical studies of donepezil hydrochloride tablets are nausea, diarrhea, insomnia, vomiting muscle cramps, fatigue and anorexia (6.1). To report SUSPECTED ADVERSE REACTIONS, contact Macleods Pharma USA, Inc. at 1-888-943-3210 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch DRUG INTERACTIONSUSE IN SPECIFIC POPULATIONS
See 17 for PATIENT COUNSELING INFORMATION. Revised: 01/2013 |
FULL PRESCRIBING INFORMATION
1 INDICATIONS & USAGE
Donepezil hydrochloride USP is indicated for the treatment of dementia of the Alzheimer's type. Efficacy has been demonstrated in patients with mild, moderate, and severe Alzheimer's disease.
2 DOSAGE & ADMINISTRATION
Donepezil hydrochloride tablets should be taken in the evening just before retiring.
2.1 Mild to Moderate Alzheimer's Disease
The dosages of donepezil hydrochloride shown to be effective in controlled clinical trials are 5 mg and 10 mg administered once per day.
The higher dose of 10 mg did not provide a statistically significantly greater clinical benefit than 5 mg. There is a suggestion, however, based upon order of group mean scores and dose trend analyses of data from these clinical trials, that a daily dose of 10 mg of donepezil hydrochloride might provide additional benefit for some patients. Accordingly, whether or not to employ a dose of 10 mg is a matter of prescriber and patient preference.
2.2Moderate to Severe Alzheimer's Disease
Donepezil hydrochloride has been shown to be effective in controlled clinical trials at a dose of 10 mg administered once daily.
2.3 Titration
The recommended starting dose of donepezil hydrochloride is 5mg once daily. Evidence from the controlled trials in mild to moderate Alzheimer's disease indicates that the 10 mg dose, with a one week titration, is likely to be associated with a higher incidence of cholinergic adverse events compared to the 5 mg dose. In open-label trials using a 6 week titration, the type and frequency of these same adverse events were similar between the 5 mg and 10 mg dose groups. Therefore, because donepezil hydrochloride steady state is achieved about 15 days after it is started and because the incidence of untoward effects may be influenced by the rate of dose escalation, a dose of 10 mg should not be administered until patients have been on a daily dose of 5 mg for 4 to 6 weeks.
3 DOSAGE FORMS & STRENGTHS
Donepezil hydrochloride is supplied as film-coated, round tablets containing either 5 mg or 10 mg of donepezil hydrochloride.
The 5 mg tablets are white to off white, round shaped, biconvex, film-coated tablets debossed with ‘ML 89’ on one side and plain on the other side.
The 10 mg tablets are yellow, round shaped, biconvex, film-coated tablets debossed with ‘ML 88’ on one side and plain on the other side.4 CONTRAINDICATIONS
Donepezil hydrochloride tablets are contraindicated in patients with known hypersensitivity to donepezil hydrochloride or to piperidine derivatives.
5 WARNINGS AND PRECAUTIONS
5.1 Anesthesia
Donepezil hydrochloride, as a cholinesterase inhibitor, is likely to exaggerate succinylcholine-type muscle relaxation during anesthesia.
5.2 Cardiovascular Conditions
Because of their pharmacological action, cholinesterase inhibitors may have vagotonic effects on the sinoatrial and atrioventricular nodes. This effect may manifest as bradycardia or heart block in patients both with and without known underlying cardiac conduction abnormalities. Syncopal episodes have been reported in association with the use of donepezil hydrochloride.
5.3 Nausea and Vomiting
Donepezil hydrochloride, as a predictable consequence of its pharmacological properties, has been shown to produce diarrhea, nausea and vomiting. These effects, when they occur, appear more frequently with the 10mg/day dose than with the 5mg/day dose.
Although in most cases, these effects have been mild and transient, sometimes lasting one to three weeks, and have resolved during continued use of donepezil hydrochloride, patients should be observed closely at the initiation of treatment and after dose increases.5.4 Peptic Ulcer disease and GI bleeding
Through their primary action, cholinesterase inhibitors may be expected to increase gastric acid secretion due to increased cholinergic activity. Therefore, patients should be monitored closely for symptoms of active or occult gastrointestinal bleeding, especially those at increased risk for developing ulcers, e.g., those with a history of ulcer disease or those receiving concurrent nonsteroidal anti-inflammatory drugs (NSAIDS).
5.6 Genitourinary Conditions
Although not observed in clinical trials of donepezil hydrochloride, cholinomimetics may cause bladder outflow obstruction.
6 ADVERSE REACTIONS
6.1 Clinical Studies Experience
Donepezil hydrochloride tablets 5 mg / day and 10 mg / day
Mild To Moderate Alzheimer's Disease
Adverse Events Leading to Discontinuation
The rates of discontinuation from controlled clinical trials of donepezil hydrochloride due to adverse events for the donepezil hydrochloride 5 mg/day treatment groups were comparable to those of placebo treatment groups at approximately 5%. The rate of discontinuation of patients who received 7-day escalations from 5 mg/day to 10 mg/day, was higher at 13%.
The most common adverse events leading to discontinuation, defined as those occurring in at least 2% of patients and at twice the incidence seen in placebo patients, are shown in Table 1.
|
Table 1. Most Frequent Adverse Events Leading to Withdrawal from Controlled Clinical Trials by Dose Group |
|||
|
Dose Group |
Placebo |
5 mg/day Donepezil hydrochloride |
10 mg/day Donepezil hydrochloride |
| Patients Randomized
|
355 |
350 |
315 |
| Event/%Discontinuing
|
|
|
|
| Nausea |
1% |
1% |
3% |
| Diarrhea
|
0% |
< 1% |
3% |
| Vomiting |
<1% |
< 1% |
2% |
Most Frequent Adverse Clinical Events Seen in Association with the Use of Donepezil hydrochloride
The most common adverse events, defined as those occurring at a frequency of at least 5% in patients receiving 10 mg/day and twice the placebo rate, are largely predicted by donepezil hydrochloride cholinomimetic effects. These include nausea, diarrhea, insomnia, vomiting, muscle cramp, fatigue and anorexia. These adverse events were often of mild intensity and transient, resolving during continued donepezil hydrochloride treatment without the need for dose modification.
There is evidence to suggest that the frequency of these common adverse events may be affected by the rate of titration. An open-label study was conducted with 269 patients who received placebo in the 15 and 30-week studies. These patients were titrated to a dose of 10 mg/day over a 6-week period. The rates of common adverse events were lower than those seen in patients titrated to 10 mg/day over one week in the controlled clinical trials and were comparable to those seen in patients on 5 mg/day.
See Table 2 for a comparison of the most common adverse events following one and six week titration regimens.
|
Table 2. Comparison of rates of adverse events in mild to moderate patients titrated to 10 mg/day over 1 and 6 weeks |
||||
|
|
No titration |
One week titration |
Six week titration |
|
| Adverse Event
|
Placebo (n=315) |
5 mg/day (n=311) |
10 mg/day (n=315) |
10 mg/day (n=269) |
| Nausea |
6% |
5% |
19% |
6% |
| Diarrhea |
5% |
8% |
15% |
9% |
| Insomnia |
6% |
6% |
14% |
6% |
| Fatigue |
3% |
4% |
8% |
3% |
| Vomiting |
3% |
3% |
8% |
5% |
| Muscle cramps |
2% |
6% |
8% |
3% |
| Anorexia |
2% |
3% |
7% |
3% |
Adverse Events Reported in Controlled Trials
The events cited reflect experience gained under closely monitored conditions of clinical trials in a highly selected patient population. In actual clinical practice or in other clinical trials, these frequency estimates may not apply, as the conditions of use, reporting behavior, and the kinds of patients treated may differ. Table 3 lists treatment emergent signs and symptoms that were reported in at least 2% of patients in placebo-controlled trials who received donepezil hydrochloride and for which the rate of occurrence was greater for patients treated with donepezil hydrochloride than with placebo. In general, adverse events occurred more frequently in female patients and with advancing age.
|
Table 3. Adverse Events Reported in Controlled Clinical Trials in Mild to Moderate Alzheimer's Disease in at Least 2% of Patients Receiving donepezil hydrochloride and at a Higher Frequency than Placebo treated Patients |
||
| Body System/Adverse Event
|
Placebo (n=355) |
Donepezil hydrochloride (n=747) |
| Percent of Patients with any Adverse Event
|
72 |
74 |
| Body as a Whole
| | |
| Headache |
9 |
10 |
| Pain, various locations |
8 |
9 |
| Accident |
6 |
7 |
| Fatigue |
3 |
5 |
| Cardiovascular System
| | |
| Syncope |
1 |
2 |
| Digestive System
| | |
| Nausea |
6 |
11 |
| Diarrhea |
5 |
10 |
| Vomiting |
3 |
5 |
| Anorexia |
2 |
4 |
| Hemic and Lymphatic System
| | |
| Ecchymosis |
3 |
4 |
| Metabolic and Nutritional Systems
| | |
| Weight Decrease |
1 |
3 |
| Musculoskeletal System
| | |
| Muscle Cramps |
2 |
6 |
| Arthritis |
1 |
2 |
| Nervous System
| | |
| Insomnia |
6 |
9 |
| Dizziness |
6 |
8 |
| Depression |
<1 |
3 |
| Abnormal Dreams |
0 |
3 |
| Somnolence |
<1 |
2 |
| Urogenital System
| | |
| Frequent Urination |
1 |
2 |
Other Adverse Events Observed During Clinical Trials
Donepezil hydrochloride has been administered to over 1700 individuals during clinical trials worldwide. Approximately 1200 of these patients have been treated for at least 3 months and more than 1000 patients have been treated for at least 6 months. Controlled and uncontrolled trials in the United States included approximately 900 patients. In regards to the highest dose of 10 mg/day, this population includes 650 patients treated for 3 months, 475 patients treated for 6 months and 116 patients treated for over 1 year. The range of patient exposure is from 1 to 1214 days.
Treatment emergent signs and symptoms that occurred during three controlled clinical trials and two open-label trials in the United States were recorded as adverse events by the clinical investigators using terminology of their own choosing. To provide an overall estimate of the proportion of individuals having similar types of events, the events were grouped into a smaller number of standardized categories using a modified COSTART dictionary, and event frequencies were calculated across all studies. These categories are used in the listing below. The frequencies represent the proportion of 900 patients from these trials who experienced that event while receiving donepezil hydrochloride. All adverse events occurring at least twice are included, except for those already listed in Tables 2 or 3, COSTART terms too general to be informative, or events less likely to be drug related. Events are classified by body system and listed using the following definitions: Frequent adverse events - those occurring in at least 1/100 patients; Infrequent adverse events - those occurring in 1/100 to 1/1000 patients. These adverse events are not necessarily related to donepezil hydrochloride treatment and in most cases were observed at a similar frequency in placebo treated patients in the controlled studies. No important additional adverse events were seen in studies conducted outside the United States
Body as a Whole: Frequent:influenza, chest pain, toothache; Infrequent: fever, edema face, periorbital edema, hernia hiatal, abscess, cellulitis, chills, generalized coldness, head fullness, listlessness.
Cardiovascular System: Frequent:hypertension, vasodilation, atrial fibrillation, hot flashes, hypotension; Infrequent: angina pectoris, postural hypotension, myocardial infarction, AV block (first degree), congestive heart failure, arteritis, bradycardia, peripheral vascular disease, supraventricular tachycardia, deep vein thrombosis.
Digestive System: Frequent:fecal incontinence, gastrointestinal bleeding, bloating, epigastric pain; Infrequent: eructation, gingivitis, increased appetite, flatulence, periodontal abscess, cholelithiasis, diverticulitis, drooling, dry mouth, fever sore, gastritis, irritable colon, tongue edema, epigastric distress, gastroenteritis, increased transaminases, hemorrhoids, ileus, increased thirst, jaundice, melena, polydipsia, duodenal ulcer, stomach ulcer.
Endocrine System: Infrequent: diabetes mellitus, goiter.
Hemic and Lymphatic System: Infrequent: anemia, thrombocythemia, thrombocytopenia, eosinophilia, erythrocytopenia.
Metabolic and Nutritional Disorders: Frequent: dehydration; Infrequent: gout, hypokalemia, increased creatine kinase, hyperglycemia, weight increase, increased lactate dehydrogenase.
Musculoskeletal System: Frequent: bone fracture; Infrequent: muscle weakness, muscle fasciculation.
Nervous System: Frequent:delusions, tremor, irritability, paresthesia, aggression, vertigo, ataxia, increased libido, restlessness, abnormal crying, nervousness, aphasia; Infrequent: cerebrovascular accident, intracranial hemorrhage, transient ischemic attack, emotional lability, neuralgia, coldness (localized), muscle spasm, dysphoria, gait abnormality, hypertonia, hypokinesia, neurodermatitis, numbness (localized), paranoia, dysarthria, dysphasia, hostility, decreased libido, melancholia, emotional withdrawal, nystagmus, pacing.
Respiratory System: Frequent:dyspnea, sore throat, bronchitis; Infrequent: epistaxis, post nasal drip, pneumonia, hyperventilation, pulmonary congestion, wheezing, hypoxia, pharyngitis, pleurisy, pulmonary collapse, sleep apnea, snoring.
Skin and Appendages: Frequent:pruritus, diaphoresis, urticaria; Infrequent: dermatitis, erythema, skin discoloration, hyperkeratosis, alopecia, fungal dermatitis, herpes zoster, hirsutism, skin striae, night sweats, skin ulcer.
Special Senses: Frequent:cataract, eye irritation, vision blurred; Infrequent: dry eyes, glaucoma, earache, tinnitus, blepharitis, decreased hearing, retinal hemorrhage, otitis externa, otitis media, bad taste, conjunctival hemorrhage, ear buzzing, motion sickness, spots before eyes.
Urogenital System: Frequent:urinary incontinence, nocturia; Infrequent: dysuria, hematuria, urinary urgency, metrorrhagia, cystitis, enuresis, prostate hypertrophy, pyelonephritis, inability to empty bladder, breast fibroadenosis, fibrocystic breast, mastitis, pyuria, renal failure, vaginitis.
Severe Alzheimer's Disease
Adverse Events Leading to Discontinuation
The rates of discontinuation from controlled clinical trials of donepezil hydrochloride due to adverse events for the donepezil hydrochloride patients were approximately 12% compared to 7% for placebo patients. The most common adverse events leading to discontinuation, defined as those occurring in at least 2% of donepezil hydrochloridepatients and at twice or morethe incidence seen in placebo patients, were anorexia (2% vs. 1% placebo), nausea (2% vs. less than 1% placebo), diarrhea (2% vs. 0% placebo) and urinary tract infection (2% vs. 1% placebo).
Most Frequent Adverse Clinical Events Seen in Association with the Use of donepezil hydrochloride
The most common adverse events, defined as those occurring at a frequency of at least 5% in patients receiving donepezil hydrochloride and twice or more the placebo rate, are largely predicted by donepezil hydrochloride’s cholinomimetic effects. These include diarrhea, anorexia, vomiting, nausea, and ecchymosis. These adverse events were often of mild intensity and transient, resolving during continued donepezil hydrochloride treatment without the need for dose modification.
Adverse Events Reported in Controlled Trials
Table 4 lists adverse events that were reported in at least 2% of patients in placebo-controlled trials who received donepezil hydrochloride and for which the rate of occurrence was greater for patients treated with donepezil hydrochloride than with placebo.
|
Table 4. Adverse Events Reported in Controlled Clinical Trials in Severe Alzheimer's Disease in at Least 2% of Patients Receiving Donepezil hydrochloride and at a Higher Frequency than Placebo treated Patients |
||
|
Body System/Adverse Event |
Placebo (n=392) |
Donepezil hydrochloride (n=501) |
| Percent of Patients with any Adverse Event
|
73 |
81 |
| Body as a Whole
| | |
| Accident |
12 |
13 |
| Infection |
9 |
11 |
| Headache |
3 |
4 |
| Pain |
2 |
3 |
| Back Pain |
2 |
3 |
| Fever |
1 |
2 |
| Chest Pain |
<1 |
2 |
| Cardiovascular System
| | |
| Hypertension |
2 |
3 |
| Hemorrhage |
1 |
2 |
| Syncope |
1 |
2 |
| Digestive System
| | |
| Diarrhea |
4 |
10 |
| Vomiting |
4 |
8 |
| Anorexia |
4 |
8 |
| Nausea |
2 |
6 |
|
Hemic and Lymphatic System | | |
| Ecchymosis |
2 |
5 |
| Metabolic and Nutritional Systems
| | |
|
Creatine Phosphokinase Increased |
1 |
3 |
| Dehydration |
1 |
2 |
| Hyperlipemia |
<1 |
2 |
| Nervous System
| | |
| Insomnia |
4 |
5 |
| Hostility |
2 |
3 |
| Nervousness |
2 |
3 |
| Hallucinations |
1 |
3 |
| Somnolence |
1 |
2 |
| Dizziness |
1 |
2 |
| Depression |
1 |
2 |
| Confusion |
1 |
2 |
| Emotional Lability |
1 |
2 |
| Personality Disorder |
1 |
2 |
| Skin And Appendages
| | |
| Eczema |
2 |
3 |
| Urogenital System
| | |
| Urinary Incontinence |
1 |
2 |
Other Adverse Events Observed During Clinical Trials
Donepezil hydrochloride has been administered to over 600 patients with severe Alzheimer's disease during clinical trials of at least 6 months duration, including three double-blind placebo-controlled trials, two of which had an open label extension. All adverse events occurring at least twice are included, except for those already listed in Table 4, COSTART terms too general to be informative, or events less likely to be drug related. Events are classified by body system using the COSTART dictionary and listed using the following definitions: Frequent adverse events - those occurring in at least 1/100 patients; Infrequent adverse events - those occurring in 1/100 to 1/1000 patients. These adverse events are not necessarily related to donepezil hydrochloride treatment and in most cases were observed at a similar frequency in placebo treated patients in the controlled studies.
Body as a Whole: Frequent:abdominal pain, asthenia, fungal infection, flu syndrome; Infrequent: allergic reaction, cellulitis, malaise, sepsis, face edema, hernia.
Cardiovascular System: Frequent: hypotension, bradycardia, ECG abnormal, heart failure; Infrequent: myocardial infarction, angina pectoris, atrial fibrillation, congestive heart failure, peripheral vascular disorder, supraventricular extrasystoles, ventricular extrasystoles, cardiomegaly.
Digestive System: Frequent:constipation, gastroenteritis, fecal incontinence, dyspepsia; Infrequent: gamma glutamyl transpeptidase increase, gastritis, dysphagia, periodontitis, stomach ulcer, periodontal abscess, flatulence, liver function tests abnormal, eructation, esophagitis, rectal hemorrhage.
Endocrine System: Infrequent: diabetes mellitus.
Hemic and Lymphatic System: Frequent: anemia; Infrequent: leukocytosis
Metabolic and Nutritional Disorders: Frequent: weight loss, peripheral edema, edema, lactic dehydrogenase increased, alkaline phosphatase increased; Infrequent: hypercholesteremia, hypokalemia, hypoglycemia, weight gain, bilirubinemia, BUN increased, B12 deficiency anemia, cachexia, creatinine increased, gout, hyponatremia, hypoproteinemia, iron deficiency anemia, SGOT increased, SGPT increased.
Musculoskeletal System: Frequent: arthritis; Infrequent: arthrosis, bone fracture, arthralgia, leg cramps, osteoporosis, myalgia.
Nervous System: Frequent:agitation, anxiety, tremor, convulsion, wandering, abnormal gait; Infrequent: apathy, vertigo, delusions, abnormal dreams, cerebrovascular accident, increased salivation, ataxia, euphoria, vasodilatation, cerebral hemorrhage, cerebral infarction, cerebral ischemia, dementia, extrapyramidal syndrome, grand mal convulsion, hemiplegia, hypertonia, hypokinesia.
Respiratory System: Frequent:pharyngitis, pneumonia, cough increased, bronchitis; Infrequent: dyspnea, rhinitis, asthma
Skin and Appendages: Frequent:rash, skin ulcer, pruritus; Infrequent: psoriasis, skin discoloration, herpes zoster, dry skin, sweating, urticaria, vesiculobullous rash.
Special Senses: Infrequent:conjunctivitis, glaucoma, abnormal vision, ear pain, lacrimation disorder.
Urogenital System: Frequent:urinary tract infection, cystitis, hematuria, glycosuria; Infrequent: vaginitis, dysuria, urinary frequency, albuminuria
6.2 Postmarketing Experience
Voluntary reports of adverse events temporally associated with donepezil hydrochloride that have been received since market introduction that are not listed above, and for which there are inadequate data to determine the causal relationship with the drug include the following: abdominal pain, agitation, cholecystitis, confusion, convulsions, hallucinations, heart block (all types), hemolytic anemia, hepatitis, hyponatremia, neuroleptic malignant syndrome, pancreatitis, and rash.
7 DRUG INTERACTIONS
7.1 Effects of Other Drugs on the Metabolism of Donepezil Hydrochloride Tablets
Ketoconazole and quinidine, inhibitors of CYP450, 3A4 and 2D6, respectively, inhibit donepezil metabolism in vitro. Whether there is a clinical effect of quinidine is not known. In a 7-day crossover study in 18 healthy volunteers, ketoconazole (200 mg q.d.) increased mean Donepezil (5 mg q.d.) concentrations (AUC 0-24 and Cmax) by 36%. The clinical relevance of this increase in concentration is unknown.
Inducers of CYP 3A4 (e.g., phenytoin, carbamazepine, dexamethasone, rifampin, and phenobarbital) could increase the rate of elimination of donepezil hydrochloride tablets.
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Category C:There are no adequate or well- controlled studies in pregnant women. Donepezil hydrochloride should be used during pregnancy only if potential benefit justifies the potential risk to the fetus.
Oral administration of donepezil to pregnant rats and rabbits during the period of organogenesis did not produce any teratogenic effects at doses up to 16 mg/kg/day (approximately 6 times the maximum recommended human dose [MRHD] of 23 mg/day on a mg/m2 basis) and 10 mg/kg/day (approximately 7 times the MRHD on a mg/m2 basis), respectively. Oral administration of donepezil (1, 3, 10 mg/kg/day) to rats during late gestation and throughout lactation to weaning produced an increase in stillbirths and reduced offspring survival through postpartum day 4 at the highest dose. The no-effect dose of 3 mg/kg/day is approximately equal to the MRHD on a mg/m2 basis.
8.3 Nursing Mothers
It is not known whether donepezil is excreted in human breast milk. Caution should be exercised when donepezil hydrochloride is administered to a nursing woman.
8.4 Pediatric Use
The safety and effectiveness of donepezil hydrochloride in children have not been established.
8.5 Geriatric Use
Alzheimer's disease is a disorder occurring primarily in individuals over 55 years of age. The mean age of patients enrolled in the clinical study with donepezil hydrochloride was 73 years; 80 % of these patients were between 65 and 84 years old, and 49 % of patients were at or above the age of 75. The efficacy and safety data presented in the clinical trials section were obtained from these patients. There were no clinically significant differences in most adverse events reported by patient groups ≥ 65 years old and < 65 years old.
10 OVERDOSAGE
Because strategies for the management of overdose are continually evolving, it is advisable to contact a PoisonControlCenter to determine the latest recommendations for the management of an overdose of any drug.
As in any case of overdose, general supportive measures should be utilized. Overdosage with cholinesterase inhibitors can result in cholinergic crisis characterized by severe nausea, vomiting, salivation, sweating, bradycardia, hypotension, respiratory depression, collapse and convulsions. Increasing muscle weakness is a possibility and may result in death if respiratory muscles are involved. Tertiary anticholinergics such as atropine may be used as an antidote for donepezil hydrochloride overdosage. Intravenous atropine sulfate titrated to effect is recommended: an initial dose of 1.0 to 2.0 mg IV with subsequent doses based upon clinical response. Atypical responses in blood pressure and heart rate have been reported with other cholinomimetics when co-administered with quaternary anticholinergics such as glycopyrrolate. It is not known whether donepezil hydrochloride and/or its metabolites can be removed by dialysis (hemodialysis, peritoneal dialysis, or hemofiltration).
Dose-related signs of toxicity in animals included reduced spontaneous movement, prone position, staggering gait, lacrimation, clonic convulsions, depressed respiration, salivation, miosis, tremors, fasciculation and lower body surface temperature.
11 DESCRIPTION
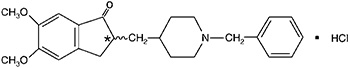
Donepezil hydrochloride USP is a reversible inhibitor of the enzyme acetylcholinesterase, known chemically as (±)-2, 3-dihydro-5, 6-dimethoxy-2-[[1-(phenylmethyl)-4-piperidinyl]methyl]-1H-inden-1-one hydrochloride. Donepezil hydrochloride USP is commonly referred to in the pharmacological literature as E2020. It has an empirical formula of C24H29NO3HCl and a molecular weight of 415.96. Donepezil hydrochloride USP is a white crystalline powder and is freely soluble in chloroform, soluble in water and in glacial acetic acid, slightly soluble in ethanol and in acetonitrile and practically insoluble in ethyl acetate and in n-hexane.
Donepezil hydrochloride USP is available for oral administration in film-coated tablets containing 5,or 10 mg of donepezil hydrochloride USP.
Inactive ingredients in 5mg and 10mg tablets are lactose monohydrate, pregelatinised starch, microcrystalline cellulose colloidal silicon dioxide and magnesium stearate. The film coating contains talc, polyethylene glycol, hypromellose and titanium dioxide. Additionally, the 10 mg tablet contains yellow iron oxide (synthetic) as a coloring agent.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Current theories on the pathogenesis of the cognitive signs and symptoms of Alzheimer's disease attribute some of them to a deficiency of cholinergic neurotransmission.
Donepezil hydrochloride is postulated to exert its therapeutic effect by enhancing cholinergic function. This is accomplished by increasing the concentration of acetylcholine through reversible inhibition of its hydrolysis by acetylcholinesterase. There is no evidence that donepezil alters the course of the underlying dementing process.
12.3 Pharmacokinetics
Pharmacokinetics of donepezil is linear over a dose range of 1 – 10mg given once daily. The rate and extent of absorption of donepezil hydrochloride tablets are not influenced by food.
Based on population pharmacokinetic analysis of plasma donepezil concentrations measured in patients with Alzheimer's disease, following oral dosing, peak plasma concentration is achieved in 3 hours for donpezil hydrochloride 10mg tablets.
The elimination half life of donepezil is about 70 hours, and the mean apparent plasma clearance (Cl/F) is 0.13 – 0.19 L/hr/kg. Following multiple dose administration, donepezil accumulates in plasma by 4-7 fold, and steady state is reached within 15 days. The steady state volume of distribution is 12 - 16 L/kg. Donepezil is approximately 96% bound to human plasma proteins, mainly to albumins (about 75%) and alpha1 - acid glycoprotein (about 21%) over the concentration range of 2-1000 ng/mL.
Donepezil is both excreted in the urine intact and extensively metabolized to four major metabolites, two of which are known to be active, and a number of minor metabolites, not all of which have been identified. Donepezil is metabolized by CYP 450 isoenzymes 2D6 and 3A4 and undergoes glucuronidation. Following administration of 14C-labeled donepezil, plasma radioactivity, expressed as a percent of the administered dose, was present primarily as intact donepezil (53%) and as 6-O-desmethyl donepezil (11%), which has been reported to inhibit AChE to the same extent as donepezil in vitro and was found in plasma at concentrations equal to about 20% of donepezil. Approximately 57% and 15% of the total radioactivity was recovered in urine and feces, respectively, over a period of 10 days, while 28% remained unrecovered, with about 17% of the donepezil dose recovered in the urine as unchanged drug. Examination of the effect of CYP2D6 genotype in Alzheimer's patients showed differences in clearance values among CYP2D6 genotype subgroups. When compared to the extensive metabolizers, poor metabolizers had a 31.5% slower clearance and ultra-rapid metabolizers had a 24% faster clearance. These results suggest CYP2D6 has a minor role in the metabolism of donepezil.
Hepatic Disease : In a study of 10 patients with stable alcoholic cirrhosis, the clearance of donepezil hydrochloride was decreased by 20% relative to 10 healthy age- and sex-matched subjects.
Renal Disease : In a study of 11 patients with moderate to severe renal impairment (ClC< 18 mL/min/1.73 m2) the clearance of donepezil hydrochloride did not differ from 11 age- and sex-matched healthy subjects.
Age : No formal pharmacokinetic study was conducted to examine age-related differences in the pharmacokinetics of donepezil hydrochloride. Population pharmacokinetic analysis suggested that clearance of donepezil in patients decreases with increasing age. When compared with 65- year old subjects, 90-year old subjects have a 17 % decrease in clearance, while 40-year old subjects have a 33 % increase in clearance. The effect of age on donepezil clearance may not be clinically significant.
Gender and Race : No specifc pharmacokinetic study was conducted to investigate the effects of gender and race on the disposition of donepezil hydrochloride. However, retrospective pharmacokinetic analysis and population pharmacokinetic analysis of plasma donepezil concentrations measured in patients with Alzheimer's disease indicates that the gender and race (Japanese and Caucasians) did not affect the clearance of donepezil hydrochloride to an important degree.
Body weight : There was a relationship noted between body weight and clearance. Over the range of weights from 50 kg to 110 kg, clearance increased from 7.77 L/h to 14.04 L/h to 14.04 L/h with a value of 10 L/hr for 70 kg individuals.
Drug Interactions: Effect of Donepezil Hydrochloride on the Metabolism of Other Drugs
No in vivo clinical trials have investigated the effect of donepezil hydrochloride on the clearance of drugs metabolized by CYP 3A4 (e.g. cisapride, terfenadine) or by CYP 2D6 (e.g. imipramine). However, in vitro studies show a low rate of binding to these enzymes (mean Ki about 50-130 µM), that, given the therapeutic plasma concentrations of donepezil (164 nM), indicates little likelihood of interference. Based on in vitro studies, donepezil shows little or no evidence of direct inhibition of CYP2B6, CYP2C8 and CYP2C19 at clinically relevant concentrations.
Whether donepezil hydrochloride has any potential for enzyme induction is not known. Formal pharmacokinetic studies evaluated the potential of donepezil hydrochloride for interaction with theophylline, cimetidine, warfarin, digoxin and ketoconazole. No effects of donepezil hydrochloride on the pharmacokinetics of these drugs were observed.
Effect of Other Drugs on the Metabolism of Donepezil Hydrochloride
A small effect of CYP2D6 inhibitors was identified in a population pharmacokinetic analysis of plasma donepezil concentrations measured in patients with Alzheimer’s disease. Donepezil clearance was reduced by approximately 17% in patients taking 10 or 23 mg in combination with a known CYP2D6 inhibitor. This result is consistent with the conclusion that CYP2D6 is a minor metabolic pathway of donepezil.
Formal pharmacokinetic studies demonstrated that the metabolism of donepezil hydrochloride is not significantly affected by concurrent administration of digoxin or cimetidine.
An in vitro study showed that donepezil was not a substrate of P-glycoprotein.
Drugs Highly Bound to Plasma Proteins
Drug displacement studies have been performed in vitro between this highly bound drug (96%) and other drugs such as furosemide, digoxin, and warfarin. Donepezil hydrochloride at concentrations of 0.3-10 micrograms/mL did not affect the binding of furosemide (5 micrograms/mL), digoxin (2 ng/mL), and warfarin (3 micrograms/mL) to human albumin. Similarly, the binding of donepezil hydrochloride to human albumin was not affected by furosemide, digoxin and warfarin.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis & Mutagenesis & Impairment Of Fertility
No evidence of a carcinogenic potential was obtained in an 88-week carcinogenicity study of donepezil hydrochloride conducted in mice at oral doses up to 180 mg/kg/day or in a 104-week carcinogenicity study in rats at doses up to 30 mg/kg/day (approximately 13 times the MRHD on a mg/m2 basis).
Donepezil was negative in a battery of genotoxicity assays (in vitro bacterial reverse mutation, in vitro mouse lymphoma tk, in vitro chromosomal aberration and in vivo mouse micronucleus).
Donepezil had no effect on fertility in rats at oral doses up to 10 mg/kg/day (approximately 4 times the MRHD on a mg/m2 basis) when administered to males and females prior to and during mating and continuing in females through implantation.
13.2 AnimalToxicology
In a published study, female rats were given single doses of donepezil and memantine by intraperitoneal injection, each alone or in combination. When given in combination with memantine, donepezil increased the incidence and severity of memantine-induced neurodegeneration. The relevance of this finding to humans is unknown.14 CLINICAL STUDIES
The effectiveness of donepezil hydrochloride as a treatment for Alzheimer's disease is demonstrated by the results of randomized, double-blind, placebo-controlled clinical investigation.
14.1 Mild to Moderate Alzheimer's disease
The effectiveness of donepezil hydrochloride as a treatment for mild to moderate Alzheimer's disease is demonstrated by the results of two randomized, double-blind, placebo-controlled clinical investigation in patients with Alzheimer's disease (diagnosed by NINCDS and DSM III-R criteria, Mini Mental State Examination ≥10 and ≤26 and Clinical Dementia Rating of 1 or 2). The mean age of patients participating in donepezil hydrochloride trials was 73 years, with a range of 50 to 94. Approximately 62 % of patients were women, and 38 % were men. The racial distribution was white 95 %, black 3 % and other races 2 %.
Study Outcome Measures:In each study, the effectiveness of treatment with donepezil hydrochloride was evaluated using a dual outcome assessment strategy.
The ability of donepezil hydrochloride to improve cognitive performance was assessed with the cognitive subscale of the Alzheimer’s Disease Assessment Scale (ADAS-cog), a multi-item instrument that has been extensively validated in longitudinal cohorts of Alzheimer’s disease patients. The ADAS-cog examines selected aspects of cognitive performance including elements of memory, orientation, attention, reasoning, language and praxis. The ADAS-cog scoring range is from 0 to 70, with higher scores indicating greater cognitive impairment. Elderly normal adults may score as low as 0 or 1, but it is not unusual for non-demented adults to score slightly higher.
The patients recruited as participants in each study had mean scores on the ADAS-cog of approximately 26 points, with a range from 4 to 61. Experience based on longitudinal studies of ambulatory patients with mild to moderate Alzheimer's disease suggest that scores on the ADAS-cog increase (worsen) by 6 - 12 points per year. However, smaller changes may be seen in patients with very mild or very advanced disease since the ADAS-cog is not uniformly sensitive to change over the course of the disease. The annualized rate of decline in the placebo patients participating in donepezil hydrochloride trials was approximately 2 to 4 points per year.
The ability of donepezil hydrochloride to produce an overall clinical effect was assessed using a Clinician's Interview-Based Impression of Change that required the use of caregiver information, the CIBIC-plus. The CIBIC-plus is not a single instrument and is not a standardized instrument like the ADAS-cog. Clinical trials for investigational drugs have used a variety of CIBIC formats, each different in terms of depth and structure.
As such, results from a CIBIC-plus reflect clinical experience from the trial or trials in which it was used and cannot be compared directly with the results of CIBIC-plus evaluations from other clinical trials. The CIBIC-plus used in donepezil hydrochloride trials was a semi-structured instrument that was intended to examine four major areas of patient function: General, Cognitive, Behavioral and Activities of Daily Living. It represents the assessment of a skilled clinician based upon his/her observations at an interview with the patient, in combination with information supplied by a caregiver familiar with the behavior of the patient over the interval rated. The CIBIC-plus is scored as a seven point categorical rating, ranging from a score of 1, indicating "markedly improved," to a score of 4, indicating "no change" to a score of 7, indicating "markedly worse." The CIBIC-plus has not been systematically compared directly to assessments not using information from caregivers (CIBIC) or other global methods.
Thirty-Week Study
In a study of 30 weeks duration, 473 patients were randomized to receive single daily doses of placebo, 5 mg/day or 10 mg/day of donepezil hydrochloride. The 30-week study was divided into a 24-week double-blind active treatment phase followed by a 6-week single-blind placebo washout period. The study was designed to compare 5 mg/day or 10 mg/day fixed doses of donepezil hydrochloride to placebo. However, to reduce the likelihood of cholinergic effects, the 10 mg/day treatment was started following an initial 7-day treatment with 5 mg/day doses.
Effects on the ADAS-cog:Figure 1 illustrates the time course for the change from baseline in ADAS-cog scores for all three dose groups over the 30 weeks of the study. After 24 weeks of treatment, the mean differences in the ADAS-cog change scores for donepezil hydrochloride treated patients compared to the patients on placebo were 2.8 and 3.1 points for the 5 mg/day and 10 mg/day treatments, respectively. These differences were statistically significant. While the treatment effect size may appear to be slightly greater for the 10 mg/day treatment, there was no statistically significant difference between the two active treatments.
Following 6 weeks of placebo washout, scores on the ADAS-cog for both the donepezil hydrochloridetreatment groups were indistinguishable from those patients who had received only placebo for 30 weeks. This suggests that the beneficial effects of donepezil hydrochloride abate over 6 weeks following discontinuation of treatment and do not represent a change in the underlying disease. There was no evidence of a rebound effect 6 weeks after abrupt discontinuation of therapy.

Figure 2 illustrates the cumulative percentages of patients from each of the three treatment groups who had attained the measure of improvement in ADAS-cog score shown on the X axis. Three change scores, (7-point and 4-point reductions from baseline or no change in score) have been identified for illustrative purposes, and the percent of patients in each group achieving that result is shown in the inset table.
The curves demonstrate that both patients assigned to placebo and donepezil hydrocloride have a wide range of responses, but that the active treatment groups are more likely to show greater improvements. A curve for an effective treatment would be shifted to the left of the curve for placebo, while an ineffective or deleterious treatment would be superimposed upon or shifted to the right of the curve for placebo.

Effects on the CIBIC-plus: Figure 3 is a histogram of the frequency distribution of CIBIC-plus scores attained by patients assigned to each of the three treatment groups who completed 24 weeks of treatment. The mean drug-placebo differences for these groups of patients were 0.35 points and 0.39 points for 5 mg/day and 10 mg/day of donpezil hydrochloride, respectively. These differences were statistically significant. There was no statistically significant difference between the two active treatments.

Fifteen-Week Study
In a study of 15 weeks duration, patients were randomized to receive single daily doses of placebo or either 5 mg/day or 10 mg/day of donepezil hydrocloride for 12 weeks, followed by a 3-week placebo washout period. As in the 30-week study, to avoid acute cholinergic effects, the 10 mg/day treatment followed an initial 7-day treatment with 5 mg/day doses.
Effects on the ADAS-Cog:Figure 4 illustrates the time course of the change from baseline in ADAS-cog scores for all three dose groups over the 15 weeks of the study. After 12 weeks of treatment, the differences in mean ADAS-cog change scores for the donepezil hydrochloride treated patients compared to the patients on placebo were 2.7 and 3.0 points each, for the 5 and 10 mg/day donepezil hydrochloridetreatment groups, respectively. These differences were statistically significant. The effect size for the 10 mg/day group may appear to be slightly larger than that for 5 mg/day. However, the differences between active treatments were not statistically significant.

Following 3 weeks of placebo washout, scores on the ADAS-cog for both the donepezil hydrochloridetreatment groups increased, indicating that discontinuation of donepezil hydrochloride resulted in a loss of its treatment effect. The duration of this placebo washout period was not sufficient to characterize the rate of loss of the treatment effect, but, the 30-week study (see above) demonstrated that treatment effects associated with the use of donepezil hydrochloride abate within 6 weeks of treatment discontinuation.
Figure 5 illustrates the cumulative percentages of patients from each of the three treatment groups who attained the measure of improvement in ADAS-cog score shown on the X axis. The same three change scores, (7-point and 4-point reductions from baseline or no change in score) as selected for the 30-week study have been used for this illustration. The percentages of patients achieving those results are shown in the inset table.
As observed in the 30-week study, the curves demonstrate that patients assigned to either placebo or to donepezil hydrochloride have a wide range of responses, but that the donepezil hydrochloride treated patients are more likely to show greater improvements in cognitive performance.

Effects on the CIBIC-plus:Figure 6 is a histogram of the frequency distribution of CIBIC-plus scores attained by patients assigned to each of the three treatment groups who completed 12 weeks of treatment. The differences in mean scores for donepezil hydrochloride treated patients compared to the patients on placebo at Week 12 were 0.36 and 0.38 points for the 5 mg/day and 10 mg/day treatment groups, respectively. These differences were statistically significant.

In both studies, patient age, sex and race were not found to predict the clinical outcome of donepezil hydrochloride treatment.
14.2 Moderate to Severe Alzheimer's Disease
The effectiveness of donepezil hydrochloride in the treatment of patients with moderate to severe Alzheimer’s Disease was established in studies employing doses of 10 mg/day.
Swedish 6 Month Study
The effectiveness of donepezil hydrochloride as a treatment for severe Alzheimer's disease is demonstrated by the results of a randomized, double-blind, placebo-controlled clinical study conducted in Sweden (6 month study) in patients with probable or possible Alzheimer's disease diagnosed by NINCDS-ADRDA and DSM-IV criteria, MMSE: range of 1-10. Two hundred and forty eight (248) patients with severe Alzheimer's disease were randomized to donepezil hydrochloride or placebo. For patients randomized to donepezil hydrochloride, treatment was initiated at 5 mg once daily for 28 days and then increased to 10 mg once daily. At the end of the 6 month treatment period, 90.5% of the donepezil hydrochloride treated patients were receiving the 10 mg/day dose. The mean age of patients was 84.9 years, with a range of 59 to 99. Approximately 77 % of patients were women, and 23 % were men. Almost all patients were Caucasian. Probable AD was diagnosed in the majority of the patients (83.6% of donepezil hydrochloride treated patients and 84.2% of placebo treated patients).
Study Outcome Measures:The effectiveness of treatment with donepezil hydrochloride was determined using a dual outcome assessment strategy that evaluated cognitive function using an instrument designed for more impaired patients and overall function through caregiver-rated assessment. This study showed that patients on donepezil hydrochloride experienced significant improvement on both measures compared to placebo.
The ability of donepezil hydrochloride to improve cognitive performance was assessed with the Severe Impairment Battery (SIB). The SIB, a multi-item instrument, has been validated for the evaluation of cognitive function in patients with moderate to severe dementia. The SIB evaluates selective aspects of cognitive performance, including elements of memory, language, orientation, attention, praxis, visuospatial ability, construction, and social interaction. The SIB scoring range is from 0 to 100, with lower scores indicating greater cognitive impairment.
Daily function was assessed using the Modified Alzheimer's Disease Cooperative Study Activities of Daily Living Inventory for Severe Alzheimer's Disease(ADCS-ADL-severe). The ADCS-ADL-severe is derived from the Alzheimer's Disease Cooperative Study Activities of Daily Living Inventory, which is a comprehensive battery of ADL questions used to measure the functional capabilities of patients. Each ADL item is rated from the highest level of independent performance to complete loss. The ADCS-ADL-severe is a subset of 19 items, including ratings of the patient's ability to eat, dress, bathe, use the telephone, get around (or travel), and perform other activities of daily living; it has been validated for the assessment of patients with moderate to severe dementia. The ADCS-ADL-severe has a scoring range of 0 to 54, with the lower scores indicating greater functional impairment. The investigator performs the inventory by interviewing a caregiver, in this study a nurse staff member, familiar with the functioning of the patient.
Effects on the SIB:
Figure 7 shows the time course for the change from baseline in SIB score for the two treatment groups over the 6 months of the study. At 6 months of treatment, the mean difference in the SIB change scores for donepezil hydrochloride treated patients compared to patients on placebo was 5.9 points. Donepezil hydrochloride treatment was statistically significantly superior to placebo.

Figure 8 illustrates the cumulative percentages of patients from each of the two treatment groups who attained the measure of improvement in SIB score shown on the X-axis. While patients assigned both to donepezil hydrochloride and to placebo have a wide range of responses, the curves show that the donepezil hydrochloride group is more likely to show a greater improvement in cognitive performance.


Effects on the ADCS-ADL-severe: Figure 9 illustrates the time course for the change from baseline in ADCS-ADL-severe scores for patients in the two treatment groups over the 6 months of the study. After 6 months of treatment, the mean difference in the ADCS-ADL-severe change scores for donepezil hydrochloride treated patients compared to patients on placebo was 1.8 points. Donepezil hydrochloride treatment was statistically significantly superior to placebo.
Figure 10 shows the cumulative percentages of patients from each treatment group with specified changes from baseline ADCS-ADL-severe scores. While both patients assigned to donepezil hydrochlorideand placebo have a wide range of responses, the curves demonstrate that the donepezil hydrochloridegroup is more likely to show a smaller decline or an improvement.

Japanese 24-Week Study
In a study of 24 weeks duration conducted in Japan, 325 patients with severe Alzheimer's disease were randomized to doses of 5 mg/day or 10 mg/day of donepezil, administered once daily, or placebo. Patients randomized to treatment with donepezil were to achieve their assigned doses by titration, beginning at 3 mg/day, and extending over a maximum of 6 weeks. Two hundred and forty eight (248) patients completed the study, with similar proportions of patients completing the study in each treatment group. The primary efficacy measures for this study were the SIB and CIBIC-plus.
At 24 weeks of treatment, statistically significant treatment differences were observed between the 10 mg/day dose of donepezil and placebo on both the SIB and CIBIC-plus. The 5 mg/day dose of donepezil showed a statistically significant superiority to placebo on the SIB, but not on the CIBIC-plus.
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 Donepezil Hyrochloride Tablets USP
Supplied as film-coated, round tablets containing either 5mg, or 10mg of donepezil hydrochloride.
The 5 mg tablets are white to off white, round shaped, biconvex, film-coated tablets debossed with ML;89 on one side and plain on the other side.
Bottles of 30 (NDC 33342-027-07 )
Bottles of 90 (NDC 33342-027-10 )
Blister Pack of 50 (5x10) (NDC 33342-027-31 )
Blister Pack of 100 (10x10) (NDC 33342-027-12)
The 10 mg tablets are yellow, round shaped, biconvex, film-coated tablets debossed with ML;88 on one side and plain on the other side.
Bottles of 30 (NDC 33342-028-07 )
Bottles of 90 (NDC 33342-028-10 )
Blister Pack of 50 (5x10) (NDC 33342-028-31 )
Blister Pack of 100 (10x10) (NDC33342-028-12)
Storage:Store at 20º-25ºC (68º-77ºF); [see USP Controlled Room Temperature].
17 PATIENT COUNSELING INFORMATION
See FDA-approved Patient Package Insert attached to this label.
To assure safe and effective use of donepezil hydrochloride, the information and instructions provided in the attached Patient Package Insert should be discussed with patients and care-givers.
Patients and care-givers should be instructed to take donepezil hydrochloride only once per day, as prescribed.
Patients and care-givers should be instructed that donepezil hydrochloride tablets should not be split or crushed. Donepezil hydrochloride tablets should be swallowed whole.
Patients and care-givers should be advised that the product may cause nausea, diarrhea, insomnia, vomiting, muscle cramps, fatigue and decreased appetite.
PATIENT PACKAGE INSERT
Donepezil hydrochloride tablets USP
· Tablets: 5mg, and 10mg
Read the Patient Information that comes with donepezil hydrochloride tablets before the patient starts taking it and each time you get a refill. There may be new information. This leaflet does not take the place of talking to the doctor about Alzheimer's disease or treatment for it. If you have questions ask the doctor or the pharmacist.
What are donepezil hydrochloride tablets?
Donepezil hydrochloride tablets come as film-coated tablets in dosage strengths of 5mg and 10mg.
Donepezil hydrochloride is a prescription medicine to treat mild, moderate and severe Alzheimer's disease (up to 10mg). Donepezil hydrochloride tablets can help in mental function and with doing daily tasks. Donepezil hydrochloride does not work the same in all people.
Some people may:
· Seem much better
· Get better in small ways or stay the same
· Get worse over time but slower than expected
· Not change and get worse as expected
Donepezil hydrochloride tablets do not cure Alzheimer's disease. All patients with Alzheimer's disease get worse over time, even if they take donepezil hydrochloride.
Donepezil hydrochloride tablets have not been approved as a treatment for any medical condition in children.
Who should not take donepezil hydrochloride tablets?
The patient should not take donepezil hydrochloride tablets if allergic to any of the ingredients in donepezil hydrochloride tablets or to medicines that contain piperidines. Ask the patient’s doctor if you are not sure. See the end of this leaflet for a list of ingredients in donepezil hydrochloride tablets.
What should I tell the doctor before the patient takes donepezil hydrochloride tablets?
Tell the doctor about all the patient’s present or past health problems.
Include:
· Any heart problems including problems with irregular, slow, or fast heartbeats
· Asthama or lung problems
· A seizure
· Stomach ulcers
· Difficulty passing urine
· Liver or kidney problems
· Trouble swallowing tablets
· Present pregnancy or plans to become pregnant. It is not known if donepezil hydrochloride can harm an unborn baby.
· Present breast-feeding. It is not known if donepezil hydrochloride passes into breast milk. Donepezil hydrochloride tablets are not for women who are breast-feeding.
Tell the doctor about all medicines the patient takes, including prescription and non-prescription medicines, vitamins and herbal products. Donepezil hydrochloride and other medicines may affect each other.
Be particularly sure to tell the doctor if the patient takes aspirin or medicines called non-steroidal anti-inflammatory drugs (NSAIDS). There are many NSAID medicines, both prescription and non-prescription. Ask your doctor or pharmacist if you are not sure if any of the patients medicines are NSAIDs. Taking NSAID and donepezil hydrochloride together may make the patient more likely to get stomach ulcers.
Donepezil hydrochloride tablets taken with certain medicines used for anesthesia may cause side effects. Tell the responsible doctor or dentist that the patient takes donepezil hydrochloride tablets before the patient has:
· surgery
· medical procedures
· dental surgery or procedures
Know the medicines that the patient takes. Keep a list of patient’s medicines. Show it to the doctor or pharmacist before the patient starts a new medicine.
How should the patient take donepezil hydrochloride tablets?
· Give donepezil hydrochloride tablets exactly as prescribed by the doctor. Do not stop donepezil hydrochloride tablets or change the dose yourself. Talk with the doctor first.
· Give donepezil hydrochloride tablets one time each day. Donepezil hydrochloride tablets can be taken with or without food.
· Donepezil hydrochloride Tablets should be swallowed whole without the tablets being broken or crushed.
· If you miss giving the patient a dose of donepezil hydrochloride tablets, just wait. Give only the next dose at the usual time. Do not give 2 doses at the same time.
· If donepezil hydrochloride tablets are missed for 7 days or more, talk with the doctor before starting again.
· If the patient takes too much donepezil hydrochloride at one time, call the doctor or poison control center, or go to the emergency room right away.
What are the possible side effects of donepezil hydrochloride tablets?
Donepezil hydrochloride tablets may cause the following serious side effects:
· slow heartbeat and fainting. This happens more often in people with heart problems. Call the doctor right away if the patient faints while taking donepezil hydrochloride tablets.
· more stomach acid: This raises the chance of ulcers and bleeding.. The risk is higher for patients who had ulcers, or take aspirin or other NSAIDs.
· worsening of lung problem in people with asthma or other lung diseases.
· siezures.
· difficulty passing urine.
Call the doctor right awayif the patient has:
· fainting.
· heartburn or stomach pain that is new or won’t go away.
· nausea or vomiting, blood in the vomit, dark vomit that looks like coffee grounds.
· bowel movement or stools that look like black tar.
· new or worse asthma or breathing problem.
· seizures.
· difficulty passing urine.
The most common side effects of donepezil hydrochloride tablets are:
· nausea
· diarrhea
· not sleeping well
· vomiting
· muscle cramps
· feeling tired
· not wanting to eat
These side effects may get better after the patient takes donepezil hydrochloride for a while. This is not a complete list of side effects with donepezil hydrochloride tablets. For more information, ask the doctor or pharmacist.
Call your doctor for medical advise about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should Donepezil hydrochloride tablets be stored?
Store donepezil hydrochloride tablets at controlled room temperature between 59º to 68º-77ºF (20º to 25ºC).
Keep donepezil hydrochloride tablets and all medicines out of the reach of children.
General information about donepezil hydrochloride tablets
Medicines are sometimes prescribed for conditions that are not mentioned in this Patient Information Leaflet. Do not use donepezil hydrochloride tablets for a condition for which it was not prescribed. Do not give donepezil hydrochloride tablets to people other than the patient, even if they have the same symptoms as the patient, as it may harm them.
This leaflet summarizes the most important information about donepezil hydrochloride tablets. If you would like more information talk with the patients doctor. You can ask your pharmacist or doctor for information about donepezil hydrochloride tablets that is written for health professionals.
For more information, call 1-888-943-3210.
What are the ingredients in donepezil hydrochloride tablets?
Active ingredient: donepezil hydrochloride USP
Inactive Ingredients:
· Donepezil hydrochloride 5mg and 10mg film-coated tablets: lactose monohydrate, pregelatinised starch, microcrystalline cellulose, colloidal silicon dioxide, and magnesium stearate. The film-coating contains talc, polyethylene glycol, hypromellose, and titanium dioxide. Additionally, the 10mg tablet contains yellow iron oxide (synthetic) as a coloring agent.
Macleods Pharma USA, INC,.
Plainsboro, NJ 08536
Manufactured by:
Macleods Pharmaceutical Ltd.
Baddi, Himachal Pradesh, India.
Rx only
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL-5 mg 30 Tablets
NDC 33342-027-07
Rx only
30 TABLETS
Donepezil Hydrochloride Tablets USP
5 mg

PACKAGE LABEL.PRINCIPAL DISPLAY PANEL-5 mg 90 Tablets
NDC 33342-027-10
Rx only
90 TABLETS
Donepezil Hydrochloride Tablets USP
5 mg

PACKAGE LABEL.PRINCIPAL DISPLAY PANEL-10 mg 30 Tablets
NDC 33342-028-07
Rx only
30 TABLETS
Label Donepezil Hydrochloride Tablets USP
10 mg

| DONEPEZIL HYDROCHLORIDE
donepezil hydrochloride tablet, film coated |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| DONEPEZIL HYDROCHLORIDE
donepezil hydrochloride tablet, film coated |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| Labeler - Macleods Pharmaceuticals Limited (862128535) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
| Macleods Pharmaceuticals Limited | 676369519 | ANALYSIS(33342-027, 33342-028), LABEL(33342-027, 33342-028), MANUFACTURE(33342-027, 33342-028), PACK(33342-027, 33342-028) | |