promethazine hydrochloride (Promethazine Hydrochloride) syrup
[Morton Grove Pharmaceuticals, Inc.]
Rx only
DESCRIPTION
Each 5 mL (teaspoonful) contains:
Promethazine Hydrochloride, USP . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 6.25 mg
(In a flavored syrup base with a pH between 4.7 and 5.2)
Alcohol . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 7.0%
In addition, the following inactive ingredients are present: artificial banana flavor, artificial fruit flavor, artificial strawberry flavor, ascorbic acid, USP, citric acid anhydrous, USP, D&C Yellow No. 10, edetate disodium, USP, FD&C Blue No. 1, FD&C Red No. 40, glycerin, USP, liquid sugar, methylparaben, NF, purified water, USP, saccharin sodium, USP, sodium benzoate, NF, sodium citrate dihydrate, USP, and sodium propionate, NF.
Promethazine hydrochloride is a racemic compound; the molecular formula is C17H20N2S•HCl and its molecular weight is 320.89.
Promethazine hydrochloride, a phenothiazine derivative, is chemically designated as 10-[2-(Dimethylamino)propyl] phenothiazine monohydrochloride. Its structural formula is:
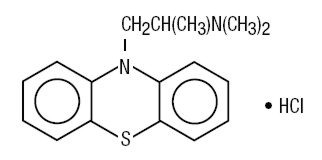
Promethazine hydrochloride occurs as a white to faint yellow, practically odorless, crystalline powder which slowly oxidizes and turns blue on prolonged exposure to air. It is freely soluble in water and soluble in alcohol.
CLINICAL PHARMACOLOGY
Promethazine is a phenothiazine derivative which differs structurally from the antipsychotic phenothiazines by the presence of a branched side chain and no ring substitution. It is thought that this configuration is responsible for its relative lack (1/10 that of chlorpromazine) of dopaminergic Central Nervous System (CNS) action.
Promethazine is an H1 receptor blocking agent. In addition to its antihistaminic action, it provides clinically useful sedative and antiemetic effects. In therapeutic dosage, promethazine produces no significant effects on the cardiovascular system.
Promethazine is well absorbed from the gastrointestinal tract. Clinical effects are apparent within 20 minutes after oral administration and generally last four to six hours, although they may persist as long as 12 hours. Promethazine is metabolized by the liver to a variety of compounds; the sulfoxides of promethazine and N-demethylpromethazine are the predominant metabolites appearing in the urine.
INDICATIONS AND USAGE
Promethazine is useful for:
Perennial and seasonal allergic rhinitis.
Vasomotor rhinitis.
Allergic conjunctivitis due to inhalant allergens and foods.
Mild, uncomplicated allergic skin manifestations of urticaria and angioedema.
Amelioration of allergic reactions to blood or plasma.
Dermographism.
Anaphylactic reactions, as adjunctive therapy to epinephrine and other standard measures, after the acute manifestations have been controlled.
Preoperative, postoperative, or obstetric sedation.
Prevention and control of nausea and vomiting associated with certain types of anesthesia and surgery.
Therapy adjunctive to meperidine or other analgesics for control of postoperative pain.
Sedation in both children and adults, as well as relief of apprehension and production of light sleep from which the patient can be easily aroused.
Active and prophylactic treatment of motion sickness.
Antiemetic therapy in postoperative patients.
CONTRAINDICATIONS
Promethazine products are contraindicated for use in pediatric patients less than two years of age.
Promethazine is contraindicated in individuals known to be hypersensitive or to have had an idiosyncratic reaction to promethazine or to other phenothiazines.
Antihistamines are contraindicated for use in the treatment of lower respiratory tract symptoms, including asthma.
WARNINGS
PROMETHAZINE SHOULD NOT BE USED IN PEDIATRIC PATIENTS LESS THAN 2 YEARS OF AGE BECAUSE OF THE POTENTIAL FOR FATAL RESPIRATORY DEPRESSION.
POSTMARKETING CASES OF RESPIRATORY DEPRESSION, INCLUDING FATALITIES, HAVE BEEN REPORTED WITH USE OF PROMETHAZINE IN PEDIATRIC PATIENTS LESS THAN 2 YEARS OF AGE. A WIDE RANGE OF WEIGHT-BASED DOSES OF PROMETHAZINE HAVE RESULTED IN RESPIRATORY DEPRESSION IN THESE PATIENTS.
CAUTION SHOULD BE EXERCISED WHEN ADMINISTERING PROMETHAZINE TO PEDIATRIC PATIENTS 2 YEARS OF AGE AND OLDER. IT IS RECOMMENDED THAT THE LOWEST EFFECTIVE DOSE OF PROMETHAZINE BE USED IN PEDIATRIC PATIENTS 2 YEARS OF AGE AND OLDER AND CONCOMITANT ADMINISTRATION OF OTHER DRUGS WITH RESPIRATORY DEPRESSANT EFFECTS BE AVOIDED.
Promethazine may cause marked drowsiness. Ambulatory patients should be cautioned against such activities as driving or operating dangerous machinery until it is known that they do not become drowsy or dizzy from promethazine therapy.
The sedative action of promethazine hydrochloride is additive to the sedative effects of CNS depressants; therefore, agents such as alcohol, narcotic analgesics, sedatives, hypnotics and tranquilizers should either be eliminated or given in reduced dosage in the presence of promethazine hydrochloride. When given concomitantly with promethazine hydrochloride, the dose of barbiturates should be reduced by at least one-half, and the dose of analgesic depressants, such as morphine or meperidine, should be reduced by one-quarter to one-half.
Promethazine may lower seizure threshold. This should be taken into consideration when administering to persons with known seizure disorders, or when giving in combination with narcotics or local anesthetics which may also affect seizure threshold.
Sedative drugs or CNS depressants should be avoided in patients with a history of sleep apnea.
Antihistamines should be used with caution in patients with narrow-angle glaucoma, stenosing peptic ulcer, pyloroduodenal obstruction and urinary bladder obstruction due to symptomatic prostatic hypertrophy and narrowing of the bladder neck.
Administration of promethazine has been associated with reported cholestatic jaundice.
Use in Pediatric Patients
PROMETHAZINE PRODUCTS ARE CONTRAINDICATED FOR USE IN PEDIATRIC PATIENTS LESS THAN TWO YEARS OF AGE.
CAUTION SHOULD BE EXERCISED WHEN ADMINISTERING PROMETHAZINE PRODUCTS TO PEDIATRIC PATIENTS 2 YEARS OF AGE AND OLDER BECAUSE OF THE POTENTIAL FOR FATAL RESPIRATORY DEPRESSION. RESPIRATORY DEPRESSION AND APNEA, SOMETIMES ASSOCIATED WITH DEATH, ARE STRONGLY ASSOCIATED WITH PROMETHAZINE PRODUCTS AND ARE NOT DIRECTLY RELATED TO INDIVIDUALIZED WEIGHT-BASED DOSING, WHICH MIGHT OTHERWISE PERMIT SAFE ADMINISTRATION. CONCOMITANT ADMINISTRATION OF PROMETHAZINE PRODUCTS WITH OTHER RESPIRATORY DEPRESSANTS HAS AN ASSOCIATION WITH RESPIRATORY DEPRESSION, AND SOMETIMES DEATH, IN PEDIATRIC PATIENTS.
ANTIEMETICS ARE NOT RECOMMENDED FOR TREATMENT OF UNCOMPLICATED VOMITING IN PEDIATRIC PATIENTS, AND THEIR USE SHOULD BE LIMITED TO PROLONGED VOMITING OF KNOWN ETIOLOGY. THE EXTRAPYRAMIDAL SYMPTOMS WHICH CAN OCCUR SECONDARY TO PROMETHAZINE PRODUCTS ADMINISTRATION MAY BE CONFUSED WITH THE CNS SIGNS OF UNDIAGNOSED PRIMARY DISEASE, e.g., ENCEPHALOPATHY OR REYE'S SYNDROME. THE USE OF PROMETHAZINE PRODUCTS SHOULD BE AVOIDED IN PEDIATRIC PATIENTS WHOSE SIGNS AND SYMPTOMS MAY SUGGEST REYE'S SYNDROME OR OTHER HEPATIC DISEASES.
Excessively large dosages of antihistamines, including Promethazine products, in pediatric patients may cause sudden death (see OVERDOSAGE). Hallucinations and convulsions have occurred with therapeutic doses and overdoses of Promethazine in pediatric patients. In pediatric patients who are acutely ill associated with dehydration, there is an increased susceptibility to dystonias with the use of promethazine HCl.
PRECAUTIONS
General
Promethazine should be used cautiously in persons with cardiovascular disease or with impairment of liver function.
Information for Patients
Promethazine may cause marked drowsiness or may impair the mental and/or physical abilities required for the performance of potentially hazardous tasks, such as driving a vehicle or operating machinery. Ambulatory patients should be told to avoid engaging in such activities until it is known that they do not become drowsy or dizzy from promethazine therapy. Children should be supervised to avoid potential harm in bike riding or in other hazardous activities.
The concomitant use of alcohol or other CNS depressants, including narcotic analgesics, sedatives, hypnotics and tranquilizers, may have an additive effect and should be avoided or their dosage reduced.
Patients should be advised to report any involuntary muscle movements or unusual sensitivity to sunlight.
Drug Interactions
The sedative action of promethazine is additive to the sedative effects of other CNS depressants, including alcohol, narcotic analgesics, sedatives, hypnotics, tricyclic antidepressants and tranquilizers; therefore, these agents should be avoided or administered in reduced dosage to patients receiving promethazine.
Drug/Laboratory Test Interactions
The following laboratory tests may be affected in patients who are receiving therapy with promethazine hydrochloride:
Pregnancy Tests
Diagnostic pregnancy tests based on immunological reactions between HCG and anti-HCG may result in false-negative or false-positive interpretations.
Glucose Tolerance Test
An increase in blood glucose has been reported in patients receiving promethazine.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term animal studies have not been performed to assess the carcinogenic potential of promethazine, nor are there other animal or human data concerning the carcinogenicity, mutagenicity or impairment of fertility with this drug. Promethazine was nonmutagenic in the Salmonella test system of Ames.
Pregnancy
Teratogenic Effects—Pregnancy Category C
Teratogenic effects have not been demonstrated in rat-feeding studies at doses of 6.25 and 12.5 mg/kg of promethazine. These doses are from approximately 2.1 to 4.2 times the maximum recommended total daily dose of promethazine for a 50-kg subject, depending upon the indication for which the drug is prescribed. Specific studies to test the action of the drug on parturition, lactation and development of the animal neonate were not done, but a general preliminary study in rats indicated no effect on these parameters. Although antihistamines, including promethazine, have been found to produce fetal mortality in rodents, the pharmacological effects of histamine in the rodent do not parallel those in man. There are no adequate and well-controlled studies of promethazine in pregnant women. Promethazine should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Nonteratogenic Effects
Promethazine taken within two weeks of delivery may inhibit platelet aggregation in the newborn.
Labor and Delivery
Promethazine, in appropriate dosage form, may be used alone or as an adjunct to narcotic analgesics during labor and delivery (See " INDICATIONS AND USAGE" and " DOSAGE AND ADMINISTRATION").
See also " Nonteratogenic Effects".
Nursing Mothers
It is not known whether promethazine is excreted in human milk. Caution should be exercised when promethazine is administered to a nursing woman.
Pediatric Use
PROMETHAZINE PRODUCTS ARE CONTRAINDICATED FOR USE IN PEDIATRIC PATIENTS LESS THAN TWO YEARS OF AGE (see WARNINGS-Black Box Warning and Use in Pediatric Patients).
Promethazine products should be used with caution in pediatric patients 2 years of age and older (see WARNINGS-Use in Pediatric Patients).
Geriatric Use
Clinical studies of Promethazine formulations did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal or cardiac function, and of concomitant disease or other drug therapy.
Sedating drugs may cause confusion and over-sedation in the elderly; elderly patients generally should be started on low doses of Promethazine products and observed closely.
ADVERSE REACTIONS
CNS–Sedation, sleepiness, occasional blurred vision, dryness of mouth, dizziness; rarely confusion, disorientation and extrapyramidal symptoms such as oculogyric crisis, torticollis and tongue protrusion (usually in association with parenteral injection or excessive dosage).
Cardiovascular–Increased or decreased blood pressure.
Dermatologic–Rash, rarely photosensitivity.
Hematologic–Rarely leukopenia, thrombocytopenia; agranulocytosis (1 case).
Gastrointestinal–Nausea and vomiting.
OVERDOSAGE
Signs and symptoms of overdosage with promethazine range from mild depression of the CNS and cardiovascular system to profound hypotension, respiratory depression and unconsciousness.
Stimulation may be evident, especially in children and geriatric patients. Convulsions may rarely occur. A paradoxical reaction has been reported in children receiving single doses of 75 mg to 125 mg orally, characterized by hyperexcitability and nightmares.
Atropine-like signs and symptoms – dry mouth, fixed dilated pupils, flushing, as well as gastrointestinal symptoms, may occur.
Treatment
The treatment of overdosage with promethazine is essentially symptomatic and supportive. Only in cases of extreme overdosage or individual sensitivity do vital signs including respiration, pulse, blood pressure, temperature and EKG need to be monitored. Activated charcoal orally or by lavage may be given, or sodium or magnesium sulfate orally as a cathartic. Attention should be given to the reestablishment of adequate respiratory exchange through provision of a patent airway and institution of assisted or controlled ventilation. Diazepam may be used to control convulsions. Acidosis and electrolyte losses should be corrected. Note that any of the depressant effects of promethazine are not reversed by naloxone. Avoid analeptics which may cause convulsions.
Severe hypotension usually responds to the administration of norepinephrine or phenylephrine. EPINEPHRINE SHOULD NOT BE USED since its use in patients with partial adrenergic blockade may further lower the blood pressure.
Limited experience with dialysis indicates that it is not helpful.
DOSAGE AND ADMINISTRATION
Promethazine products are contraindicated for children under 2 years of age (see WARNINGS-Black Box Warning and Use in Pediatric Patients).
ALLERGY
The average oral dose is 25 mg taken before retiring; however, 12.5 mg may be taken before meals and on retiring, if necessary. Children tolerate this product well. Single 25 mg doses at bedtime or 6.25 to 12.5 mg taken three times daily will usually suffice. After initiation of treatment in children or adults, dosage should be adjusted to the smallest amount adequate to relieve symptoms.
Promethazine hydrochloride rectal suppositories may be used if the oral route is not feasible, but oral therapy should be resumed as soon as possible if continued therapy is indicated.
The administration of promethazine hydrochloride in 25 mg doses will control minor transfusion reactions of an allergic nature.
MOTION SICKNESS
The average adult dose is 25 mg taken twice daily. The initial dose should be taken one-half to one hour before anticipated travel and be repeated 8 to 12 hours later, if necessary. On succeeding days of travel, it is recommended that 25 mg be given on arising and again before the evening meal. For children, promethazine tablets, syrup or rectal suppositories, 12.5 to 25 mg, twice daily, may be administered.NAUSEA AND VOMITING
The average effective dose of promethazine for the active therapy of nausea and vomiting in children or adults is 25 mg. When oral medication cannot be tolerated, the dose should be given parenterally (cf. promethazine hydrochloride injection) or by rectal suppository. 12.5 to 25 mg doses may be repeated as necessary at 4- to 6-hour intervals.
For nausea and vomiting in children, the usual dose is 0.5 mg per pound of body weight, and the dose should be adjusted to the age and weight of the patient and the severity of the condition being treated.
For prophylaxis of nausea and vomiting, as during surgery and the postoperative period, the average dose is 25 mg repeated at 4- to 6-hour intervals, as necessary.
SEDATION
This product relieves apprehension and induces a quiet sleep from which the patient can be easily aroused. Administration of 12.5 to 25 mg promethazine by the oral route or by rectal suppository at bedtime will provide sedation in children. Adults usually require 25 to 50 mg for nighttime, presurgical, or obstetrical sedation.
PRE- AND POSTOPERATIVE USE
Promethazine in 12.5 to 25 mg doses for children and 50 mg doses for adults the night before surgery relieves apprehension and produces a quiet sleep.
For preoperative medication, children require doses of 0.5 mg per pound of body weight in combination with an equal dose of meperidine and the appropriate dose of an atropine-like drug.
Usual adult dosage is 50 mg promethazine with an equal amount of meperidine and the required amount of a belladonna alkaloid.
Postoperative sedation and adjunctive use with analgesics may be obtained by the administration of 12.5 to 25 mg in children and 25 to 50 mg doses in adults.
Promethazine Syrup Plain is contraindicated for children under 2 years of age.
HOW SUPPLIED
Promethazine Hydrochloride Syrup, USP 6.25 mg/5 mL is supplied as a clear, green syrup. Available in 4 fl oz (118 mL), 8 fl oz (237 mL) and pint (473 mL) bottles.
RECOMMENDED STORAGE
Store at 20 ° to 25 °C (68 ° to 77 °F). [See USP Controlled Room Temperature].
KEEP TIGHTLY CLOSED
PROTECT FROM LIGHT
Dispense in a tight, light-resistant container as defined in the USP.
Rx Only
Product No.: 7608
Manufactured By:
Morton Grove Pharmaceuticals, Inc.
Morton Grove, IL 60053
A50-7608-04
REV. 07-05
| Promethazine Hydrochloride (Promethazine Hydrochloride) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Revised: 02/2006