NEXAVAR
-
sorafenib tablet, film coated
----------
NEXAVAR®
(sorafenib)
tablets, oral
|
|||||||||||||||||||||||||
FULL PRESCRIBING INFORMATION
1. INDICATIONS AND USAGE
1.1 Hepatocellular Carcinoma
NEXAVAR is indicated for the treatment of patients with unresctable hepatocellular carcinoma (HCC).
1.2 Renal Cell Carcinoma
NEXAVAR is indicated for the treatment of patients with advanced renal cell carcinoma (RCC).
2. DOSAGE AND ADMINISTRATION
The recommended daily dose of NEXAVAR is 400 mg (2 x 200 mg tablets) taken twice daily without food (at least 1 hour before or 2 hours after a meal). Treatment should continue until the patient is no longer clinically benefiting from therapy or until unacceptable toxicity occurs.
Management of suspected adverse drug reactions may require temporary interruption and/or dose reduction of NEXAVAR therapy. When dose reduction is necessary, the NEXAVAR dose may be reduced to 400 mg once daily. If additional dose reduction is required, NEXAVAR may be reduced to a single 400 mg dose every other day [see Warnings and Precautions (5)].
Suggested dose modifications for skin toxicity are outlined in Table 1.
| Skin Toxicity Grade | Occurrence | Suggested Dose Modification |
| Grade 1: Numbness, dysesthesia, paresthesia, tingling, painless swelling, erythema or discomfort of the hands or feet which does not disrupt the patient’s normal activities | Any occurrence | Continue treatment with NEXAVAR and consider topical therapy for symptomatic relief |
| Grade 2: Painful erythema and swelling of the hands or feet and/or discomfort affecting the patient’s normal activities | 1st occurrence |
Continue treatment with NEXAVAR and consider topical therapy for symptomatic relief If no improvement within 7 days, see below |
| No improvement within 7 days or 2nd or 3rd occurrence |
Interrupt NEXAVAR treatment until toxicity resolves to Grade 0-1 When resuming treatment, decrease NEXAVAR dose by one dose level (400 mg daily or 400 mg every other day) |
|
| 4th occurrence | Discontinue NEXAVAR treatment | |
| Grade 3: Moist desquamation, ulceration, blistering or severe pain of the hands or feet, or severe discomfort that causes the patient to be unable to work or perform activities of daily living | 1st or 2nd occurrence |
Interrupt NEXAVAR treatment until toxicity resolves to Grade 0-1 When resuming treatment, decrease NEXAVAR dose by one dose level (400 mg daily or 400 mg every other day) |
| 3rd occurrence | Discontinue NEXAVAR treatment |
No dose adjustment is required on the basis of patient age, gender, or body weight.
Concomitant strong CYP3A4 inducers: The use of concomitant strong CYP3A4 inducers may decrease sorafenib plasma concentrations and should be avoided (e.g., St. John's Wort, dexamethasone, phenytoin, carbamazepine, rifampin, rifabutin, phenobarbital). Although a dose increase has not been studied, if a strong CYP3A4 inducer must be co-administered, a NEXAVAR dose increase may be considered. If the dose of NEXAVAR is increased, the patient should be monitored carefully for toxicity [see Drug Interactions (7.6)].
3. DOSAGE FORMS AND STRENGTHS
Tablets containing sorafenib tosylate (274 mg) equivalent to 200 mg of sorafenib.
NEXAVAR tablets are round, biconvex, red film-coated tablets, debossed with the “Bayer cross” on one side and “200” on the other side.
4. CONTRAINDICATIONS
NEXAVAR is contraindicated in patients with known severe hypersensitivity to sorafenib or any other component of NEXAVAR.
5. WARNINGS AND PRECAUTIONS
5.1 Risk of Cardiac Ischemia and/or Infarction
In the HCC study, the incidence of cardiac ischemia/infarction was 2.7% in NEXAVAR patients compared with 1.3% in the placebo group and in RCC Study 1, the incidence of cardiac ischemia/infarction was higher in the NEXAVAR group (2.9%) compared with the placebo group (0.4%). Patients with unstable coronary artery disease or recent myocardial infarction were excluded from this study. Temporary or permanent discontinuation of NEXAVAR should be considered in patients who develop cardiac ischemia and/or infarction.
5.2 Risk of Hemorrhage
An increased risk of bleeding may occur following NEXAVAR administration. In the HCC study, an excess of bleeding regardless of causality was not apparent and the rate of bleeding from esophageal varices was 2.4% in NEXAVAR patients and 4% in placebo patients. Bleeding with a fatal outcome from any site was reported in 2.4% of NEXAVAR patients and 4% in placebo patients. In RCC Study 1, bleeding regardless of causality was reported in 15.3% of patients in the NEXAVAR group and 8.2% of patients in the placebo group. The incidence of CTCAE Grade 3 and 4 bleeding was 2% and 0%, respectively, in NEXAVAR patients, and 1.3% and 0.2%, respectively, in placebo patients. There was one fatal hemorrhage in each treatment group in RCC Study 1. If any bleeding necessitates medical intervention, permanent discontinuation of NEXAVAR should be considered.
5.3 Risk of Hypertension
Blood pressure should be monitored weekly during the first 6 weeks of NEXAVAR therapy and thereafter monitored and treated, if required, in accordance with standard medical practice. In the HCC study, hypertension was reported in approximately 9.4% of NEXAVAR-treated patients and 4.3% of patients in the placebo group. In RCC Study 1, hypertension was reported in approximately 16.9% of NEXAVAR-treated patients and 1.8% of patients in the placebo group. Hypertension was usually mild to moderate, occurred early in the course of treatment, and was managed with standard antihypertensive therapy. In cases of severe or persistent hypertension, despite institution of antihypertensive therapy, temporary or permanent discontinuation of NEXAVAR should be considered. Permanent discontinuation due to hypertension occurred in 1 of 297 NEXAVAR patients in the HCC study and 1 of 451 NEXAVAR patients in RCC Study 1.
5.4 Risk of Dermatologic Toxicities
Hand-foot skin reaction and rash represent the most common adverse reactions attributed to NEXAVAR. Rash and hand-foot skin reaction are usually CTCAE Grade 1 and 2 and generally appear during the first six weeks of treatment with NEXAVAR. Management of dermatologic toxicities may include topical therapies for symptomatic relief, temporary treatment interruption and/or dose modification of NEXAVAR, or in severe or persistent cases, permanent discontinuation of NEXAVAR. Permanent discontinuation of therapy due to hand-foot skin reaction occurred in 4 of 297 NEXAVAR HCC patients and 3 of 451 NEXAVAR RCC patients.
5.5 Risk of Gastrointestinal Perforation
Gastrointestinal perforation is an uncommon adverse reaction and has been reported in less than 1% of patients taking NEXAVAR. In some cases this was not associated with apparent intra-abdominal tumor. In the event of a gastrointestinal perforation, NEXAVAR therapy should be discontinued.
5.6 Warfarin Co-Administration
Infrequent bleeding or elevations in the International Normalized Ratio (INR) have been reported in some patients taking warfarin while on NEXAVAR therapy. Patients taking concomitant warfarin should be monitored regularly for changes in prothrombin time, INR or clinical bleeding episodes.
5.7 Wound Healing Complications
No formal studies of the effect of NEXAVAR on wound healing have been conducted. Temporary interruption of NEXAVAR therapy is recommended in patients undergoing major surgical procedures. There is limited clinical experience regarding the timing of reinitiation of NEXAVAR therapy following major surgical intervention. Therefore, the decision to resume NEXAVAR therapy following a major surgical intervention should be based on clinical judgment of adequate wound healing.
5.8 Interactions with UGT1A1 Substrates
Sorafenib can cause increases in plasma concentrations of drugs that are substrates of UGT1A1. Caution is recommended when administering NEXAVAR with compounds that are metabolized/eliminated predominantly by the UGT1A1 pathway (e.g. irinotecan) [see Drug Interactions (7.1)].
5.9 Interaction with Docetaxel
Sorafenib can cause increases in plasma concentrations of docetaxel. Caution is recommended when NEXAVAR is co-administered with docetaxel [see Drug Interactions (7.2)].
5.10 Interaction with Doxorubicin
Sorafenib can cause increases in plasma concentrations of doxorubicin. Caution is recommended when NEXAVAR is co-administered with doxorubicin [see Drug Interactions (7.3)].
5.11 Pregnancy
Pregnancy Category D
Sorafenib may cause fetal harm when administered to a pregnant woman. In rats and rabbits, sorafenib has been shown to be teratogenic and to induce embryo-fetal toxicity (including increased post-implantation loss, resorptions, skeletal retardations, and retarded fetal weight). The effects occurred at doses considerably below the recommended human dose of 400 mg twice daily (approximately 500 mg/m2/day on a body surface area basis). Adverse intrauterine development effects were seen at doses ≥1.2 mg/m2/day in rats and 3.6 mg/m2/day in rabbits (approximately 0.008 times the AUC seen in cancer patients at the recommended human dose). A NOAEL (no observed adverse effect level) was not defined for either species, since lower doses were not tested.
There are no adequate and well-controlled studies in pregnant women using NEXAVAR. Women of childbearing potential should be advised to avoid becoming pregnant while on NEXAVAR. If this drug is used during pregnancy, or if the patient becomes pregnant while taking this drug, the patient should be apprised of the potential hazard to the fetus.
5.12 Hepatic Impairment
Hepatic impairment may reduce plasma concentrations of sorafenib. Comparison of data across studies suggests that sorafenib levels are lower in HCC patients than in non-HCC patients (without hepatic impairment). The AUC of sorafenib is similar between HCC patients with mild (Child-Pugh A) and moderate (Child-Pugh B) hepatic impairment. The optimal dose in non-HCC patients with hepatic impairment is not established [see Use in Specific Populations (8.6) and Clinical Pharmacology (12.3)].
6. ADVERSE REACTIONS
The following risks are discussed in greater detail in the WARNINGS AND PRECAUTIONS section (5):
- Cardiac ischemia, infarction [see Warnings and Precautions (5.1)]
- Hemorrhage [see Warnings and Precautions (5.2)]
- Hypertension [see Warnings and Precautions (5.3)]
- Hand-foot skin reaction and rash [see Warnings and Precautions (5.4)]
- Gastrointestinal perforation [see Warnings and Precautions (5.5)]
- Wound healing complications [see Warnings and Precautions (5.7)]
- Teratogenicity and embryofetal toxicity [see Warnings and Precautions (5.11)]
The data described in sections 6.1 and 6.2 reflect exposure to NEXAVAR in 748 patients who participated in placebo controlled studies in hepatocellular carcinoma (N=297) or advanced renal cell carcinoma (N=451).
The most common adverse reactions (≥20%), which were considered to be related to NEXAVAR, in patients with HCC or RCC are fatigue, weight loss, rash/desquamation, hand-foot skin reaction, alopecia, diarrhea, anorexia, nausea and abdominal pain.
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
6.1 Adverse Reactions in HCC Study
Table 2 shows the percentage of HCC patients experiencing adverse reactions that were reported in at least 10% of patients and at a higher rate in the NEXAVAR arm than the placebo arm. CTCAE Grade 3 adverse reactions were reported in 39% of patients receiving NEXAVAR compared to 24% of patients receiving placebo. CTCAE Grade 4 adverse reactions were reported in 6% of patients receiving NEXAVAR compared to 8% of patients receiving placebo.
| NEXAVAR | Placebo | |||||
| N=297 | N=302 | |||||
|
Adverse Reaction NCI- CTCAE v3 Category/Term |
All Grades % |
Grade 3 % |
Grade 4 % |
All Grades % |
Grade 3 % |
Grade 4 % |
| Any Adverse Reaction | 98 | 39 | 6 | 96 | 24 | 8 |
| Constitutional symptoms | ||||||
| Fatigue | 46 | 9 | 1 | 45 | 12 | 2 |
| Weight loss | 30 | 2 | 0 | 10 | 1 | 0 |
| Dermatology/ Skin | ||||||
| Rash/ desquamation | 19 | 1 | 0 | 14 | 0 | 0 |
| Pruritus | 14 | <1 | 0 | 11 | <1 | 0 |
| Hand-foot skin reaction | 21 | 8 | 0 | 3 | <1 | 0 |
| Dry skin | 10 | 0 | 0 | 6 | 0 | 0 |
| Alopecia | 14 | 0 | 0 | 2 | 0 | 0 |
| Gastrointestinal | ||||||
| Diarrhea | 55 | 10 | <1 | 25 | 2 | 0 |
| Anorexia | 29 | 3 | 0 | 18 | 3 | <1 |
| Nausea | 24 | 1 | 0 | 20 | 3 | 0 |
| Vomiting | 15 | 2 | 0 | 11 | 2 | 0 |
| Constipation | 14 | 0 | 0 | 10 | 0 | 0 |
| Hepatobiliary/ pancreas | ||||||
| Liver dysfunction | 11 | 2 | 1 | 8 | 2 | 1 |
| Pain | ||||||
| Pain, abdomen | 31 | 9 | 0 | 26 | 5 | 1 |
Hypertension was reported in 9% of patients treated with NEXAVAR and 4% of those treated with placebo. CTCAE Grade 3 hypertension was reported in 4% of NEXAVAR treated patients and 1% of placebo treated patients. No patients were reported with CTCAE Grade 4 reactions in either treatment group.
Hemorrhage/bleeding was reported in 18% of those receiving NEXAVAR and 20% of placebo patients. The rates of CTCAE Grade 3 and 4 bleeding were also higher in the placebo group (CTCAE Grade 3 - 3% NEXAVAR and 5% placebo and CTCAE Grade 4 - 2% NEXAVAR and 4% placebo). Bleeding from esophageal varices was reported in 2.4% in NEXAVAR treated patients and 4% of placebo treated patients.
Renal failure was reported in <1% of patients treated with NEXAVAR and 3% of placebo treated patients.
The rate of adverse reactions (including those associated with progressive disease) resulting in permanent discontinuation was similar in both the NEXAVAR and placebo groups (32% of NEXAVAR patients and 35% of placebo patients).
Laboratory Abnormalities
The following laboratory abnormalities were observed in HCC patients:
Hypophosphatemia was a common laboratory finding, observed in 35% of NEXAVAR-treated patients compared to 11% of placebo patients; CTCAE Grade 3 hypophosphatemia (1–2 mg/dL) occurred in 11% of NEXAVAR-treated patients and 2% of patients in the placebo group; there was 1 case of CTCAE Grade 4 hypophosphatemia (<1 mg/dL) reported in the placebo group. The etiology of hypophosphatemia associated with NEXAVAR is not known.
Elevated lipase was observed in 40% of patients treated with NEXAVAR compared to 37% of patients in the placebo group. CTCAE Grade 3 or 4 lipase elevations occurred in 9% of patients in each group. Elevated amylase was observed in 34% of patients treated with NEXAVAR compared to 29% of patients in the placebo group. CTCAE Grade 3 or 4 amylase elevations were reported in 2% of patients in each group. Many of the lipase and amylase elevations were transient, and in the majority of cases NEXAVAR treatment was not interrupted. Clinical pancreatitis was reported in 1 of 297 NEXAVAR-treated patients (CTCAE Grade 2).
Elevations in liver function tests were comparable between the 2 arms of the study. Hypoalbuminemia was observed in 59% of NEXAVAR-treated patients and 47% of placebo patients; no CTCAE Grade 3 or 4 hypoalbuminemia was observed in either group.
INR elevations were observed in 42% of NEXAVAR-treated patients and 34% of placebo patients; CTCAE Grade 3 INR elevations were reported in 4% of NEXAVAR-treated patients and 2% of placebo patients; there was no CTCAE Grade 4 INR elevation in either group.
Lymphopenia was observed in 47% of NEXAVAR-treated patients and 42% of placebo patients.
Thrombocytopenia was observed in 46% of NEXAVAR-treated patients and 41% of placebo patients; CTCAE Grade 3 or 4 thrombocytopenia was reported in 4% of NEXAVAR-treated patients and less than 1% of placebo patients.
6.2 Adverse Reactions in RCC Study 1
Table 3 shows the percentage of RCC patients experiencing adverse reactions that were reported in at least 10% of patients and at a higher rate in the NEXAVAR arm than the placebo arm. CTCAE Grade 3 adverse reactions were reported in 31% of patients receiving NEXAVAR compared to 22% of patients receiving placebo. CTCAE Grade 4 adverse reactions were reported in 7% of patients receiving NEXAVAR compared to 6% of patients receiving placebo.
|
NEXAVAR N=451 |
Placebo N=451 |
|||||
|
Adverse Reactions NCI- CTCAE v3 Category/Term |
All Grades % |
Grade 3 % |
Grade 4 % |
All Grades % |
Grade 3 % |
Grade 4 % |
| Any Adverse Reactions | 95 | 31 | 7 | 86 | 22 | 6 |
| Cardiovascular, General | ||||||
| Hypertension | 17 | 3 | <1 | 2 | <1 | 0 |
| Constitutional symptoms | ||||||
| Fatigue | 37 | 5 | <1 | 28 | 3 | <1 |
| Weight loss | 10 | <1 | 0 | 6 | 0 | 0 |
| Dermatology/skin | ||||||
| Rash/desquamation | 40 | <1 | 0 | 16 | <1 | 0 |
| Hand-foot skin reaction | 30 | 6 | 0 | 7 | 0 | 0 |
| Alopecia | 27 | <1 | 0 | 3 | 0 | 0 |
| Pruritus | 19 | <1 | 0 | 6 | 0 | 0 |
| Dry skin | 11 | 0 | 0 | 4 | 0 | 0 |
| Gastrointestinal symptoms | ||||||
| Diarrhea | 43 | 2 | 0 | 13 | <1 | 0 |
| Nausea | 23 | <1 | 0 | 19 | <1 | 0 |
| Anorexia | 16 | <1 | 0 | 13 | 1 | 0 |
| Vomiting | 16 | <1 | 0 | 12 | 1 | 0 |
| Constipation | 15 | <1 | 0 | 11 | <1 | 0 |
| Hemorrhage/bleeding | ||||||
| Hemorrhage – all sites | 15 | 2 | 0 | 8 | 1 | <1 |
| Neurology | ||||||
| Neuropathy-sensory | 13 | <1 | 0 | 6 | <1 | 0 |
| Pain | ||||||
| Pain, abdomen | 11 | 2 | 0 | 9 | 2 | 0 |
| Pain, joint | 10 | 2 | 0 | 6 | <1 | 0 |
| Pain, headache | 10 | <1 | 0 | 6 | <1 | 0 |
| Pulmonary | ||||||
| Dyspnea | 14 | 3 | <1 | 12 | 2 | <1 |
The rate of adverse reactions (including those associated with progressive disease) resulting in permanent discontinuation was similar in both the NEXAVAR and placebo groups (10% of NEXAVAR patients and 8% of placebo patients).
Laboratory Abnormalities
The following laboratory abnormalities were observed in RCC patients in Study 1:
Hypophosphatemia was a common laboratory finding, observed in 45% of NEXAVAR-treated patients compared to 11% of placebo patients. CTCAE Grade 3 hypophosphatemia (1–2 mg/dL) occurred in 13% of NEXAVAR-treated patients and 3% of patients in the placebo group. There were no cases of CTCAE Grade 4 hypophosphatemia (<1 mg/dL) reported in either NEXAVAR or placebo patients. The etiology of hypophosphatemia associated with NEXAVAR is not known.
Elevated lipase was observed in 41% of patients treated with NEXAVAR compared to 30% of patients in the placebo group. CTCAE Grade 3 or 4 lipase elevations occurred in 12% of patients in the NEXAVAR group compared to 7% of patients in the placebo group. Elevated amylase was observed in 30% of patients treated with NEXAVAR compared to 23% of patients in the placebo group. CTCAE Grade 3 or 4 amylase elevations were reported in 1% of patients in the NEXAVAR group compared to 3% of patients in the placebo group. Many of the lipase and amylase elevations were transient, and in the majority of cases NEXAVAR treatment was not interrupted. Clinical pancreatitis was reported in 3 of 451 NEXAVAR-treated patients (one CTCAE Grade 2 and two Grade 4) and 1 of 451 patients (CTCAE Grade 2) in the placebo group.
Lymphopenia was observed in 23% of NEXAVAR-treated patients and 13% of placebo patients. CTCAE Grade 3 or 4 lymphopenia was reported in 13% of NEXAVAR-treated patients and 7% of placebo patients. Neutropenia was observed in 18% of NEXAVAR-treated patients and 10% of placebo patients. CTCAE Grade 3 or 4 neutropenia was reported in 5% of NEXAVAR-treated patients and 2% of placebo patients.
Anemia was observed in 44% of NEXAVAR-treated patients and 49% of placebo patients. CTCAE Grade 3 or 4 anemia was reported in 2% of NEXAVAR-treated patients and 4% of placebo patients.
Thrombocytopenia was observed in 12% of NEXAVAR-treated patients and 5% of placebo patients. CTCAE Grade 3 or 4 thrombocytopenia was reported in 1% of NEXAVAR-treated patients and 0% of placebo patients.
6.3 Additional Data from Multiple Clinical Trials
The following additional drug-related adverse reactions and laboratory abnormalities were reported from clinical trials of NEXAVAR as monotherapy (very common 10% or greater, common 1 to less than 10%, uncommon 0.1% to less than 1%):
Cardiovascular:Uncommon: hypertensive crisis*, myocardial ischemia and/or infarction*, congestive heart failure*
Dermatologic:Very common: erythema Common: exfoliative dermatitis, acne, flushing Uncommon: folliculitis, eczema, erythema multiforme, keratoacanthomas/squamous cell cancer of the skin
Digestive:Very common: increased lipase, increased amylase Common: mucositis, stomatitis (including dry mouth and glossodynia), dyspepsia, dysphagia Uncommon: pancreatitis, gastrointestinal reflux, gastritis, gastrointestinal perforations*
Note that elevations in lipase are very common (41%, see below); a diagnosis of pancreatitis should not be made solely on the basis of abnormal laboratory values
General Disorders:Very common: hemorrhage (including gastrointestinal* & respiratory tract* and uncommon cases of cerebral hemorrhage*), asthenia, pain (including mouth, bone, and tumor pain) Common: decreased appetite, influenza-like illness, pyrexia Uncommon: infection
Hematologic:Very common: leukopenia, lymphopenia Common: anemia, neutropenia, thrombocytopenia Uncommon: INR abnormal
Hypersensitivity:Uncommon: hypersensitivity reactions (including skin reactions and urticaria)
Metabolic and Nutritional:Very common: hypophosphatemia Common: transient increases in transaminases Uncommon: dehydration, hyponatremia, transient increases in alkaline phosphatase, increased bilirubin (including jaundice), hypothyroidism, Cholecystitis, cholangitis
Musculoskeletal:Common: arthralgia, myalgia
Nervous System and Psychiatric:Common: depression Uncommon: tinnitus, reversible posterior leukoencephalopathy*
Renal and Genitourinary: Common : renal failure
Reproductive:Common: erectile dysfunction Uncommon: gynecomastia
Respiratory:Common: hoarseness Uncommon: rhinorrhea
*adverse reactions may have a life-threatening or fatal outcome.
In addition, the following medically significant adverse reactions were uncommon during clinical trials of NEXAVAR: transient ischemic attack, arrhythmia, thromboembolism, acute renal failure. For these adverse reactions, the causal relationship to NEXAVAR has not been established.
7. DRUG INTERACTIONS
7.1 UGT1A1 and UGT1A9 Substrates
Caution is recommended when administering NEXAVAR with compounds that are metabolized/eliminated predominantly by the UGT1A1 pathway (e.g. irinotecan). Sorafenib inhibits glucuronidation by the UGT1A1 (Ki value: 1 µM) and UGT1A9 pathways (Ki value: 2 µM). Systemic exposure to substrates of UGT1A1 and UGT1A9 may increase when co-administered with NEXAVAR [see Warnings and Precautions (5.8)].
In clinical studies, when NEXAVAR was administered with irinotecan, whose active metabolite SN-38 is further metabolized by the UGT1A1 pathway, there was a 67-120% increase in the AUC of SN-38 and a 26-42% increase in the AUC of irinotecan. The clinical significance of these findings is unknown.
7.2 Docetaxel
Concomitant use of docetaxel (75 or 100 mg/m2 administered every 21 days) with NEXAVAR (200 or 400 mg twice daily), administered with a 3-day break in dosing around administration of docetaxel, resulted in a 36-80% increase in docetaxel AUC and a 16-32% increase in docetaxel Cmax. Caution is recommended when NEXAVAR is co-administered with docetaxel [see Warnings and Precautions (5.9)].
7.3 Doxorubicin
Concomitant treatment with NEXAVAR resulted in a 21% increase in the AUC of doxorubicin. Caution is recommended when administering doxorubicin with NEXAVAR. The clinical significance of these findings is unknown [see Warnings and Precautions (5.10)].
7.4 Fluorouracil
Both increases (21%-47%) and decreases (10%) in the AUC of fluorouracil were observed with concomitant treatment with NEXAVAR. Caution is recommended when NEXAVAR is co-administered with fluorouracil/leucovorin.
7.5 CYP2B6 and CYP2C8 Substrates
Sorafenib inhibits CYP2B6 and CYP2C8 in vitro with Ki values of 6 and 1-2 µM, respectively. Systemic exposure to substrates of CYP2B6 and CYP2C8 is expected to increase when co-administered with NEXAVAR. Caution is recommended when administering substrates of CYP2B6 and CYP2C8 with NEXAVAR.
7.6 CYP3A4 Inducers
Continuous concomitant administration of NEXAVAR and rifampicin resulted in an average 37% reduction of sorafenib AUC. Other inducers of CYP3A4 activity (e.g. Hypericum perforatum also known as St. John’s wort, phenytoin, carbamazepine, phenobarbital, and dexamethasone) may also increase metabolism of sorafenib and thus decrease sorafenib concentrations [see Dosage and Administration (2)].
7.7 CYP3A4 Inhibitors and CYP Isoform Substrates
In vitro data indicate that sorafenib is metabolized by CYP3A4 and UGT1A9 pathways. Ketoconazole (400 mg), a potent inhibitor of CYP3A4, administered once daily for 7 days did not alter the mean AUC of a single oral 50 mg dose of sorafenib in healthy volunteers. Therefore, sorafenib metabolism is unlikely to be altered by CYP3A4 inhibitors.
Studies with human liver microsomes demonstrated that sorafenib is a competitive inhibitor of CYP2C19, CYP2D6, and CYP3A4 as indicated by Ki values of 17 µM, 22 µM, and 29 µM, respectively. Administration of NEXAVAR 400 mg twice daily for 28 days did not alter the exposure of concomitantly administered midazolam (CYP3A4 substrate), dextromethorphan (CYP2D6 substrate), and omeprazole (CYP2C19 substrate). This indicates that sorafenib is unlikely to alter the metabolism of substrates of these enzymes in vivo.
Studies with human liver microsomes demonstrated that sorafenib is a competitive inhibitor of CYP2C9 with a Ki value of 7-8 µM. The possible effect of sorafenib on the metabolism of the CYP2C9 substrate warfarin was assessed indirectly by measuring PT-INR. The mean changes from baseline in PT-INR were not higher in NEXAVAR patients compared to placebo patients, suggesting that sorafenib did not inhibit warfarin metabolism in vivo[see Warnings and Precautions (5.6)].
7.8 In Vitro Studies: CYP Enzyme Induction
CYP1A2 and CYP3A4 activities were not altered after treatment of cultured human hepatocytes with sorafenib, indicating that sorafenib is unlikely to be an inducer of CYP1A2 or CYP3A4.
7.9 Combination with Other Antineoplastic Agents
In clinical studies, NEXAVAR has been administered with a variety of other antineoplastic agents at their commonly used dosing regimens, including gemcitabine, oxaliplatin, paclitaxel and carboplatin, doxorubicin, docetaxel, and irinotecan. Sorafenib had no effect on the pharmacokinetics of gemcitabine or oxaliplatin. Concomitant use of paclitaxel (225 mg/m2) and carboplatin (AUC=6) with NEXAVAR (100, 200 or 400 mg twice daily), administered with a 3-day break in dosing around administration of paclitaxel/carboplatin, resulted in no significant effect on the pharmacokinetics of paclitaxel. See Drug Interactions (7.1, 7.2, 7.3 and 7.4 ) for information about interactions with irinotecan, docetaxel, doxorubicin and fluorouracil/leucovorin
8. USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Category D
[see Warnings and Precautions (5.11)].
8.3 Nursing Mothers
It is not known whether sorafenib is excreted in human milk. Because many drugs are excreted in human milk and because of the potential for serious adverse reactions in nursing infants from NEXAVAR, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Following administration of radiolabeled sorafenib to lactating Wistar rats, approximately 27% of the radioactivity was secreted into the milk. The milk to plasma AUC ratio was approximately 5:1.
8.4 Pediatric Use
The safety and effectiveness of NEXAVAR in pediatric patients have not been studied.
Repeat dosing of sorafenib to young and growing dogs resulted in irregular thickening of the femoral growth plate at daily sorafenib doses ≥ 600 mg/m2 (approximately 0.3 times the AUC at the recommended human dose), hypocellularity of the bone marrow adjoining the growth plate at 200 mg/m2/day (approximately 0.1 times the AUC at the recommended human dose), and alterations of the dentin composition at 600 mg/m2/day. Similar effects were not observed in adult dogs when dosed for 4 weeks or less.
8.5 Geriatric Use
In total, 59% of HCC patients treated with NEXAVAR were age 65 years or older, and 19% were 75 and older. In total, 32% of RCC patients treated with NEXAVAR were age 65 years or older, and 4% were 75 and older. No differences in safety or efficacy were observed between older and younger patients, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
8.6 Patients with Hepatic Impairment
In vitro and in vivo data indicate that sorafenib is primarily metabolized by the liver. Comparison of data across studies suggests that patients with mild (Child-Pugh A) and moderate (Child-Pugh B) hepatic impairment have sorafenib AUCs that may be 23- 65% lower than subjects with normal hepatic function. Systemic exposure and safety data were comparable in HCC patients with Child-Pugh A and B hepatic impairment. NEXAVAR has not been studied in patients with Child-Pugh C hepatic impairment [see Warnings and Precautions (5.12) and Clinical Pharmacology (12.3)].
8.7 Patients with Renal Impairment
NEXAVAR has not been studied in patients undergoing dialysis. No dosage adjustment is necessary when administering NEXAVAR to patients with mild, moderate or severe renal impairment not undergoing dialysis. Monitoring of fluid balance and electrolytes in patients at risk with renal dysfunction is advised [see Clinical Pharmacology (12.3)].
10. OVERDOSAGE
There is no specific treatment for NEXAVAR overdose.
The highest dose of NEXAVAR studied clinically is 800 mg twice daily. The adverse reactions observed at this dose were primarily diarrhea and dermatologic. No information is available on symptoms of acute overdose in animals because of the saturation of absorption in oral acute toxicity studies conducted in animals.
In cases of suspected overdose, NEXAVAR should be withheld and supportive care instituted.
11. DESCRIPTION
NEXAVAR, a kinase inhibitor, is the tosylate salt of sorafenib.
Sorafenib tosylate has the chemical name 4-(4-{3-[4-Chloro-3-(trifluoromethyl)phenyl]ureido}phenoxy)N2-methylpyridine-2-carboxamide 4-methylbenzenesulfonate and its structural formula is:
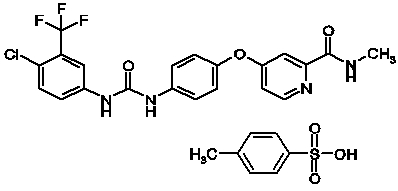
Sorafenib tosylate is a white to yellowish or brownish solid with a molecular formula of C21H16ClF3N4O3 x C7H8O3S and a molecular weight of 637.0 g/mole. Sorafenib tosylate is practically insoluble in aqueous media, slightly soluble in ethanol and soluble in PEG 400.
Each red, round NEXAVAR film-coated tablet contains sorafenib tosylate (274 mg) equivalent to 200 mg of sorafenib and the following inactive ingredients: croscarmellose sodium, microcrystalline cellulose, hypromellose, sodium lauryl sulphate, magnesium stearate, polyethylene glycol, titanium dioxide and ferric oxide red.
12. CLINICAL PHARMACOLOGY
12.1 Mechanism Of Action
Sorafenib is a kinase inhibitor that decreases tumor cell proliferation in vitro. Sorafenib was shown to inhibit multiple intracellular (CRAF, BRAF and mutant BRAF) and cell surface kinases (KIT, FLT-3, RET, VEGFR-1, VEGFR-2, VEGFR-3, and PDGFR-ß). Several of these kinases are thought to be involved in tumor cell signaling, angiogenesis, and apoptosis. Sorafenib inhibited tumor growth and angiogenesis of human hepatocellular carcinoma and renal cell carcinoma, and several other human tumor xenografts in immunocompromised mice.
12.3 Pharmacokinetics
After administration of NEXAVAR tablets, the mean relative bioavailability is 38-49% when compared to an oral solution. The mean elimination half-life of sorafenib is approximately 25-48 hours. Multiple dosing of NEXAVAR for 7 days resulted in a 2.5- to 7-fold accumulation compared to single dose administration. Steady-state plasma sorafenib concentrations are achieved within 7 days, with a peak-to-trough ratio of mean concentrations of less than 2.
Absorption and Distribution
Following oral administration, sorafenib reaches peak plasma levels in approximately 3 hours. When given with a moderate-fat meal (30% fat; 700 calories), bioavailability was similar to that in the fasted state. With a high-fat meal (50% fat; 900 calories), sorafenib bioavailability was reduced by 29% compared to administration in the fasted state. It is recommended that NEXAVAR be administered without food [see DOSAGE AND ADMINISTRATION (2)].
Mean Cmax and AUC increased less than proportionally beyond doses of 400 mg administered orally twice daily.
In vitro binding of sorafenib to human plasma proteins is 99.5%.
Metabolism and Elimination
Sorafenib is metabolized primarily in the liver, undergoing oxidative metabolism, mediated by CYP3A4, as well as glucuronidation mediated by UGT1A9.
Sorafenib accounts for approximately 70-85% of the circulating analytes in plasma at steady-state. Eight metabolites of sorafenib have been identified, of which five have been detected in plasma. The main circulating metabolite of sorafenib in plasma, the pyridine N-oxide, shows in vitro potency similar to that of sorafenib. This metabolite comprises approximately 9-16% of circulating analytes at steady-state.
Following oral administration of a 100 mg dose of a solution formulation of sorafenib, 96% of the dose was recovered within 14 days, with 77% of the dose excreted in feces, and 19% of the dose excreted in urine as glucuronidated metabolites. Unchanged sorafenib, accounting for 51% of the dose, was found in feces but not in urine.
Special Populations
Age
Analyses of demographic data suggest that no dose adjustments are necessary for age.
Gender
Analyses of demographic data suggest that no dose adjustments are necessary for gender.
Race
A study of the pharmacokinetics of sorafenib indicated that the mean AUC of sorafenib in Asians (N=78) was 30% lower than in Caucasians (N=40).
Pediatric
There are no pharmacokinetic data in pediatric patients.
Hepatic Impairment
Sorafenib is cleared primarily by the liver.
Comparison of data across studies suggests that in HCC patients with mild (Child-Pugh A) or moderate (Child-Pugh B) hepatic impairment, 400 mg doses of sorafenib appear to be associated with AUC values that were 23 to 65% lower than those of other subjects without hepatic impairment. The AUC of sorafenib is similar between HCC patients with mild (Child-Pugh A) and moderate (Child-Pugh B) hepatic impairment. The pharmacokinetics of sorafenib have not been studied in patients with severe (Child-Pugh C) hepatic impairment [see Warnings and Precautions (5.12) and Use in Specific Populations (8.6)].
Renal Impairment
In a study of drug disposition after a single oral dose of radiolabeled sorafenib to healthy subjects, 19% of the administered dose of sorafenib was excreted in urine.
In a clinical pharmacology study, the pharmacokinetics of sorafenib were evaluated following administration of a single 400 mg dose to subjects with normal renal function, and in subjects with mild (CrCl > 50-80 ml/min), moderate (CrCl 30-50 ml/min), or severe (CrCl < 30 ml/min) renal impairment, not undergoing dialysis. There was no relationship observed between sorafenib exposure and renal function. No dosage adjustment is necessary based on mild, moderate or severe renal impairment not undergoing dialysis. Monitoring of fluid balance and electrolytes in patients with renal dysfunction is advised [see Use in Specific Populations (8.7)].
13. NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment Of Fertility
Carcinogenicity studies have not been performed with sorafenib.
Sorafenib was clastogenic when tested in an in vitro mammalian cell assay (Chinese hamster ovary) in the presence of metabolic activation. Sorafenib was not mutagenic in the in vitro Ames bacterial cell assay or clastogenic in an in vivo mouse micronucleus assay. One intermediate in the manufacturing process, which is also present in the final drug substance (<0.15%), was positive for mutagenesis in an in vitro bacterial cell assay (Ames test) when tested independently.
No specific studies with sorafenib have been conducted in animals to evaluate the effect on fertility. However, results from the repeat-dose toxicity studies suggest there is a potential for sorafenib to impair reproductive function and fertility. Multiple adverse effects were observed in male and female reproductive organs, with the rat being more susceptible than mice or dogs. Typical changes in rats consisted of testicular atrophy or degeneration, degeneration of epididymis, prostate, and seminal vesicles, central necrosis of the corpora lutea and arrested follicular development. Sorafenib-related effects on the reproductive organs of rats were manifested at daily oral doses ≥30 mg/m2 (approximately 0.5 times the AUC in cancer patients at the recommended human dose). Dogs showed tubular degeneration in the testes at 600 mg/m2/day (approximately 0.3 times the AUC at the recommended human dose) and oligospermia at 1200 mg/m2/day of sorafenib.
Adequate contraception should be used during therapy and for at least 2 weeks after completing therapy.
14. CLINICAL STUDIES
The clinical safety and efficacy of NEXAVAR have been studied in patients with hepatocellular carcinoma (HCC) and renal cell carcinoma (RCC).
14.1 Hepatocellular Carcinoma
The HCC Study was a Phase 3, international, multicenter, randomized, double blind, placebo-controlled trial in patients with unresectable hepatocellular carcinoma. Overall survival was the primary endpoint. A total of 602 patients were randomized; 299 to NEXAVAR 400 mg twice daily and 303 to matching placebo.
Demographics and baseline disease characteristics were similar between the NEXAVAR and placebo groups with regard to age, gender, race, performance status, etiology (including hepatitis B, hepatitis C and alcoholic liver disease), TNM stage (stage I: <1% vs. <1%; stage II: 10.4% vs. 8.3%; stage III: 37.8% vs. 43.6%; stage IV: 50.8% vs. 46.9%), absence of both macroscopic vascular invasion and extrahepatic tumor spread (30.1% vs. 30.0%), and Barcelona Clinic Liver Cancer stage (stage B: 18.1% vs. 16.8%; stage C: 81.6% vs. 83.2%; stage D: <1% vs. 0%). Liver impairment by Child-Pugh score was comparable between the NEXAVAR and placebo groups
(Class A: 95% vs. 98%; B: 5% vs. 2%). Only one patient with Child-Pugh class C was entered. Prior treatments included surgical resection procedures (19.1% vs. 20.5%), locoregional therapies (including radiofrequency ablation, percutaneous ethanol injection and transarterial chemoembolization; 38.8% vs. 40.6%), radiotherapy (4.3% vs. 5.0%) and systemic therapy (3.0% vs. 5.0%).
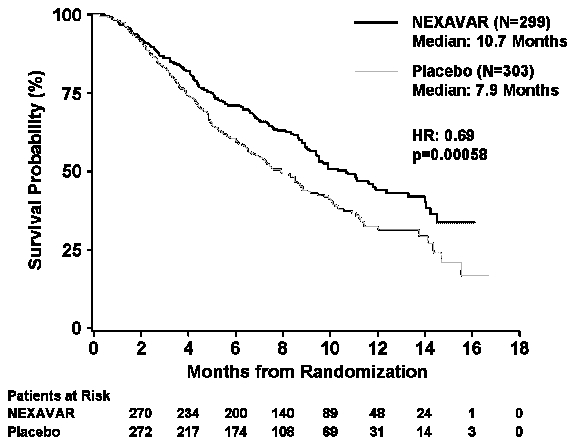
The trial was stopped for efficacy following a pre-specified second interim analysis for survival showing a statistically significant advantage for NEXAVAR over placebo for overall survival (HR: 0.69, p= 0.00058) (see Table 4 and Figure 1). This advantage was consistent across all subsets analyzed.
Final analysis of time to tumor progression (TTP) based on data from an earlier time point (by independent radiologic review) also was significantly longer in the NEXAVAR arm (HR: 0.58, p=0.000007) (see Table 4).
| CI=Confidence interval | ||||
|
||||
| Efficacy Parameter |
NEXAVAR (N=299) |
Placebo (N=303) | Hazard Ratio *(95% CI) | P-value (log-rank test †) |
|
Overall Survival Median, months (95% CI) No. of events |
10.7 (9.4, 13.3) 143 |
7.9 (6.8, 9.1) 178 |
0.69 (0.55, 0.87) | 0.00058 |
|
Time to Progression ‡ Median, months (95% CI) No. of events |
5.5 (4.1, 6.9) 107 |
2.8 (2.7, 3.9) 156 |
0.58 (0.45, 0.74) | 0.000007 |
14.2 Renal Cell Carcinoma
The safety and efficacy of NEXAVAR in the treatment of advanced renal cell carcinoma (RCC) were studied in the following two randomized controlled clinical trials.
RCC Study 1 was a Phase 3, international, multicenter, randomized, double blind, placebo-controlled trial in patients with advanced renal cell carcinoma who had received one prior systemic therapy. Primary study endpoints included overall survival and progression-free survival (PFS). Tumor response rate was a secondary endpoint. The PFS analysis included 769 patients stratified by MSKCC (Memorial Sloan Kettering Cancer Center)
prognostic risk category (low or intermediate) and country and randomized to NEXAVAR 400 mg twice daily
(N=384) or to placebo (N=385).
Table 5 summarizes the demographic and disease characteristics of the study population analyzed. Baseline demographics and disease characteristics were well balanced for both treatment groups. The median time from initial diagnosis of RCC to randomization was 1.6 and 1.9 years for the NEXAVAR and placebo groups, respectively.
|
||||
| Characteristics |
NEXAVAR N=384 |
Placebo N=385 |
||
| n | (%) | n | (%) | |
| Gender | ||||
| Male | 267 | (70) | 287 | (75) |
| Female | 116 | (30) | 98 | (25) |
| Race | ||||
| White | 276 | (72) | 278 | (73) |
| Black/Asian/ | ||||
| Hispanic/Other | 11 | (3) | 10 | (2) |
| Not reported* | 97 | (25) | 97 | (25) |
| Age group | ||||
| < 65 years | 255 | (67) | 280 | (73) |
| ≥ 65 years | 127 | (33) | 103 | (27) |
| ECOG performance status at baseline | ||||
| 0 | 184 | (48) | 180 | (47) |
| 1 | 191 | (50) | 201 | (52) |
| 2 | 6 | (2) | 1 | (<1) |
| Not reported | 3 | (<1) | 3 | (<1) |
| MSKCC prognostic risk category | ||||
| Low | 200 | (52) | 194 | (50) |
| Intermediate | 184 | (48) | 191 | (50) |
| Prior IL-2 and/or interferon | ||||
| Yes | 319 | (83) | 313 | (81) |
| No | 65 | (17) | 72 | (19) |
Progression-free survival, defined as the time from randomization to progression or death from any cause, whichever occurred earlier, was evaluated by blinded independent radiological review using RECIST criteria.
Figure 2 depicts Kaplan-Meier curves for PFS. The PFS analysis was based on a two-sided Log-Rank test stratified by MSKCC prognostic risk category and country.
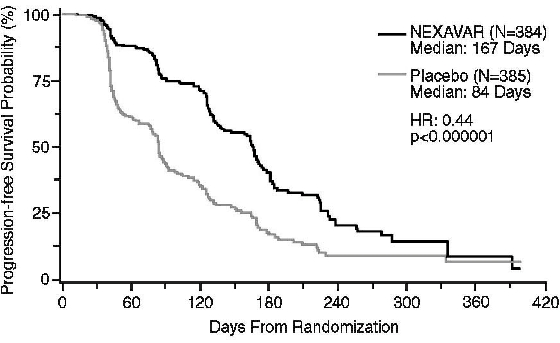
Figure 2: Kaplan-Meier Curves for Progression-free Survival - RCC Study 1
NOTE: HR is from Cox regression model with the following covariates: MSKCC prognostic risk category and country. P-value is from two-sided Log-Rank test stratified by MSKCC prognostic risk category and country.
The median PFS for patients randomized to NEXAVAR was 167 days compared to 84 days for patients randomized to placebo. The estimated hazard ratio (risk of progression with NEXAVAR compared to placebo) was 0.44 (95% CI: 0.35, 0.55).
A series of patient subsets were examined in exploratory univariate analyses of PFS. The subsets included age
above or below 65 years, ECOG PS 0 or 1, MSKCC prognostic risk category, whether the prior therapy was for
progressive metastatic disease or for an earlier disease setting, and time from diagnosis of less than or greater than 1.5 years. The effect of NEXAVAR on PFS was consistent across these subsets, including patients with no prior IL-2 or interferon therapy (N=137; 65 patients receiving NEXAVAR and 72 placebo), for whom the median PFS was 172 days on NEXAVAR compared to 85 days on placebo.
Tumor response was determined by independent radiologic review according to RECIST criteria. Overall, of 672 patients who were evaluable for response, 7 (2%) NEXAVAR patients and 0 (0%) placebo patients had a confirmed partial response. Thus the gain in PFS in NEXAVAR-treated patients primarily reflects the stable disease population.
At the time of a planned interim survival analysis, based on 220 deaths, overall survival was longer for NEXAVAR than placebo with a hazard ratio (NEXAVAR over placebo) of 0.72. This analysis did not meet the prespecified criteria for statistical significance. Additional analyses are planned as the survival data mature.
RCC Study 2 was a Phase 2 randomized discontinuation trial in patients with metastatic malignancies, including RCC. The primary endpoint was the percentage of randomized patients remaining progression-free at 24 weeks. All patients received NEXAVAR for the first 12 weeks. Radiologic assessment was repeated at week 12. Patients with <25% change in bi-dimensional tumor measurements from baseline were randomized to NEXAVAR or placebo for a further 12 weeks. Patients who were randomized to placebo were permitted to cross over to open-label NEXAVAR upon progression. Patients with tumor shrinkage ≥25% continued NEXAVAR, whereas patients with tumor growth ≥25% discontinued treatment.
Two hundred and two patients with advanced RCC were enrolled into RCC Study 2, including patients who had received no prior therapy and patients with tumor histology other than clear cell carcinoma. After the initial 12 weeks of NEXAVAR therapy, 79 RCC patients continued on open-label NEXAVAR, and 65 patients were randomized to NEXAVAR or placebo. After an additional 12 weeks, at week 24, for the 65 randomized patients, the progression-free rate was significantly higher in patients randomized to NEXAVAR (16/32, 50%) than in patients randomized to placebo (6/33, 18%) (p=0.0077). Progression-free survival was significantly longer in the NEXAVAR group (163 days) than in the placebo group (41 days) (p=0.0001, HR=0.29).
16. HOW SUPPLIED/STORAGE AND HANDLING
NEXAVAR tablets are supplied as round, biconvex, red film-coated tablets, debossed with the “Bayer cross” on one side and “200” on the other side, each containing sorafenib tosylate equivalent to 200 mg of sorafenib.
Bottles of 120 tablets NDC 0026-8488-58
Storage
Store at 25°C (77°F); excursions permitted to 15-30°C (59-86°F) (see USP controlled room temperature). Store in a dry place.
17. PATIENT COUNSELING INFORMATION
See FDA-approved Patient Labeling (17.5)
17.1 Cardiac Ischemia; Infarction
Physicians should also discuss with patients that cardiac ischemia and/or infarction has been reported during NEXAVAR treatment, and that they should immediately report any episodes of chest pain or other symptoms of cardiac ischemia and/or infarction [see Warnings and Precautions (5.1)]
17.2 Bleeding; Gastrointestinal Perforation
Physicians should inform patients that NEXAVAR may increase the risk of bleeding and that they should promptly report any episodes of bleeding.
Patients should be advised that cases of gastrointestinal perforation have been reported in patients taking NEXAVAR [see Warnings and Precautions (5.2 and 5.5)]
17.3 Skin Reactions; Hypertension
Patients should be advised of the possible occurrence of hand-foot skin reaction and rash during NEXAVAR treatment and appropriate countermeasures.
Patients should be informed that hypertension may develop during NEXAVAR treatment, especially during the first six weeks of therapy, and that blood pressure should be monitored regularly during treatment [see Warnings and Precautions (5.3 and 5.4)]
17.4 Birth Defects and Fetal Loss
Physicians should inform female patients that NEXAVAR may cause birth defects or fetal loss and that they should not become pregnant during treatment with NEXAVAR and for at least 2 weeks after stopping treatment. Both male and female patients should be counseled to use effective birth control during treatment with NEXAVAR and for at least 2 weeks after stopping treatment. Female patients should also be advised against breast-feeding while receiving NEXAVAR [see Warnings and Precautions (5.11)]
17.5 FDA-approved Patient Labeling
Patient Information:
NEXAVAR® (NEX-A-VAR)
(sorafenib) tablets, oral
Read the Patient Information that comes with NEXAVAR before you start taking it and each time you get a refill. There may be new information. This leaflet does not take the place of talking with your doctor about your medical condition or your treatment.
What is the most important information I should know about NEXAVAR?
NEXAVAR may cause birth defects or death of an unborn baby.
- Women should not get pregnant during treatment with NEXAVAR and for at least 2 weeks after stopping treatment.
- Men and women should use effective birth control during treatment with NEXAVAR and for at least 2 weeks after stopping treatment. Talk with your doctor about effective birth control methods.
Call your doctor right away if you become pregnant during treatment with NEXAVAR.
What is NEXAVAR?
NEXAVAR is an anticancer medicine used to treat a certain type of liver or kidney cancer called:
1. Hepatocellular carcinoma (HCC, a type of liver cancer), when it can not be treated with surgery
2. Renal cell carcinoma (RCC, a type of kidney cancer)
NEXAVAR has not been studied in children.
What should I tell my doctor before starting NEXAVAR?
Tell your doctor about all of your health conditions, including if you:
- have any allergies
- have heart problems or chest pain
- have bleeding problems
- have high blood pressure
- have kidney problems in addition to kidney cancer
- have liver problems in addition to liver cancer
- are pregnant or planning to become pregnant. See “What is the most important information I should know about NEXAVAR?”
- are breast-feeding or planning to breast-feed. It is not known if NEXAVAR passes into your breast milk. You and your doctor should decide if you will take NEXAVAR or breast-feed. You should not do both.
Tell your doctor about all the medicines you take, including prescription and non-prescription medicines, vitamins and herbal supplements. NEXAVAR and certain other medicines can interact with each other and cause serious side effects. Especially, tell your doctor if you take warfarin (Coumadin®)*.
Know the medicines you take. Keep a list of your medicines and show it to your doctor and pharmacist when you get a new medicine. Do not take other medicines with NEXAVAR until you have talked with your doctor.
How do I take NEXAVAR?
- Take NEXAVAR exactly as prescribed by your doctor.
- The usual dose of NEXAVAR is 2 tablets taken two times a day (for a total of 4 tablets each day). Your doctor may change your dose during treatment or stop treatment for some time if you have side effects.
- Swallow NEXAVAR tablets whole with water.
- Take NEXAVAR without food (at least 1 hour before or 2 hours after a meal).
- If you miss a dose of NEXAVAR, skip the missed dose, and take your next dose at your regular time. Do not double your dose of NEXAVAR. Call your doctor right away if you take too much NEXAVAR.
What are possible side effects of NEXAVAR?
NEXAVAR may cause serious side effects, including:
- decreased blood flow to the heart and heart attack. Get emergency help right away and call your doctor if you get symptoms such as chest pain, shortness of breath, feel lightheaded or faint, nausea, vomiting, sweating a lot.
- bleeding problems. NEXAVAR may increase your chance of bleeding. Tell your doctor if you have any bleeding while taking NEXAVAR.
- high blood pressure. Your blood pressure should be checked every week during the first 6 weeks of starting NEXAVAR. Your blood pressure should be checked regularly and any high blood pressure should be treated while you are receiving NEXAVAR.
- a skin problem called hand-foot skin reaction. This causes redness, pain, swelling, or blisters on the palms of your hands or soles of your feet. If you get this side effect, your doctor may change your dose or stop treatment for some time.
- perforation of the bowel. Tell your doctor right away if you get high fever, nausea, vomiting, severe abdominal pain.
- possible wound healing problems. If you need to have a surgical or dental procedure, tell your doctor that you are taking NEXAVAR. NEXAVAR may need to be stopped until your wound heals after some types of surgery.
- birth defects or death of an unborn baby. See “What is the most important information I should know about NEXAVAR?”
Other side effects with NEXAVAR may include:
- rash, redness, itching or peeling of your skin
- hair thinning or patchy hair loss
- diarrhea (frequent or loose bowel movements)
- nausea or vomiting
- mouth sores
- weakness
- loss of appetite
- numbness, tingling or pain in your hands and feet
- abdominal pain
- tiredness
- weight loss
Tell your doctor if you have any side effects that bother you or that do not go away. These are not all the side effects with NEXAVAR. Ask your doctor or pharmacist for more information.
How should I store NEXAVAR?
- Store NEXAVAR tablets at room temperature between 59° to 86° F (15° to 30° C), in a dry place.
- Keep NEXAVAR and all medicines out of the reach of children.
General information about NEXAVAR
Medicines are sometimes prescribed for purposes other than those listed in the patient information leaflet. Do not use NEXAVAR for a condition for which it is not prescribed. Do not give NEXAVAR to other people even if they have the same symptoms you have. It may harm them.
This patient information leaflet summarizes the most important information about NEXAVAR. If you would like more information, talk with your doctor. You can ask your doctor or pharmacist for information about NEXAVAR that is written for healthcare professionals. You can visit our website at www.NEXAVAR.com, or call 1-866-NEXAVAR (1-866-639-2827).
What are the ingredients in NEXAVAR?
Active Ingredient: sorafenib tosylate
Inactive Ingredients: croscarmellose sodium, microcrystalline cellulose, hypromellose, sodium lauryl sulphate, magnesium stearate, polyethylene glycol, titanium dioxide and ferric oxide red.
*Coumadin (warfarin sodium) is a trademark of Bristol-Myers Squibb Company
Manufactured by:
Bayer HealthCare AG
Leverkusen, Germany
Manufactured for:
Bayer HealthCare Pharmaceuticals Inc.
Wayne, NJ 07470
Onyx Pharmaceuticals, Inc.,
2100 Powell Street, Emeryville, CA 94608
Distributed and marketed by:
Bayer HealthCare Pharmaceuticals Inc.
Wayne, NJ 07470
Marketed by:
Onyx Pharmaceuticals, Inc.,
2100 Powell Street, Emeryville, CA 94608
80771372, R.2 013632
©2008 Bayer HealthCare Pharmaceuticals Inc. Printed in U.S.A.
| NEXAVAR
sorafenib tablet, film coated |
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
Revised: 10/2008