DESOXIMETASONE
-
desoximetasone gel
DESOXIMETASONE
-
desoximetasone cream
DESOXIMETASONE
-
desoximetasone ointment
Taro Pharmaceuticals U.S.A., Inc.
----------
Desoximetasone
Cream USP, 0.05%, Cream USP, 0.25%
Gel USP, 0.05%, Ointment USP, 0.25%
For external use only. Not for ophthalmic use.
Rx only
DESCRIPTION
Desoximetasone Cream USP, 0.05%; Desoximetasone Cream USP, 0.25%; Desoximetasone Gel USP, 0.05% and Desoximetasone Ointment USP, 0.25% contain desoximetasone a synthetic fluorinated corticosteroid for topical dermatologic use. The corticosteroids constitute a class of primarily synthetic steroids used topically as anti-inflammatory and anti-pruritic agents.
Chemically, desoximetasone is Pregna-1, 4-diene-3, 20-dione, 9-fluoro-11,21-dihydroxy-16-methyl-, (11β, 16α)-. It has the molecular formula C22H29FO4 and a molecular weight of 376.47. The CAS Registry Number is 382-67-2. The chemical structure is:
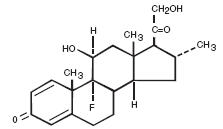
Desoximetasone is a white to off-white, odorless, crystalline powder and is soluble in alcohol, acetone, chloroform, and hot ethyl acetate; slightly soluble in ether and benzene; and insoluble in water, dilute aqueous acids and alkalis.
Each gram of Desoximetasone Cream USP, 0.05% contains 0.5 mg of desoximetasone in an emollient cream consisting of white petrolatum, purified water, isopropyl myristate, lanolin alcohols, mineral oil, cetostearyl alcohol and edetate disodium.
Each gram of Desoximetasone Cream USP, 0.25% contains 2.5 mg of desoximetasone in an emollient cream consisting of white petrolatum, purified water, isopropyl myristate, lanolin alcohols, mineral oil and cetostearyl alcohol.
Each gram of Desoximetasone Gel USP, 0.05% contains 0.5 mg of desoximetasone in a gel base consisting of purified water, docusate sodium, edetate disodium, isopropyl myristate, carbomer 940, trolamine, and SDAG-3 95% alcohol.
Each gram of Desoximetasone Ointment USP, 0.25% contains 2.5 mg of desoximetasone in an ointment base consisting of white petrolatum and fractionated coconut oil.
CLINICAL PHARMACOLOGY
The corticosteroids are a class of compounds comprising steroid hormones secreted by the adrenal cortex and their synthetic analogs. In pharmacologic doses, corticosteroids are used primarily for their anti-inflammatory and/or immunosuppressive effects.
Topical corticosteroids, such as desoximetasone, are effective in the treatment of corticosteroid-responsive dermatoses primarily because of their anti-inflammatory, antipruritic, and vasoconstrictive actions. However, while the physiologic, pharmacologic, and clinical effects of the corticosteroids are well known, the exact mechanisms of their actions in each disease are uncertain. Desoximetasone, a corticosteroid, has been shown to have topical (dermatologic) and systemic pharmacologic and metabolic effects characteristic of this class of drugs.
Pharmacokinetics
The extent of percutaneous absorption of topical corticosteroids is determined by many factors including the vehicle and the integrity of the epidermal barrier, and the use of occlusive dressings.
Topical corticosteroids can be absorbed through normal intact skin. Inflammation and/or other disease processes in the skin may increase percutaneous absorption. Occlusive dressings substantially increase the percutaneous absorption of topical corticosteroids.
Once absorbed through the skin, topical corticosteroids enter pharmacokinetic pathways similar to systemically administered corticosteroids. Corticosteroids are bound to plasma proteins in varying degrees, are metabolized primarily in the liver and excreted by the kidneys. Some of the topical corticosteroids and their metabolites are also excreted into the bile.
Pharmacokinetic studies in men with Desoximetasone Cream USP, 0.25% with tagged desoximetasone showed a total of 5.2% ± 2.9% excretion in urine (4.1% ± 2.3%) and feces (1.1% ± 0.6%) and no detectable level (limit of sensitivity: 0.005 µg/mL) in the blood when it was applied topically on the back followed by occlusion for 24 hours. Seven days after application, no further radioactivity was detected in urine or feces. The half-life of the material was 15 ± 2 hours (for urine) and 17 ± 2 hours (for feces) between the third and fifth trial day. Studies with other similarly structured steroids have shown that predominant metabolite reaction occurs through conjugation to form the glucuronide and sulfate ester. Studies performed with Desoximetasone Cream USP, 0.05%; Desoximetasone Cream USP, 0.25%; Desoximetasone Gel USP, 0.05% and Desoximetasone Ointment USP, 0.25% indicate that they are in the high range of potency as compared with other topical corticosteroids.
INDICATIONS AND USAGE
Topical corticosteroids are high potency corticosteroids indicated for the relief of the inflammatory and pruritic manifestations of corticosteroid responsive dermatoses.
CONTRAINDICATIONS
Topical corticosteroids are contraindicated in those patients with a history of hypersensitivity to any of the components of the preparation.
WARNINGS
Desoximetasone Cream USP, 0.05%; Desoximetasone Cream USP, 0.25%; Desoximetasone Gel USP, 0.05% and Desoximetasone Ointment USP, 0.25% are not for ophthalmic use.
Keep out of reach of children.
PRECAUTIONS
General
Systemic absorption of topical corticosteroids can produce reversible hypothalamic-pituitary-adrenal (HPA) axis suppression with the potential for glucocorticosteroid insufficiency after withdrawal of treatment. Manifestations of Cushing's syndrome, hyperglycemia, and glucosuria can also be produced in some patients by systemic absorption of topical corticosteroids while on treatment. Use of more than one corticosteroid-containing product at the same time may increase total systemic glucocorticoid exposure. (See DOSAGE AND ADMINISTRATION section.)
Patients applying a topical steroid to a large surface area or to areas under occlusion should be evaluated periodically for evidence of HPA axis suppression. This may be done by using ACTH stimulation and urinary free cortisol tests.
If HPA axis suppression is noted, an attempt should be made to withdraw the drug, to reduce the frequency of application, or to substitute a less potent corticosteroid. Recovery of HPA axis function is generally prompt upon discontinuation of topical corticosteroids. Infrequently, signs and symptoms of glucocorticosteroid insufficiency may occur requiring supplemental systemic corticosteroids. For information on systemic supplementation, see prescribing information for those products.
Pediatric patients may be more susceptible to systemic toxicity from equivalent doses due to their larger skin surface to body mass ratios. (See PRECAUTIONS – Pediatric Use.)
If irritation develops, Desoximetasone Cream USP, 0.05%; Desoximetasone Cream USP, 0.25%; Desoximetasone Gel USP, 0.05% or Desoximetasone Ointment USP, 0.25% should be discontinued and appropriate therapy instituted. Allergic contact dermatitis with corticosteroids is usually diagnosed by observing failure to heal rather than noting a clinical exacerbation as with most topical products not containing corticosteroids. Such an observation should be corroborated with appropriate diagnostic patch testing.
If concomitant skin infections are present or develop, an appropriate antifungal or antibacterial agent should be used. If a favorable response does not occur promptly, use of Desoximetasone Cream USP, 0.05%; Desoximetasone Cream USP, 0.25%; Desoximetasone Gel USP, 0.05% or Desoximetasone Ointment USP, 0.25% should be discontinued until the infection has been adequately controlled.
This medication should not be used on the face, underarm or groin area.
As with other corticosteroids, therapy should be discontinued when control is achieved. If no improvement is seen within two weeks, the diagnosis and therapy should be re-evaluated.
Information for the Patient
Patients using topical corticosteroids should receive the following information and instructions. This information is intended to aid in the safe and effective use of this medication. It is not a disclosure of all possible adverse or unintended effects:
- This medication is to be used as directed by the physician. It is for external use only. Avoid contact with the eyes.
- This medication should not be used for any disorder other than that for which it was prescribed.
- The treated skin area should not be bandaged, otherwise covered or wrapped, so as to be occlusive unless directed by the physician.
- Patients should report to their physician any signs of local adverse reactions.
- Other topical corticosteroid-containing products should not be used with Desoximetasone Cream USP, 0.05% or Desoximetasone Cream USP, 0.25% or Desoximetasone Gel USP, 0.05% or Desoximetasone Ointment USP, 0.25% without first talking to your physician.
- If no improvement is seen in 2 weeks, contact your physician.
Laboratory Tests
The following tests may be helpful in evaluating patients for HPA axis suppression:
- ACTH stimulation test
- Urinary free cortisol test
Carcinogenesis, Mutagenesis, and Impairment of Fertility
Long-term animal studies have not been performed to evaluate the carcinogenic potential or the effect on fertility of desoximetasone.
Desoximetasone was nonmutagenic in the Ames test.
Pregnancy
Teratogenic Effects
Pregnancy Category C
Corticosteroids have been shown to be teratogenic in laboratory animals when administered systemically at relatively low dosage levels. Some corticosteroids have been shown to be teratogenic after dermal application in laboratory animals.
Desoximetasone has been shown to be teratogenic and embryotoxic in mice, rats, and rabbits when given by subcutaneous or dermal routes of administration in doses 3 to 30 times the human dose of Desoximetasone Cream USP, 0.25% or Desoximetasone Ointment USP, 0.25% or in doses 15 to 150 times the human dose of Desoximetasone Cream USP, 0.05% or Desoximetasone Gel USP, 0.05%.
There are no adequate and well-controlled studies in pregnant women. Desoximetasone Cream USP, 0.05%; Desoximetasone Cream USP, 0.25%; Desoximetasone Gel USP, 0.05% and Desoximetasone Ointment USP, 0.25% should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Nursing Mothers
It is not known whether topical administration of corticosteroids can result in sufficient systemic absorption to produce detectable quantities in breast milk. Systemically administered corticosteroids are secreted into breast milk in quantities not likely to have a deleterious effect on the infant. Nevertheless, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Pediatric Use
Pediatric patients may demonstrate greater susceptibility to topical corticosteroid-induced HPA axis suppression and Cushing's syndrome than adult patients because of a larger skin surface area to body weight ratio.
Hypothalamic-pituitary-adrenal (HPA) axis suppression, Cushing's syndrome and intracranial hypertension have been reported in pediatric patients receiving topical corticosteroids. Manifestations of adrenal suppression in pediatric patients include linear growth retardation, delayed weight gain, low plasma cortisol levels, and absence of response to ACTH stimulation. Manifestations of intracranial hypertension include bulging fontanelles, headaches, and bilateral papilledema. Chronic corticosteroid therapy may interfere with linear bone growth in pediatric patients.
Geriatric Use
Clinical studies of Desoximetasone Cream USP, 0.05%; Desoximetasone Cream USP, 0.25%; Desoximetasone Gel USP, 0.05% and Desoximetasone Ointment USP, 0.25% did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In generally, dose selection for an elderly patient should be cautious.
ADVERSE REACTIONS
In controlled clinical studies the incidence of adverse reactions was low (0.8%) for Desoximetasone Cream USP, 0.05% and included pruritus, erythema, vesiculation and burning sensation. The incidence of adverse reactions was also 0.8% for Desoximetasone Cream USP, 0.25% and included burning, folliculitis, and folliculopustular lesions. The incidence of adverse reactions for Desoximetasone Gel USP, 0.05% was 0.3% with one subject reporting stinging and burning at the site of application. The incidence of adverse reactions for Desoximetasone Ointment USP, 0.25% was also 0.3% and consisted of development of comedones at the site of application.
The following local adverse reactions are reported infrequently when topical corticosteroids are used as recommended but may occur more frequently with the use of occlusive dressings and higher potency corticosteroids. These are listed in an approximate decreasing order of occurrence: burning, itching, irritation, dryness, folliculitis, hypertrichosis, acneiform eruptions, hypopigmentation, perioral dermatitis, allergic contact dermatitis, maceration of the skin, secondary infection, skin atrophy, striae, and miliaria.
Systemic absorption of topical corticosteroids has produced hypothalamic-pituitary-adrenal (HPA) axis suppression, manifestations of Cushing's syndrome, hyperglycemia, and glucosuria in some patients.
OVERDOSAGE
Topically applied Desoximetasone Cream USP, 0.05%; Desoximetasone Cream USP, 0.25%; Desoximetasone Gel USP, 0.05% and Desoximetasone Ointment USP, 0.25% can be absorbed in sufficient amounts to produce systemic effects (see PRECAUTIONS).
DOSAGE AND ADMINISTRATION
Apply a thin film of Desoximetasone Cream USP, 0.05% or Desoximetasone Cream USP, 0.25% or Desoximetasone Gel USP, 0.05% or Desoximetasone Ointment USP, 0.25% to the affected skin areas twice daily. Rub in gently.
HOW SUPPLIED
Desoximetasone Cream USP, 0.05% is supplied in 15 gram (NDC 51672-1271-1) and 60 gram (NDC 51672-1271-3) tubes.
Desoximetasone Cream USP, 0.25% is supplied in 15 gram (NDC 51672-1270-1) and 60 gram (NDC 51672-1270-3) tubes.
Desoximetasone Gel USP, 0.05% is supplied in 15 gram (NDC 51672-1261-1) and 60 gram (NDC 51672-1261-3) tubes.
Desoximetasone Ointment USP, 0.25% is supplied in 15 gram (NDC 51672-1262-1) and 60 gram (NDC 51672-1262-3) tubes.
Store at 20° - 25°C (68° - 77°F) [see USP Controlled Room Temperature].
Mfd. by: Taro Pharmaceuticals Inc., Brampton, Ontario, Canada L6T 1C1
Revised: November, 2006
PK-0964-6 1025
| DESOXIMETASONE
desoximetasone gel |
|||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||
| DESOXIMETASONE
desoximetasone cream |
|||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||
| DESOXIMETASONE
desoximetasone cream |
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
| DESOXIMETASONE
desoximetasone ointment |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
Revised: 11/2008Taro Pharmaceuticals U.S.A., Inc.