CORIFACT- factor xiii concentrate (human)
CSL Behring GmbH
----------
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use Corifact safely and effectively. See full prescribing information for Corifact.
Corifact, Factor XIII Concentrate (Human) For Intravenous Use. Lyophilized Concentrate for Reconstitution Initial U.S. Approval: 2011 INDICATIONS AND USAGECorifact is indicated for routine prophylactic treatment of congenital Factor XIII (FXIII) deficiency. The effectiveness of Corifact is based on maintaining a trough FXIII activity level of approximately 5% to 20%. There are no controlled trials demonstrating a direct benefit on treatment of bleeding episodes with Corifact (1). DOSAGE AND ADMINISTRATIONFor intravenous use only. Reconstitute prior to use. Initial Dose (2.2)
Subsequent Dosing (2.2)
Dose Adjustment Using the Berichrom Activity Assay (2.2, Table 1)
Preparation and Reconstitution (2.3)
Administration (2.4)
DOSAGE FORMS AND STRENGTHS
CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
ADVERSE REACTIONS
To report SUSPECTED ADVERSE REACTIONS, contact the CSL Behring Pharmacovigilance Department at 1-866-915-6958 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. USE IN SPECIFIC POPULATIONS
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling. Revised: 09/2012 |
FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
Corifact, Factor XIII (FXIII) Concentrate (Human) is indicated for routine prophylactic treatment of congenital FXIII deficiency. The effectiveness of Corifact is based on maintaining a trough FXIII activity level of approximately 5% to 20%. There are no controlled trials demonstrating a direct benefit on treatment of bleeding episodes with Corifact.
2 DOSAGE AND ADMINISTRATION
2.1 Dosage
Corifact dosing regimen should be individualized based on body weight, laboratory values, and the patient's clinical condition.
2.2 Dosing Schedule
Initial dose
- 40 International Units (units) per kg body weight
- The injection rate should not exceed 4 mL per minute.
Subsequent dosing
- Dosing should be guided by the most recent trough FXIII activity level, with dosing every 28 days (4 weeks) to maintain a trough FXIII activity level of approximately 5% to 20%.1 Recommended dosing adjustments of ±5 units per kg should be based on trough FXIII activity levels as shown in Table 1 and the patient's clinical condition [see Clinical Pharmacology (12.3)].
- Dosing adjustments should be guided based on a specific assay used to determine FXIII levels. An example of dose adjustment using the Berichrom activity assay is outlined in Table 1 below.
| Factor XIII Activity Trough Level (%) | Dosage Change |
|---|---|
| One trough level of <5% | Increase by 5 units per kg |
| Trough level of 5% to 20% | No change |
| Two trough levels of >20% | Decrease by 5 units per kg |
| One trough level of >25% | Decrease by 5 units per kg |
The potency expressed in units is determined using the Berichrom activity assay, referenced to the current International Standard for Blood Coagulation Factor XIII, Plasma. Therefore, a unit herein is equivalent to an International Unit.
2.3 Preparation and Reconstitution
- For intravenous use only.
- Prepare and administer using aseptic techniques.
- Do not use Corifact beyond the expiration date on the vial label and carton.
- Perform a visual inspection of the reconstituted solution. It should be colorless to slightly yellowish, slightly opalescent, and free from visible particles.
- Administer Corifact using aseptic techniques to maintain product sterility.
- Corifact is for single use only. Contains no preservatives. The product must be used within 4 hours after reconstitution. Do not refrigerate or freeze the reconstituted solution. Discard partially used vials.
The procedures below are provided as general guidelines for the preparation and reconstitution of Corifact.
Reconstitute Corifact at room temperature as follows:
| |
| |
| |
|  Fig. 1 |
| 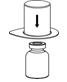 Fig. 2 |
| 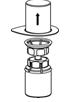 Fig. 3 |
| 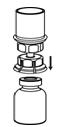 Fig. 4 |
| 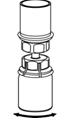 Fig. 5 |
| 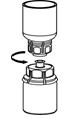 Fig. 6 |
| 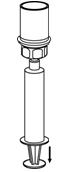 Fig. 7 |
| 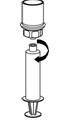 Fig. 8 |
| |
| |
|
2.4 Administration
- Do not mix Corifact with other medicinal products, and administer Corifact through a separate infusion line.
- Use aseptic techniques when administering Corifact.
- Administer Corifact after it has been brought to room temperature. It should be administered by slow intravenous injection at a rate not exceeding 4 mL per minute.
- Record the batch number of the product in the patient's medical records every time Corifact is administered to the patient.
3 DOSAGE FORMS AND STRENGTHS
Corifact is available as a single-use vial containing 1000-1600 units of FXIII as a lyophilized concentrate. A 20 mL vial of Sterile Water for Injection, USP, is provided for reconstitution.
The actual units of potency of FXIII are stated on each Corifact vial label and carton.
4 CONTRAINDICATIONS
Corifact is contraindicated in patients with known anaphylactic or severe systemic reactions to human plasma-derived products or to any components in Corifact [see Description (11)].
5 WARNINGS AND PRECAUTIONS
5.1 Hypersensitivity
Hypersensitivity reactions (including allergy, rash, pruritus, and erythema) have been observed with Corifact. If signs or symptoms of anaphylaxis or hypersensitivity reactions (including urticaria, rash, tightness of the chest, wheezing, hypotension) occur, immediately discontinue administration [see Patient Counseling Information (17)] and institute appropriate treatment.
5.2 Immunogenicity
Development of inhibitory antibodies against FXIII has been detected in patients receiving Corifact. Monitor patients for possible development of inhibitory antibodies. Presence of inhibitory antibodies may manifest as an inadequate response to treatment. If expected plasma FXIII activity levels are not attained, or if breakthrough bleeding occurs while receiving prophylaxis, an assay that measures FXIII inhibitory antibody concentrations should be performed. One case of inhibitory antibodies against FXIII has been reported in the clinical studies [see Adverse Reactions (6.1)]. Cases of inhibitory antibodies against FXIII in patients with congenital FXIII deficiency have also been reported in postmarketing surveillance.
5.3 Thromboembolic Risk
Thromboembolic complications have been reported in postmarketing surveillance [see Adverse Reactions (6.2)]. Benefits and risks should be carefully assessed in pregnant women because of their hypercoagulable state and potential for increased risk of thromboembolic events.
5.4 Transmission of Infectious Agents
Corifact is made from human plasma. Because this product is made from human blood, it may carry a risk of transmitting infectious agents, e.g., viruses, and theoretically, the Creutzfeldt-Jakob disease (CJD) agent. There is also the possibility that unknown infectious agents may be present in such products. The risk that such products could transmit viruses has been reduced by screening plasma donors for prior exposure to certain viruses, by testing for the presence of certain current virus infections, and by inactivating and removing certain viruses during manufacture [see Description (11)]. Despite these measures, such products may still potentially transmit disease.
Consider appropriate vaccination (against hepatitis A and B virus) for patients in regular/repeated receipt of Corifact. All infections thought by a physician to have been possibly transmitted by this product should be reported by the physician or other healthcare provider to the CSL Behring Pharmacovigilance Department at 1-866-915-6958 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
5.5 Monitoring Laboratory Tests
- Monitoring of patient's trough FXIII activity level [see Dosage and Administration (2.2)] is recommended during treatment with Corifact.
- If breakthrough bleeding occurs, or if expected peak plasma FXIII activity levels are not attained [see Clinical Pharmacology (12.3)], an investigation to determine the presence of FXIII inhibitory antibodies should be performed.
6 ADVERSE REACTIONS
The most common adverse reactions reported in clinical trials in greater than one subject (frequency >1%) following Corifact treatment are hypersensitivity reactions (including allergy, rash, pruritus, and erythema), chills/rise in temperature, arthralgia, headache, elevated thrombin-antithrombin levels, and an increase in hepatic enzymes. Adverse reactions are those adverse events (AEs) considered to be "at least possibly related" to the infusion of Corifact.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Twelve clinical studies were conducted and included 187 unique subjects, 90 subjects were <16 years of age [see Use in Specific Populations (8.4)]. These 187 subjects received a total of approximately 3930 infusions of Corifact.
The most common adverse events reported in clinical trials in greater than three subjects (frequency >2%) are flu-like syndrome, abdominal pain, diarrhea, contusion, joint injury, limb injury, road traffic accident, upper respiratory tract infection, arthralgia, headache, epistaxis, vomiting, fever, hematoma, head injury, and rash. Adverse events (AEs) are defined as treatment-emergent AEs that started after the first Corifact study infusion.
In the 12-week prospective, open-label, multicenter, pharmacokinetic and safety study conducted in 7 females and 7 males with congenital FXIII deficiency, ranging in age from 5 to 42 years (3 children, 2 adolescents, and 9 adults), there were no reports of deaths, life-threatening events, or adverse events that led to discontinuation or withdrawal from the study. No breakthrough bleeding episodes were reported in this study.
A case of neutralizing antibodies against FXIII was reported in the ongoing postmarketing clinical study. The patient received prophylactic treatment with Corifact for ten years. Concomitant medications included interferon for hepatitis C infection. This patient presented with bruising, and post-infusion FXIII levels were found to be lower than expected. Over several weeks, FXIII recovery values decreased, so the dose and frequency of treatments were increased. Neutralizing antibodies to FXIII were detected, interferon treatment was discontinued, and the subject underwent plasmapheresis. Within a month, neutralizing antibodies were no longer detectable, FXIII recovery levels improved, and the previous prophylactic regimen was resumed.
6.2 Postmarketing Experience
Because postmarketing reporting of adverse reactions is voluntary and from a population of uncertain size, it is not always possible to reliably estimate the frequency of these reactions or establish a causal relationship to product exposure.
The adverse reactions spontaneously reported after administration of Corifact during postmarketing surveillance outside the US since 1993, identified by system organ class are provided in Table 2.
| MedDRA System Organ Class | MedDRA Preferred Term/Symptoms |
|---|---|
|
|
| Immune system disorders | Allergic/anaphylactic reaction (including cutaneous reactions, alteration in blood pressure, nausea, dyspnea, fever, and chills) |
| General disorders and administration site conditions | Pyrexia |
| Blood and lymphatic system disorders | Factor XIII inhibitor formation |
| Vascular disorders | Thrombosis, embolism |
| Infections and infestations | Transmission of an infectious agent via medicinal products* made from human plasma [see Warnings and Precautions (5.4)] |
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Category C. Animal reproduction studies have not been conducted with Corifact. Safety and effectiveness in pregnancy have not been established. Corifact should be given to a pregnant woman only if clearly needed [see Warnings and Precautions (5.3)].
8.2 Labor and Delivery
Corifact has not been studied for use during labor and delivery. Safety and effectiveness in labor and delivery have not been established.
8.3 Nursing Mothers
It is not known whether Corifact is excreted in human milk. Use Corifact only if clearly needed when treating nursing women.
8.4 Pediatric Use
Of the 187 unique subjects in the Corifact clinical studies, 90 were subjects <16 years of age at the time of enrollment (<1 month, n=2; 1 month to <2 years, n=14; 2 to 11 years, n=52; 12 to <16 years, n=22). In the pharmacokinetic study [see Clinical Pharmacology (12.3)], 5 of the 14 subjects ranged in age from 2 to <16 years. Subjects less than 16 years had a shorter half-life (5.7 ± 1.00 days) and faster clearance (0.29 ± 0.12 mL/hr/kg) compared to adults (half-life: 7.1 ± 2.74 days, clearance: 0.22 ± 0.07 mL/hr/kg). The number of subjects less than 16 years of age limits the statistical interpretation. There were no apparent differences in the safety profile in children as compared to adults. All pediatric subjects were treated for congenital Factor XIII deficiency.
11 DESCRIPTION
Corifact is a heat-treated, lyophilized FXIII (coagulation factor XIII) concentrate made from pooled human plasma. Each vial contains 1000-1600 units FXIII, 120 to 200 mg human albumin, 120 to 320 mg total protein, 80 to 120 mg glucose and 140 to 220 mg sodium chloride. Sodium hydroxide may have been used to adjust the pH.
All plasma used in the manufacture of Corifact is obtained from US donors and is tested using serological assays for hepatitis B surface antigen and antibodies to HIV-1/2 and HCV. The plasma is tested with Nucleic Acid Testing (NAT) for HCV, HIV-1, HAV and HBV and found to be non-reactive (negative), and the plasma is also tested by NAT for Human Parvovirus B19. Only plasma that passed virus screening is used for production, and the limit for Parvovirus B19 in the fractionation pool is set not to exceed 104 International Units of Parvovirus B19 DNA per mL.
Corifact is manufactured from cryo-depleted plasma into an ethanol precipitate, which is then purified by the following steps:
- Precipitation/adsorption
- Ion exchange chromatography
- Heat-treatment (+60°C for 10 hours in an aqueous solution)
- Virus filtration over two 20 nm filters in series
The sterile filtered final bulk solution is filled into vials and lyophilized. These four manufacturing steps were independently validated in a series of in vitro experiments for their capacity to inactivate or remove both enveloped and non-enveloped viruses. Table 3 shows the virus clearance capacity of the Corifact manufacturing process, expressed as mean log10 reduction factor.
| Manufacturing Steps | Virus Reduction Factor (log10) | |||||||
|---|---|---|---|---|---|---|---|---|
| Enveloped Viruses | Non-Enveloped Viruses | |||||||
| HIV | BVDV | WNV | PRV | HAV | CPV | |||
| HIV, Human immunodeficiency virus type 1, model for HIV-1 and HIV-2 | ||||||||
| BVDV, bovine viral diarrhea virus, model for HCV | ||||||||
| WNV, West Nile virus | ||||||||
| PRV, pseudorabies virus, a model for large enveloped DNA viruses | ||||||||
| HAV, Hepatitis A virus | ||||||||
| CPV, canine parvovirus, model for B19V | ||||||||
| B19V, Human parvovirus B19 | ||||||||
| N/A, not applicable | ||||||||
| n.d., not done | ||||||||
| Al(OH)3 Adsorption / Vitacel® and Defibrination | n.d. | n.d. | n.d. | 6.9 | n.d. | n.d. | ||
| Ion Exchange Chromatography | 5.0 | 3.3 | n.d. | ≥8.0 | 3.4 | 3.7 | ||
| Heat Treatment | ≥7.7 | ≥8.1 | ≥7.4 | N/A* | 4.3 | 1.0† | ||
| 20 nm / 20 nm Virus Filtration | ≥6.1 | ≥5.0 | ≥7.4 | ≥6.4 | ≥5.6 | 6.1 | ||
| Overall Virus Reduction Factor (log10) | ≥18.8 | ≥16.4 | ≥14.8 | ≥21.3 | ≥13.3 | 10.8 | ||
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Corifact (FXIII) is an endogenous plasma glycoprotein consisting of two A-subunits and two B-subunits. FXIII circulates in blood and is present in platelets, monocytes, and macrophages. FXIII appears in 2 forms, a heterotetrameric (A2B2) plasma protein with a molecular weight of about 320 kilodaltons and a homodimeric (A2) cellular form. FXIII is a proenzyme that is activated, in the presence of calcium ions, by thrombin cleavage of the A-subunit to become activated FXIII (FXIIIa). Intracellularly, the homodimeric form of only the A-subunits (A2) are found. The B-subunits in plasma have no enzymatic activity, and function as carrier molecules for the A-subunits. They stabilize the structure of the A-subunits and protect them from proteolysis.
FXIIIa promotes cross-linking of fibrin during coagulation and is essential to the physiological protection of the clot against fibrinolysis. FXIIIa is a transglutaminase enzyme that catalyzes the cross-linking of the fibrin α- and γ-chains for fibrin stabilization and renders the fibrin clot more elastic and resistant to fibrinolysis.2,3 FXIIIa also cross-links α2-plasmin inhibitor to the α-chain of fibrin, resulting in protection of the fibrin clot from degradation by plasmin. Cross-linked fibrin is the end result of the coagulation cascade, and provides tensile strength to a primary hemostatic platelet plug.3
12.2 Pharmacodynamics
In clinical studies, the intravenous administration of Corifact demonstrated an increase in plasma levels of FXIII lasting approximately 28 days.
In the pharmacokinetic study, after the third 40 units per kg dose (steady state), the mean increase in FXIII activity levels was 83% with a range of 48 to 114% over the baseline [see Clinical Pharmacology (12.3)].
12.3 Pharmacokinetics
A 12-week prospective, open-label, multicenter pharmacokinetic and safety study was conducted in 7 females and 7 males with congenital FXIII deficiency, ranging in age from 5 to 42 years (3 children, 2 adolescents, 9 adults). One adult male did not complete the pharmacokinetic study.
Each subject received 40 units per kg Corifact intravenously every 28 days for a total of three doses administered at approximately 250 units per min. Blood samples for doses 1 and 2 were drawn from patients to determine the FXIII activity level at baseline and 30 and 60 minutes after the infusion. Following the infusion of the third dose of Corifact, blood samples were drawn at regular intervals up to 28 days to determine the pharmacokinetic parameters. The pharmacokinetic parameters based on baseline adjusted FXIII activity (Berichrom assay) are shown in Table 4.
| Parameters | Mean ±SD |
|---|---|
| AUC ss, (0-inf) = Area under the plasma concentration curve from time 0 to infinity at steady state | |
| Css, max: Peak concentration at steady state | |
| Css, min: Trough concentration at steady state | |
| Tmax: Time to peak concentration | |
| CL: Clearance | |
| Vss: Volume of distribution at steady state | |
| MRT = Mean residence time | |
| SD = Standard deviation | |
|
|
| AUC ss, 0-inf (units∙hr/mL) | 184.0 ±65.78 |
| Css, max (units/mL)* | 0.9 ±0.20 |
| Css, min (units/mL)* | 0.05 ±0.05 |
| Tmax (hr) | 1.7 ±1.44 |
| Half-life [days] | 6.6 ±2.29 |
| CL [mL/hr/kg] | 0.25 ±0.09 |
| Vss [mL/kg] | 51.1 ±12.61 |
| MRT [days] | 10.0 ±3.45 |
Due to the small sample size, the impact of age, gender, and race on the pharmacokinetics of Corifact could not be reliably evaluated.
13 NONCLINICAL TOXICOLOGY
13.2 Animal Toxicology and/or Pharmacology
Corifact was studied in an acute toxicity study in mice and rats at doses up to 3550 units per kg and 1420 units per kg, respectively. Repeat dose toxicity was studied in rats at daily doses up to 350 units per kg for a period of 14 days. No signs of toxicity were observed in the single dose and repeat dose studies.
A local tolerance study demonstrated no clinical or histopathological changes at the injection site after intravenous, intra-arterial or para-venous administration of Corifact to rabbits.
A thrombogenicity test was performed in rabbits at doses up to 350 units per kg. Corifact showed no thrombogenic potential at the doses tested.
14 CLINICAL STUDIES
The pharmacokinetic study evaluated three doses at 40 units per kg every 28 days for each subject [see Clinical Pharmacology (12.3)]. Blood sampling before and after infusion for the first two doses was to determine FXIII activity and a complete PK analysis was conducted after the third dose (steady state). FXIII activity levels were determined by the Berichrom activity assay.
Maintaining the FXIII activity trough level between 5% and 20% is predicted to provide a clinical benefit. The clinical benefit will be verified in a post-marketing study to measure the prevention of spontaneous bleeding episodes with routine prophylactic treatment for patients with congenital FXIII deficiency. Dosing will be individualized and adjusted with the objective to maintain FXIII activity trough levels of 5% to 20%.
15 REFERENCES
- Hsieh L, Nugent D. Factor XIII deficiency. Haemophilia 2008; 14:1190-1200.
- Lauer P, Metzner HJ, Zettlmeißl G, Li M, et al. Targeted Inactivation of the Mouse Locus Encoding Coagulation Factor XIII-A: Hemostatic Abnormalities in Mutant Mice and Characterization of the Coagulation Deficit. Thromb Haemost. 2002;88:967-74.
- Dardik R, Loscalzo J, Inbal A. Factor XIII (FXIII) and Angiogenesis. J Thromb Haemost. 2006;4:19-25.
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
- Corifact is supplied in a single-use vial.
- Each carton (kit) contains one single-use vial of Corifact for reconstitution with 20 mL of Sterile Water for Injection, USP, a Mix2Vial filter transfer set, and one alcohol swab.
- The actual units of potency of FXIII Concentrate (Human) are stated on each Corifact vial label and carton.
- The Corifact packaging components are not made with natural rubber latex.
The product presentation includes a package insert and the following components:
| Presentation | Carton NDC Number | Components |
|---|---|---|
| 1000-1600 units | 63833-518-02 |
|
16.2 Storage and Handling
- Store Corifact in a refrigerator at 2-8°C (36-46°F). Corifact is stable for 24 months, up to the expiration date on the carton and vial labels. Within the expiration date, Corifact may be stored at room temperature not to exceed 25°C (77°F) for up to 6 months.
- Do not return the product to the refrigerator after it is stored at room temperature. Clearly mark the beginning date of room temperature storage on the carton label.
- Do not use beyond the expiration date on the carton and vial labels, or end of the period for room temperature storage, whichever comes first.
- Store the vial in the original carton in order to protect it from light. Do not freeze.
- Administer promptly after reconstitution. This product does not contain a preservative. The product must be used within 4 hours after reconstitution. Do not refrigerate or freeze the reconstituted solution.
17 PATIENT COUNSELING INFORMATION
- Inform patients of the signs and symptoms of allergic hypersensitivity reactions, such as urticaria, rash, tightness of the chest, wheezing, hypotension and/or anaphylaxis experienced during or after injection of Corifact [see Warnings and Precautions (5.1)].
- Inform patients of the signs and symptoms of immunogenicity such as breakthrough bleeding [see Warnings and Precautions (5.2)].
- Inform patients of signs and symptoms of thrombosis, such as limb or abdomen swelling and/or pain, chest pain, shortness of breath, loss of sensation or motor power, altered consciousness, vision, or speech [see Warnings and Precautions (5.3)].
- Inform patients that because Corifact is made from human blood, it may carry a risk of transmitting infectious agents, e.g., viruses, and theoretically, the Creutzfeldt-Jakob disease (CJD) agent [see Warnings and Precautions (5.4) and Description (11)].
- FDA-Approved Patient Labeling – Patient Product Information (PPI) is provided following this section.
Manufactured by:
CSL Behring GmbH
35041 Marburg Germany
US License No. 1765
Distributed by:
CSL Behring LLC
Kankakee, IL 60901 USA
Berichrom® Factor XIII assay is a trademark of Siemens Healthcare Diagnostics.
Vitacel® is a trademark of J. Rettenmaier & Söhne GmbH and Co.
Mix2Vial® is a trademark of West Pharmaceuticals Services, Inc. or one of its subsidiaries.
-----------------------------------------------------------------------------------------------------------------------------------------
Corifact
Factor XIII Concentrate (Human)
FDA-Approved Patient Labeling – Patient Product Information (PPI)
This leaflet summarizes important information about Corifact. Please read it carefully before using Corifact and each time you get a refill. There may be new information provided. This information does not take the place of talking with your healthcare provider, and it does not include all of the important information about Corifact. If you have any questions after reading this, ask your healthcare provider.
What is CORIFACT?
Corifact is an injectable medicine used for routine prophylactic treatment of congenital Factor XIII (FXIII) deficiency in adults and pediatric patients. Corifact is a coagulation FXIII concentrate made from human plasma, and has important functions in hemostasis (stopping of bleeding).
Who should not use CORIFACT?
You should not use Corifact if you have experienced hypersensitivity reactions, including anaphylactic or severe systemic reactions to human plasma-derived products.
What should I tell my healthcare provider before CORIFACT is given?
Tell your healthcare provider about all of your medical conditions, including:
- If you are pregnant or planning to become pregnant. It has not been established if Corifact can harm your unborn baby.
- Labor and Delivery: Safety and effectiveness in labor and delivery have not been established.
- Breast feeding: It has not been established if Corifact passes into your milk.
Tell your healthcare provider and pharmacist about all of the medicines you take, including all prescription and non-prescription medicines such as over-the-counter medicines, supplements, or herbal remedies.
How is CORIFACT given?
Corifact is administered into your vein (intravenous injection). Before infusing, Corifact is dissolved using sterile water provided in the package. Your healthcare provider will prescribe the dose that you receive.
What could be the possible side effects of CORIFACT?
Call your healthcare provider or the emergency department right away if you have any of the following symptoms after using Corifact:
- shortness of breath
- rash
- pruritus (itching)
- erythema (redness of the skin)
- fainting/dizziness
- chest pain
- signs of a blood clot including pain, swelling, warmth, redness, or a lump in your legs or arms.
Other possible side effects may include:
- chills/rise in temperature
- arthralgia (joint pain)
- headache
- increase in liver enzymes
- breakthrough bleeding and pain resulting from formation of antibodies against Corifact.
Because Corifact is made from human blood, it may carry a risk of transmitting infectious agents, e.g., viruses, and, theoretically, the Creutzfeldt-Jakob (CJD) agent.
These are not all the possible side effects of Corifact.
Tell your healthcare provider about any side effect that bothers you or that does not go away. You can also report side effects to the FDA at 1-800-FDA-1088.
What else should I know about CORIFACT?
Medicines are sometimes prescribed for purposes other than those listed here. Do not use Corifact for a condition for which it is not prescribed. Do not share Corifact with other people, even if they have the same symptoms that you have.
This leaflet summarizes the most important information about Corifact. If you would like more information, talk to your healthcare provider. You can ask your healthcare provider or pharmacist for information about Corifact that was written for healthcare professionals.
Talk to your healthcare provider before traveling.
This Patient Package Insert has been approved by the US Food and Drug Administration.
PRINCIPAL DISPLAY PANEL - Single-Use Vial Label
FXIII Concentrate (Human)
Corifact®
For Intravenous Use Only
Rx only
Each Corifact® single-use vial contains 1000–1600
International Units (units) of lyophilized FXIII concentrate
for reconstitution with 20 mL of Sterile Water for
Injection, USP. Store Corifact® in a refrigerator at
2–8°C (36–46°F). Corifact® is stable for 24 months,
up to the expiration date on the carton and vial labels.
Do not use if the reconstituted solution is cloudy or
contains particulates. Do not freeze. Protect from light.
Contains no preservative.

| CORIFACT
factor xiii concentrate (human) kit |
||||||||||||||
|
||||||||||||||
|
||||||||||||||
|
||||||||||||||
|
||||||||||||||
|
||||||||||||||
|
||||||||||||||
|
||||||||||||||
|
||||||||||||||
|
||||||||||||||
|
||||||||||||||
|
||||||||||||||
|
||||||||||||||
|
||||||||||||||
|
||||||||||||||
|
||||||||||||||
| Labeler - CSL Behring GmbH (326530474) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
| CSL Behring GmbH | 326530474 | MANUFACTURE | |
