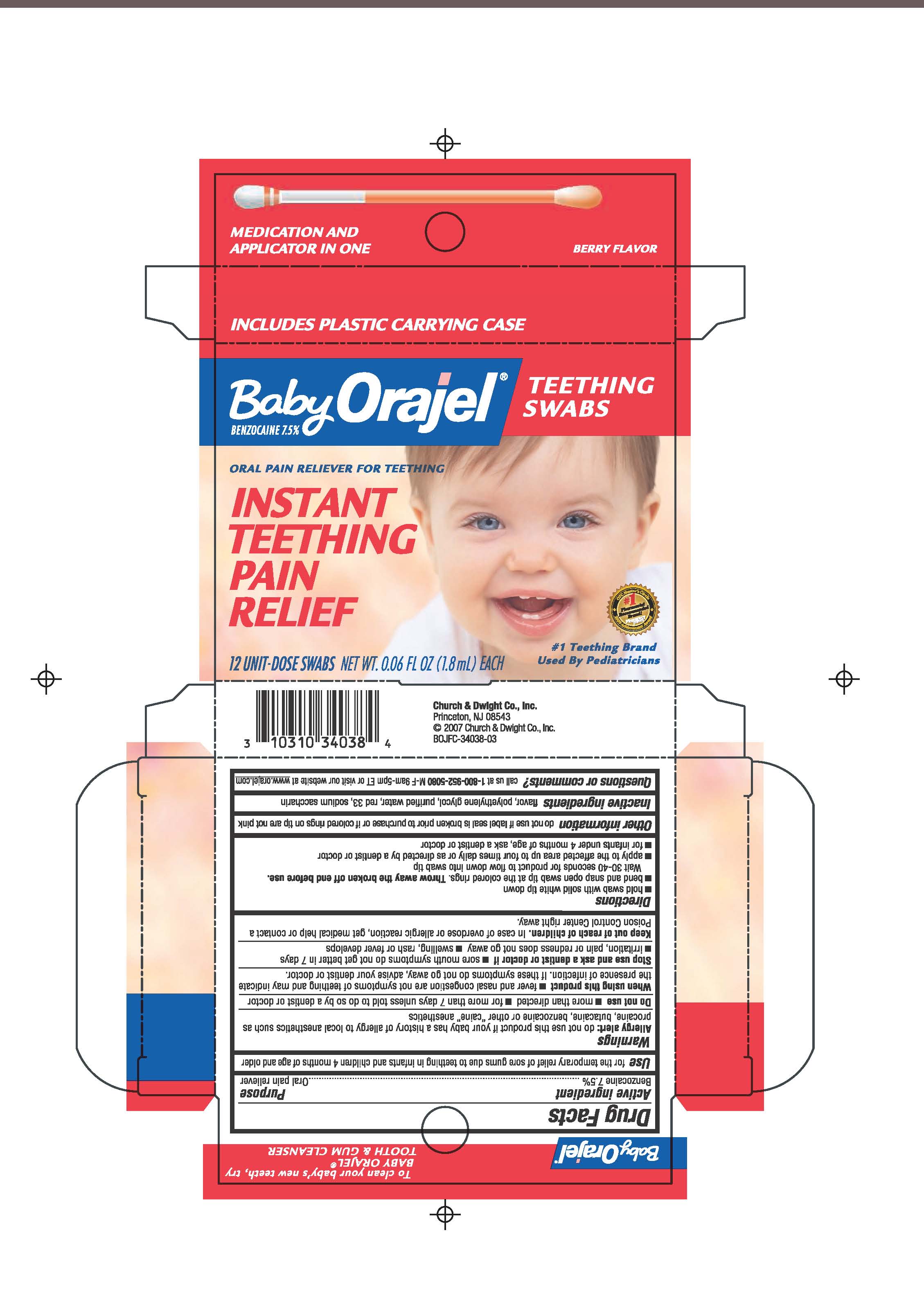
BABY ORAJEL TEETHING PAIN RELIEF
-
benzocaine swab
Church & Dwight Co., Inc.
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Use
for the temporary relief of sore gums due to teething in infants and children 4 months of age and older
Warnings
Allergy Alert
do not use this product if our baby has a history of allergy to local anesthetics such as procaine, butacaine, benzocaine or other "caine" anesthetics
Do not use
more than directed, for more than 7 days unless told to do so by a dentist or doctor
When using this product fever and nasal congestion are not symptoms of teething and may indicate the presence of infection. If these symptoms do not go away, advise your dentist or doctor.
Stop use and ask a dentist of doctor if sore mouth symptoms do not get better in 7 days; irritation, pain or redness does not go away; swelling, rash or fever develops
Keep out of reach of children. in case of overdose or allergic reaction, get medical help or contact a Poison Control Center right away
Directions
hold swab with solid white tip down
bend and snap open swab tip at the colored rings. Throw away the broken off end before use. Wait 30-40 seconds for product to flow down into swab tip
apply to the affected area up to four times daily or as directed by a dentist or doctor
for infants under 4 months of age, ask a dentist or doctor
Other information do not use if label seal is broken prior to purchase, or if colored rings on tip are not pink
inactive ingredients flavor, polyethylene glycol, purified water, red 33 sodium saccharin
Questions or comments? call us at 1-800-952-5080 M-F 9am-5pm ET or visit our website at www.orajel.comBaby Orajel Teething Swabs
Instant Teething Pain Relief
12 unit dose swabs Net wt. 0.06 FL OZ (1.8mL) EACH
| BABY ORAJEL TEETHING PAIN RELIEF
benzocaine swab |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| OTC monograph not final | part356 | 08/01/2010 | 04/24/2012 |
| Labeler - Church & Dwight Co., Inc. (001211952) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| SwabPlus, Inc. | 876441549 | manufacture | |
Revised: 08/2010 Church & Dwight Co., Inc.