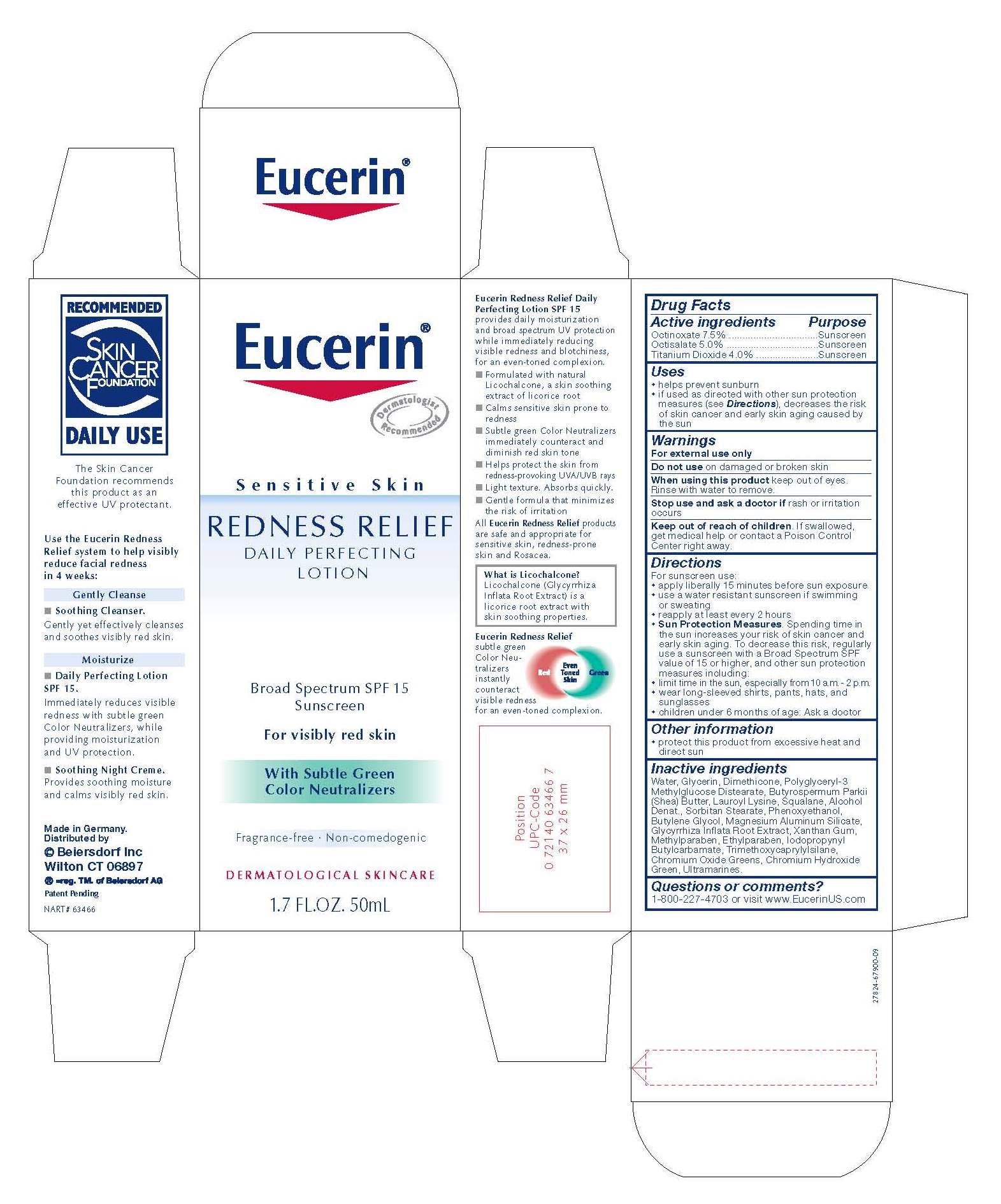
EUCERIN REDNESS RELIEF DAILY PERFECTING
-
octinoxate,
octisalate and
titanium dioxide lotion
Beiersdorf Inc
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Uses
• helps prevent sunburn
• if used as directed with other sun protection measures (see Directions),
decreases the risk of skin cancer and early skin aging caused by the sun
Directions For sunscreen use:
• apply liberally 15 minutes before sun exposure
• use a water resistant sunscreen if swimming or sweating
• reapply at least every 2 hours
• children under 6 months of age: Ask a doctor
• Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and
early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF
value of 15 or higher, and other sun protection measures including:
• limit time in the sun, especially from 10 a.m. - 2 p.m.
• wear long-sleeved shirts, pants, hats, and sunglasses.
• children under 6 months of age: Ask a doctor.
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Inactive ingredients
Water, Glycerin, Dimethicone, Polyglyceryl-3 Methylglucose Distearate, Butyrospermum Parkii
(Shea) Butter, Lauroyl Lysine, Squalane, Alcohol Denat., Sorbitan Stearate, Phenoxyethanol,
Butylene Glycol, Magnesium Aluminum Silicate, Glycyrrhiza Inflata Root Extract, Xanthan Gum,
Methylparaben, Ethylparaben, Iodopropynyl Butylcarbamate, Trimethoxycaprylylsilane,
Chromium Oxide Greens, Chromium Hydroxide Green, Ultramarines.
Eucerin
REDNESS RELIEF
DAILY PERFECTING LOTION
Broad Spectrum SPF 15
Sunscreen
For visibly red skin
With Subtle Green Color Neutralizers
Fragrance-free · Non-comedogenic
DERMATOLOGICAL SKIN CARE
| EUCERIN REDNESS RELIEF DAILY PERFECTING
octinoxate, octisalate, titanium dioxide lotion |
||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| OTC monograph not final | part352 | 01/02/2009 | |
| Labeler - Beiersdorf Inc (001177906) |
Revised: 04/2012 Beiersdorf Inc