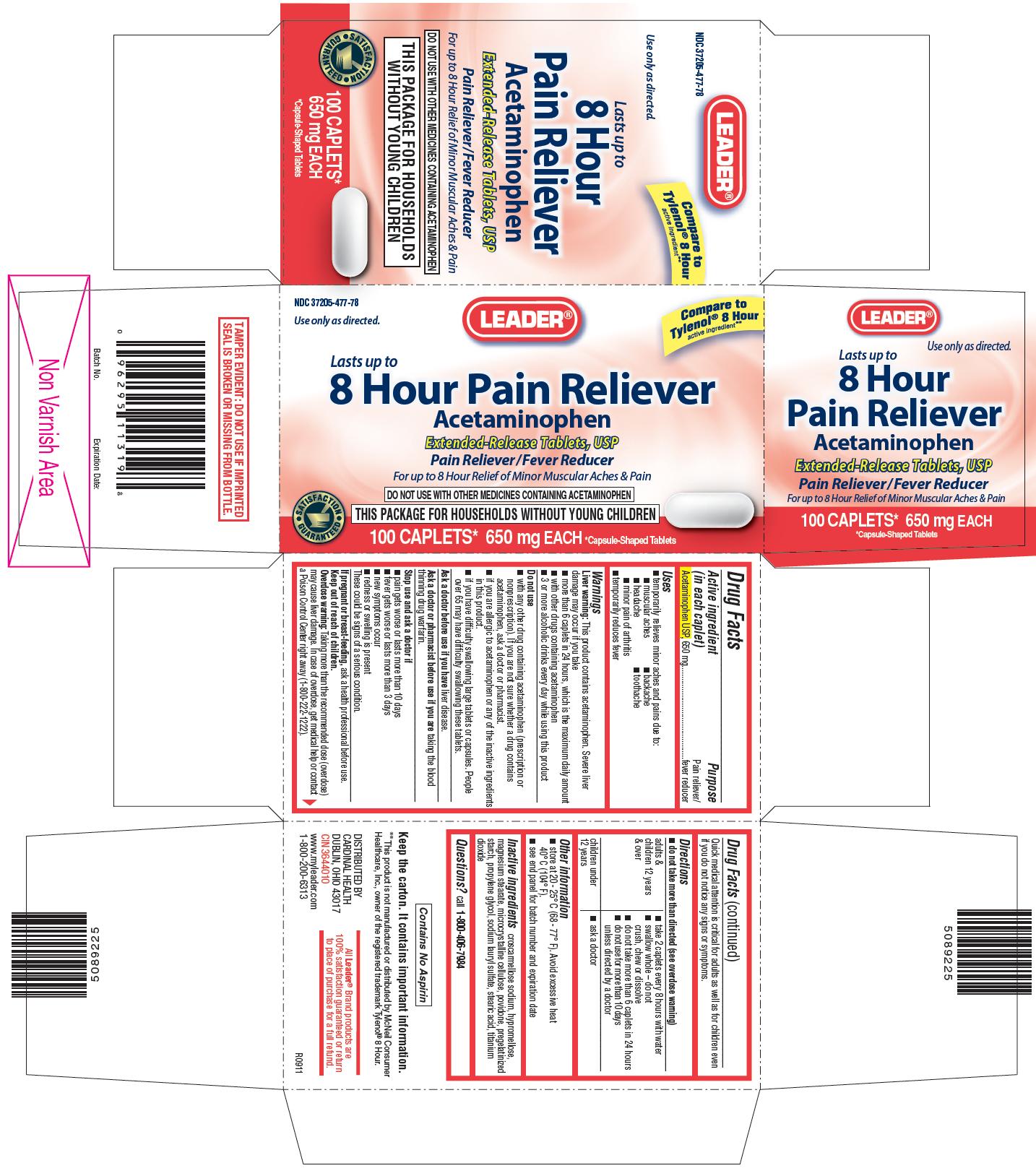
LEADER 8 HOUR PAIN RELIEVER
-
acetaminophen tablet, film coated, extended release
Cardinal Health
----------
USES
- temporarily relieves minor aches and pains due to:
- muscular aches
- backache
- headache
- toothache
- minor pain of arthritis
- temporarily reduces fever
WARNINGS
This product contains acetaminophen. Severe liver damage may occur if you take
- more than 6 caplets in 24 hours, which is the maximum daily amount
- with other drugs containing acetaminophen
- 3 or more alcoholic drinks every day while using this product
Do not use
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- if you are allergic to acetaminophen or any of the inactive ingredients in this product.
- if you have difficulty swallowing large tablets or capsules. People over 65 may have difficulty swallowing these tablets.
Stop use and ask a doctor if
- pain gets worse or lasts more than 10 days
- fever gets worse or lasts more than 3 days
- new symptoms occur
- redness or swelling is present
Keep out of reach of children.
Taking more than the recommended dose (overdose) may cause liver damage. In case of overdose, get medical help or contact a Poison Control Center right away (1-800-222-1222). Quick medical attention is critical for adults as well as for children even if you do not notice any signs or symptoms.
DIRECTIONS
- do not take more than directed (see overdose warning)
Adults & children 12 years & over:
- take 2 caplets every 8 hours with water
- swallow whole - do not crush, chew or dissolve
- do not take more than 6 caplets in 24 hours
- do not use for more than 10 days unless directed by a doctor
- ask a doctor
OTHER INFORMATION
- store at 20 - 25° C (68 - 77° F). Avoid excessive heat 40° C (104° F).
- see end panel for batch number and expiration date
INACTIVE INGREDIENTS
croscarmellose sodium, hypromellose, magnesium stearate, microcrystalline cellulose, povidone, pregelatinized starch, propylene glycol, sodium lauryl sulfate, stearic acid, titanium dioxide
QUESTIONS?
TAMPER EVIDENT: DO NOT USE IF IMPRINTED SEAL IS BROKEN OR MISSING FROM BOTTLE. (for bottle cartons/label and stand-alone labels only)
THIS PACKAGE FOR HOUSEHOLDS WITHOUT YOUNG CHILDREN. (for non CRC packages)
PRINCIPAL DISPLAY PANEL
Lasts up to 8 Hour Pain Reliever
Acetaminophen Extended-Release Tablets, USP
For up to 8 Hour Relief of Minor Muscular Aches & Pain
DO NOT USE WITH OTHER MEDICINES CONTAING ACETAMINOPHEN
THIS PACKAGE FOR HOUSEHOLD WITHOUT YOUNG CHILDREN
Compare to Tylenol® 8 Hour active ingredient**
**This product is not manufactured or distributed by McNeil Consumer Healthcare, Inc., owner of the registered trademark Tylenol® 8 Hour.
| LEADER 8 HOUR PAIN RELIEVER
acetaminophen tablet, film coated, extended release |
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| ANDA | ANDA076200 | 04/30/2002 | |
| Labeler - Cardinal Health (097537435) |
| Registrant - Ohm Laboratories Inc. (051565745) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| Ohm Laboratories Inc. | 184769029 | manufacture | |
Revised: 03/2012 Cardinal Health