ENTEROCINA
-
loperamide hydrochloride tablet
DLC Laboratories, Inc.
Disclaimer: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
----------
Warnings
Do not use if you ever had a rash or other allergic reaction to loperamide HCl
Directions
drink plenty of clear fluids to hep prevent dehydration, which may accompany diarrhea
Find right dose on chart. If possible, use weight to dose, otherwise, use age.
Adults and children 12 years of age and older:
Take 2 tablets after the first loose bowel movement and 1 tablet after each subsequent loose bowel movement and 1 tablet after each subsequent loose bowel movement; but no more than 4 tablets a day.
Children 9-11 years (60-95lbs):
Take 1 tablet after the first loose bowel movement and 1/2 tablet after each subsequent loose bowel movement; but not more than 3 tablets a day.
Children 6-8 years (48-59 lbs):
Take 1 tablet after the first loose bowel movement and 1/2 tablet after each
subsequent loose bowel movement; but not more than 2 tablets a day.
CHILDREN UNDER 6 YEARS OLD (UP TO 47LBS): Consult a physician. Not intended for use in children under 6years old.
Other information
Store at room temperature 15 to 30C (59-86F). See side panel for lot number and expiration date.
Inactive Ingredients
Croscarmellose Sodium Crospovidone, DandC Yellow No. 10, FDandC Blue No. 1 Hydrogenated Vegetable Oil, Lactose Anhydrous, Magnesium Stearate, Powedered Cellulose, Pregelatinized starch.
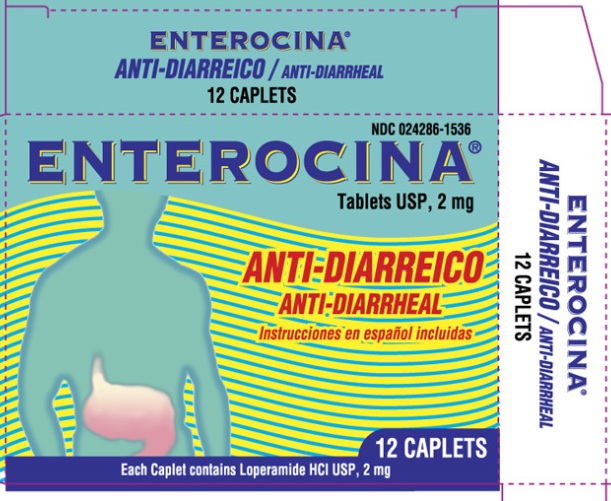
Package Labeling
Enterocina Tablets USP, 2mg
Anti- Diarrheal
12 Caplets
Each caplet contains Loperamide HCl USP, 2mg
Distributed by:
De La Cruz Producs, A division of DLC Laboratories, Inc, Paramount, CA 97723
www.dlclaboratories.com

| ENTEROCINA
loperamide hcl tablet |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| unapproved drug other | 02/28/2012 | ||
| Labeler - DLC Laboratories, Inc. (093351930) |
Revised: 02/2012 DLC Laboratories, Inc.