HAND SANITIZER
-
alcohol gel
HOME DEPOT U.S.A. INC.
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
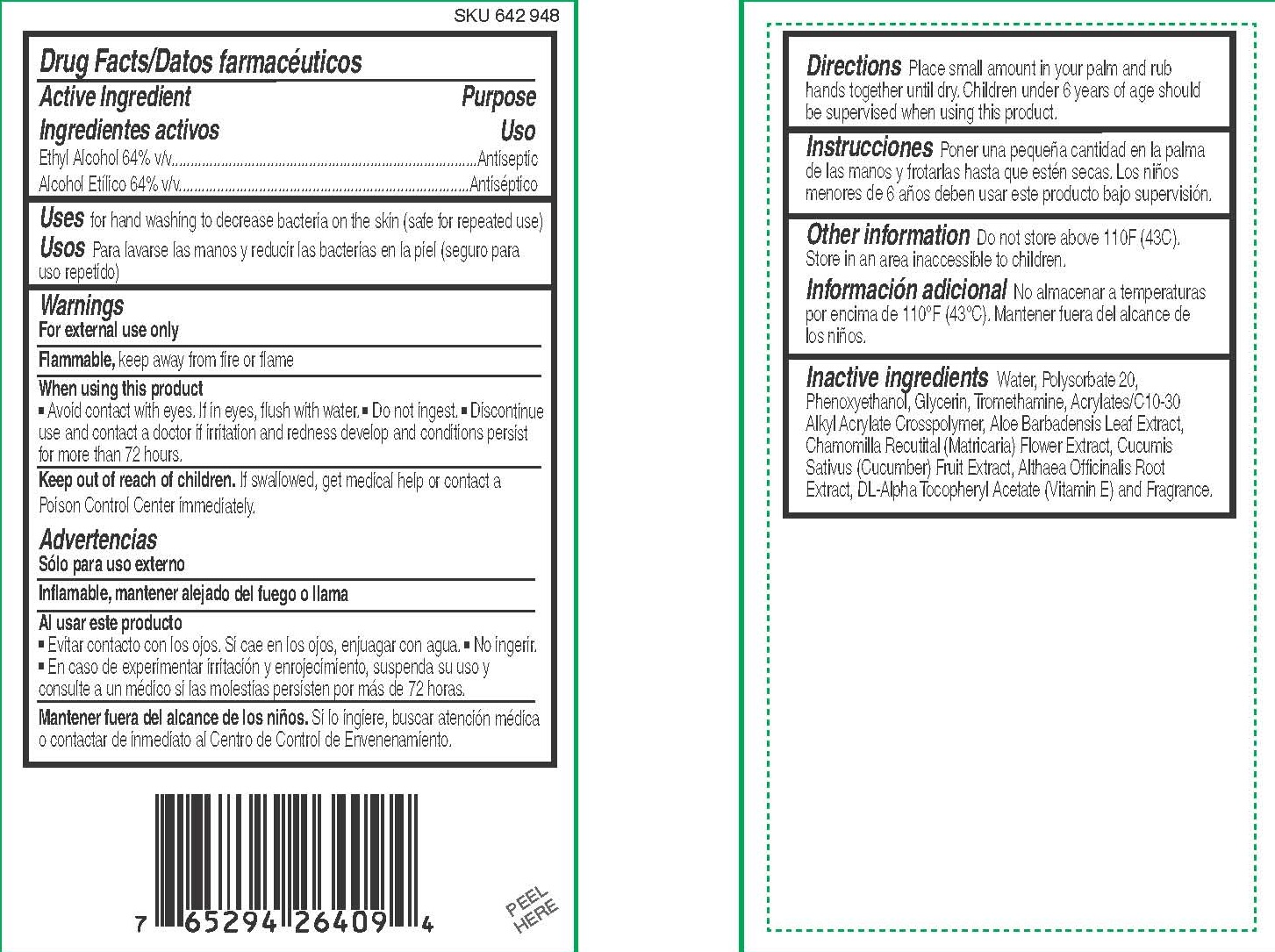
When using this product
■ Avoid contact with eyes. If in eyes, flush with water.
■ Do not ingest.
■ Discontinue use and contact a doctor if irritation and redness develop and conditions persist for more than 72 hours.
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center immediately.
Directions
Place small amount in your palm and rub hands together until dry. Children under 6 years of age should be supervised when using this product.
Inactive ingredients
Water, Polysorbate 20, Phenoxyethanol, Glycerin, Tromethamine, Acrylates/C10-30 Alkyl Acrylate Crosspolymer, Aloe Barbadensis Leaf Extract, Chamomilla Recutital (Matricaria) Flower Extract, Cucumis Sativus (Cucumber) Fruit Extract, Althaea Officinalis Root Extract, DL-Alpha Tocopheryl Acetate (Vitamin E) and Fragrance.
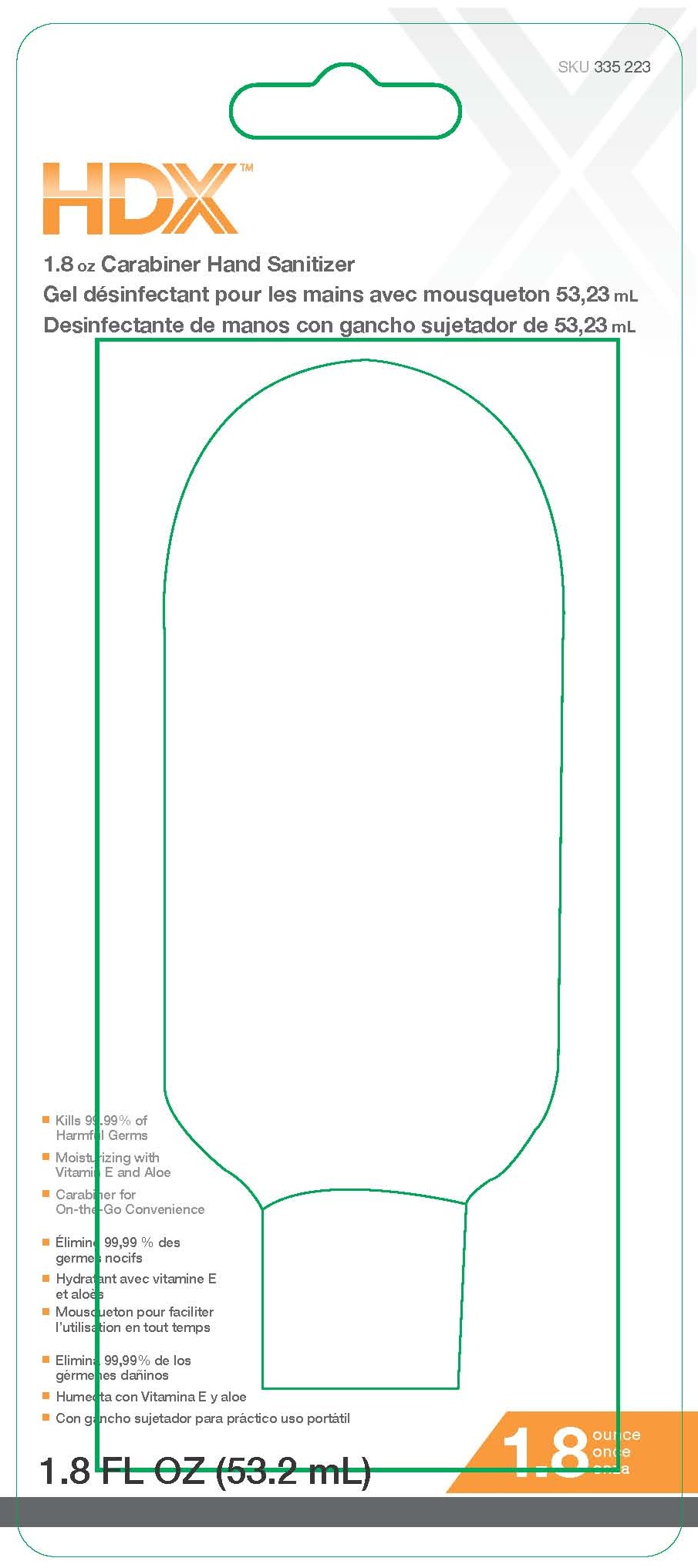
HDX
8 OZ HAND SANITIZER
- Kills 99.99% of Harmful Germs
- Moisturizing with Vitamin E and Aloe
- Carabiner for On-the-Go Convenient Use




| HAND SANITIZER
ethyl alcohol gel |
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| OTC monograph not final | part333E | 02/14/2012 | |
| Labeler - HOME DEPOT U.S.A. INC. (783266950) |
Revised: 02/2012 HOME DEPOT U.S.A. INC.