DRAMAMINE FOR KIDS
-
dimenhydrinate tablet, chewable
Medtech Products Inc.
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
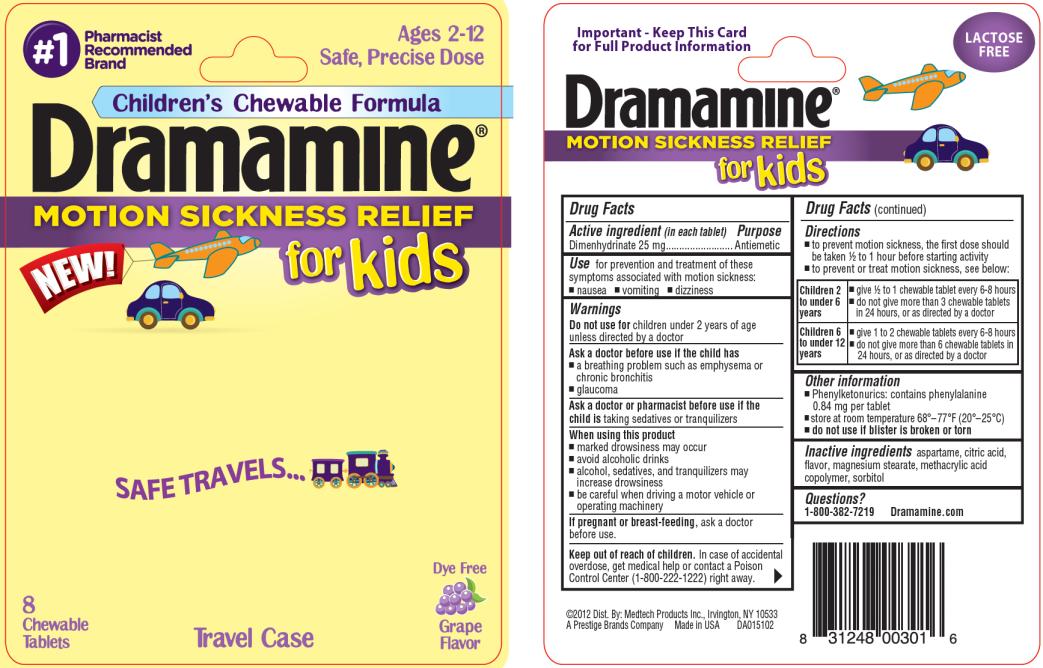
Use
for prevention and treatment of these symptoms associated with motion sickness:
- nausea
- vomiting
- dizziness
Warnings
Ask a doctor before use if the child has
- a breathing problem such as emphysema or chronic bronchitis
- glaucoma
Directions
- to prevent motion sickness, the first dose should be taken ½ to 1 hour before starting activity
- to prevent or treat motion sickness, see below:
| Children 2
to under 6 years |
|
| Children 6
to under 12 years |
|
Other information
- Phenylketonurics: contains phenylalanine 0.84 mg per tablet
- store at room temperature 68º - 77ºF (20º - 25ºC)
- do not use if blister is broken or torn
Inactive ingredients
aspartame, citric acid, flavor, magnesium stearate, methacrylic acid copolymer, sorbitol
PRINCIPAL DISPLAY PANEL
#1Pharmacist Recommended Brand
Ages 2 – 12
Safe, Precise Dose
Children’s Chewable Formula
Dramamine MOTION SICKNESS RELIEF for Kids
8 Chewable Tablets
Dye Free Grape Flavor
| DRAMAMINE FOR KIDS
dimenhydrinate tablet, chewable |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| OTC MONOGRAPH FINAL | part336 | 02/15/2012 | |
| Labeler - Medtech Products Inc. (122715688) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| Contract Pharmacal Corporation | 057795122 | MANUFACTURE | |
Revised: 02/2012 Medtech Products Inc.