AMLODIPINE BESYLATE - amlodipine besylate tablet
Aurobindo Pharma Limited
----------
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use amlodipine besylate safely and effectively. See full prescribing information for amlodipine besylate tablets, USP.
Amlodipine Besylate Tablets, USP for oral administration Initial U.S. Approval: 1987 INDICATIONS AND USAGEAmlodipine besylate tablets, USP are a calcium channel blocker and may be used alone or in combination with other antihypertensive and antianginal agents for the treatment of: DOSAGE AND ADMINISTRATION
Important Limitation: Doses in excess of 5 mg daily have not been studied in pediatric patients. (2.2) DOSAGE FORMS AND STRENGTHS
CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
ADVERSE REACTIONSMost common adverse reactions are headache and edema which occurred in a dose related manner. Other adverse experiences not dose related but reported with an incidence >1% are headache, fatigue, nausea, abdominal pain, and somnolence. (6) To report SUSPECTED ADVERSE REACTIONS, contact Aurobindo Pharma USA, Inc. at 1-866-850-2876 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. DRUG INTERACTIONS
USE IN SPECIFIC POPULATIONS
See 17 for FDA-approved patient labeling. Revised: 12/2011 |
FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
1.1 Hypertension
Amlodipine besylate tablets, USP are indicated for the treatment of hypertension. They may be used alone or in combination with other antihypertensive agents.
1.2 Coronary Artery Disease (CAD)
Chronic Stable Angina
Amlodipine besylate tablets, USP are indicated for the symptomatic treatment of chronic stable angina. Amlodipine besylate tablets, USP may be used alone or in combination with other antianginal agents.
Vasospastic Angina (Prinzmetal’s or Variant Angina)
Amlodipine besylate tablets, USP are indicated for the treatment of confirmed or suspected vasospastic angina. Amlodipine besylate tablets, USP may be used as monotherapy or in combination with other antianginal agents.
Angiographically Documented CAD
In patients with recently documented CAD by angiography and without heart failure or an ejection fraction <40%, amlodipine besylate tablets, USP are indicated to reduce the risk of hospitalization due to angina and to reduce the risk of a coronary revascularization procedure.
2 DOSAGE AND ADMINISTRATION
2.1 Adults
The usual initial antihypertensive oral dose of amlodipine besylate tablets is 5 mg once daily with a maximum dose of 10 mg once daily.
Small, fragile, or elderly patients, or patients with hepatic insufficiency may be started on 2.5 mg once daily and this dose may be used when adding amlodipine besylate tablets to other antihypertensive therapy.
Adjust dosage according to each patient’s need. In general, titration should proceed over 7 to 14 days so that the physician can fully assess the patient’s response to each dose level. Titration may proceed more rapidly, however, if clinically warranted, provided the patient is assessed frequently.
The recommended dose for chronic stable or vasospastic angina is 5 to 10 mg, with the lower dose suggested in the elderly and in patients with hepatic insufficiency. Most patients will require 10 mg for adequate effect [see Adverse Reactions (6)].
The recommended dose range for patients with coronary artery disease is 5 to 10 mg once daily. In clinical studies, the majority of patients required 10 mg [see Clinical Studies (14.4)].
2.2 Children
The effective antihypertensive oral dose in pediatric patients ages 6 to 17 years is 2.5 mg to 5 mg once daily. Doses in excess of 5 mg daily have not been studied in pediatric patients [see Clinical Pharmacology (12.4), Clinical Studies (14.1)].
3 DOSAGE FORMS AND STRENGTHS
- 2.5 mg (amlodipine besylate equivalent to 2.5 mg of amlodipine per tablet) are white to off-white, flat, bevel edged round uncoated tablets, debossed with ‘C’ on one side and ‘57’ on the other side.
- 5 mg (amlodipine besylate equivalent to 5 mg of amlodipine per tablet) are white to off-white, flat, bevel edged barrel shaped uncoated tablets, debossed with ‘C’ on one side and ‘58’ on the other side.
- 10 mg (amlodipine besylate equivalent to 10 mg of amlodipine per tablet) are white to off-white, flat, bevel edged round uncoated tablets, debossed with ‘C’ on one side and ‘59’ on the other side.
4 CONTRAINDICATIONS
Amlodipine besylate tablets are contraindicated in patients with known sensitivity to amlodipine.
5 WARNINGS AND PRECAUTIONS
5.1 Hypotension
Symptomatic hypotension is possible, particularly in patients with severe aortic stenosis. Because of the gradual onset of action, acute hypotension is unlikely.
5.2 Increased Angina or Myocardial Infarction
Worsening angina and acute myocardial infarction can develop after starting or increasing the dose of amlodipine besylate, particularly in patients with severe obstructive coronary artery disease.
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Amlodipine besylate has been evaluated for safety in more than 11,000 patients in U.S. and foreign clinical trials. In general, treatment with amlodipine besylate was well-tolerated at doses up to 10 mg daily. Most adverse reactions reported during therapy with amlodipine besylate were of mild or moderate severity. In controlled clinical trials directly comparing amlodipine besylate (N=1730) at doses up to 10 mg to placebo (N=1250), discontinuation of amlodipine besylate due to adverse reactions was required in only about 1.5% of patients and was not significantly different from placebo (about 1%). The most common side effects are headache and edema. The incidence (%) of side effects that occurred in a dose related manner are as follows:
| Adverse Event | 2.5 mg N=275 | 5 mg N=296 | 10 mg N=268 | Placebo N=520 |
|---|---|---|---|---|
| Edema
| 1.8
| 3
| 10.8
| 0.6
|
| Dizziness
| 1.1
| 3.4
| 3.4
| 1.5
|
| Flushing
| 0.7
| 1.4
| 2.6
| 0
|
| Palpitation | 0.7
| 1.4
| 4.5
| 0.6 |
Other adverse experiences that were not clearly dose related but were reported with an incidence greater than 1% in placebo-controlled clinical trials include the following:
| Placebo-Controlled Studies | ||
|---|---|---|
| Amlodipine Besylate (%) (N=1730) | Placebo (%) (N=1250) |
|
| Headache | 7.3 | 7.8 |
| Fatigue | 4.5 | 2.8 |
| Nausea | 2.9 | 1.9 |
| Abdominal Pain | 1.6 | 0.3 |
| Somnolence | 1.4 | 0.6 |
For several adverse experiences that appear to be drug and dose related, there was a greater incidence in women than men associated with amlodipine treatment as shown in the following table:
| Adverse Event | Amlodipine Besylate | Placebo | ||
|---|---|---|---|---|
| Male=% (N=1218) | Female=% (N=512) | Male=% (N=914) | Female=% (N=336) |
|
| Edema | 5.6 | 14.6 | 1.4 | 5.1 |
| Flushing | 1.5 | 4.5 | 0.3 | 0.9 |
| Palpitations | 1.4 | 3.3 | 0.9 | 0.9 |
| Somnolence | 1.3 | 1.6 | 0.8 | 0.3 |
The following events occurred in <1% but >0.1% of patients in controlled clinical trials or under conditions of open trials or marketing experience where a causal relationship is uncertain; they are listed to alert the physician to a possible relationship:
Cardiovascular: arrhythmia (including ventricular tachycardia and atrial fibrillation), bradycardia, chest pain, hypotension, peripheral ischemia, syncope, tachycardia, postural dizziness, postural hypotension, vasculitis.
Central and Peripheral Nervous System: hypoesthesia, neuropathy peripheral, paresthesia, tremor, vertigo.
Gastrointestinal: anorexia, constipation, dyspepsia,1 dysphagia, diarrhea, flatulence, pancreatitis, vomiting, gingival hyperplasia.
General: allergic reaction, asthenia,1 back pain, hot flushes, malaise, pain, rigors, weight gain, weight decrease.
Musculoskeletal System: arthralgia, arthrosis, muscle cramps,1 myalgia.
Psychiatric: sexual dysfunction (male1and female), insomnia, nervousness, depression, abnormal dreams, anxiety, depersonalization.
Respiratory System: dyspnea,1 epistaxis.
Skin and Appendages: angioedema, erythema multiforme, pruritus,1 rash,1 rash erythematous, rash maculopapular.
Special Senses: abnormal vision, conjunctivitis, diplopia, eye pain, tinnitus.
Urinary System: micturition frequency, micturition disorder, nocturia.
Autonomic Nervous System: dry mouth, sweating increased.
Metabolic and Nutritional: hyperglycemia, thirst.
Hemopoietic: leukopenia, purpura, thrombocytopenia.
1 These events occurred in less than 1% in placebo-controlled trials, but the incidence of these side effects was between 1% and 2% in all multiple dose studies.
The following events occurred in <0.1% of patients: cardiac failure, pulse irregularity, extrasystoles, skin discoloration, urticaria, skin dryness, alopecia, dermatitis, muscle weakness, twitching, ataxia, hypertonia, migraine, cold and clammy skin, apathy, agitation, amnesia, gastritis, increased appetite, loose stools, coughing, rhinitis, dysuria, polyuria, parosmia, taste perversion, abnormal visual accommodation, and xerophthalmia.
Other reactions occurred sporadically and cannot be distinguished from medications or concurrent disease states such as myocardial infarction and angina.
Amlodipine besylate therapy has not been associated with clinically significant changes in routine laboratory tests. No clinically relevant changes were noted in serum potassium, serum glucose, total triglycerides, total cholesterol, HDL cholesterol, uric acid, blood urea nitrogen, or creatinine.
In the CAMELOT and PREVENT studies [see Clinical Studies (14.4)], the adverse event profile was similar to that reported previously (see above), with the most common adverse event being peripheral edema.
6.2 Postmarketing Experience
Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
The following postmarketing event has been reported infrequently where a causal relationship is uncertain: gynecomastia. In postmarketing experience, jaundice and hepatic enzyme elevations (mostly consistent with cholestasis or hepatitis), in some cases severe enough to require hospitalization, have been reported in association with use of amlodipine.
Amlodipine besylate has been used safely in patients with chronic obstructive pulmonary disease, well-compensated congestive heart failure, coronary artery disease, peripheral vascular disease, diabetes mellitus, and abnormal lipid profiles.
7 DRUG INTERACTIONS
7.1 In Vitro Data
In vitro data indicate that amlodipine besylate has no effect on the human plasma protein binding of digoxin, phenytoin, warfarin, and indomethacin.
7.2 Cimetidine
Coadministration of amlodipine besylate with cimetidine did not alter the pharmacokinetics of amlodipine besylate.
7.3 Grapefruit Juice
Coadministration of 240 mL of grapefruit juice with a single oral dose of amlodipine 10 mg in 20 healthy volunteers had no significant effect on the pharmacokinetics of amlodipine.
7.4 Magnesium and Aluminum Hydroxide Antacid
Coadministration of a magnesium and aluminum hydroxide antacid with a single dose of amlodipine besylate had no significant effect on the pharmacokinetics of amlodipine besylate.
7.5 Sildenafil
A single 100 mg dose of sildenafil in subjects with essential hypertension had no effect on the pharmacokinetic parameters of amlodipine besylate. When amlodipine besylate and sildenafil were used in combination, each agent independently exerted its own blood pressure lowering effect.
7.6 Atorvastatin
Coadministration of multiple 10 mg doses of amlodipine besylate with 80 mg of atorvastatin resulted in no significant change in the steady-state pharmacokinetic parameters of atorvastatin.
7.7 Simvastatin
Coadministration of multiple doses of 10 mg of amlodipine with 80 mg simvastatin resulted in a 77% increase in exposure to simvastatin compared to simvastatin alone. Limit the dose of simvastatin in patients on amlodipine to 20 mg daily.
7.8 Digoxin
Coadministration of amlodipine besylate with digoxin did not change serum digoxin levels or digoxin renal clearance in normal volunteers.
7.9 Ethanol
Single and multiple 10 mg doses of amlodipine besylate had no significant effect on the pharmacokinetics of ethanol.
7.10 Warfarin
Coadministration of amlodipine besylate with warfarin did not change the warfarin prothrombin response time.
7.11 CYP3A4 Inhibitors
Coadministration of a 180 mg daily dose of diltiazem with 5 mg amlodipine in elderly hypertensive patients resulted in a 60% increase in amlodipine systemic exposure. Erythromycin coadministration in healthy volunteers did not significantly change amlodipine systemic exposure. However, strong inhibitors of CYP3A4 (e.g., ketoconazole, itraconazole, ritonavir) may increase the plasma concentrations of amlodipine to a greater extent. Monitor for symptoms of hypotension and edema when amlodipine is coadministered with CYP3A4 inhibitors.
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Category C
There are no adequate and well-controlled studies in pregnant women. Amlodipine should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
No evidence of teratogenicity or other embryo/fetal toxicity was found when pregnant rats and rabbits were treated orally with amlodipine maleate at doses up to 10 mg amlodipine/kg/day (respectively, 8 times2 and 23 times2 the maximum recommended human dose of 10 mg on a mg/m2 basis) during their respective periods of major organogenesis. However, litter size was significantly decreased (by about 50%) and the number of intrauterine deaths was significantly increased (about 5-fold) in rats receiving amlodipine maleate at a dose equivalent to 10 mg amlodipine/kg/day for 14 days before mating and throughout mating and gestation. Amlodipine maleate has been shown to prolong both the gestation period and the duration of labor in rats at this dose.
2 Based on patient weight of 50 kg.
8.3 Nursing Mothers
It is not known whether amlodipine is excreted in human milk. In the absence of this information, it is recommended that nursing be discontinued while amlodipine besylate is administered.
8.4 Pediatric Use
Effect of amlodipine besylate on blood pressure in patients less than 6 years of age is not known.
8.5 Geriatric Use
Clinical studies of amlodipine besylate did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy. Elderly patients have decreased clearance of amlodipine with a resulting increase of AUC of approximately 40 to 60%, and a lower initial dose may be required [see Dosage and Administration (2.1)].
10 OVERDOSAGE
Overdosage might be expected to cause excessive peripheral vasodilation with marked hypotension and possibly a reflex tachycardia. In humans, experience with intentional overdosage of amlodipine besylate is limited.
Single oral doses of amlodipine maleate equivalent to 40 mg amlodipine/kg and 100 mg amlodipine/kg in mice and rats, respectively, caused deaths. Single oral amlodipine maleate doses equivalent to 4 or more mg amlodipine/kg or higher in dogs (11 or more times the maximum recommended human dose on a mg/m2 basis) caused a marked peripheral vasodilation and hypotension.
If massive overdose should occur, initiate active cardiac and respiratory monitoring. Frequent blood pressure measurements are essential. Should hypotension occur, provide cardiovascular support including elevation of the extremities and the judicious administration of fluids. If hypotension remains unresponsive to these conservative measures, consider administration of vasopressors (such as phenylephrine) with attention to circulating volume and urine output. As amlodipine besylate is highly protein bound, hemodialysis is not likely to be of benefit.
11 DESCRIPTION
Amlodipine besylate is the besylate salt of amlodipine, a long-acting calcium channel blocker.
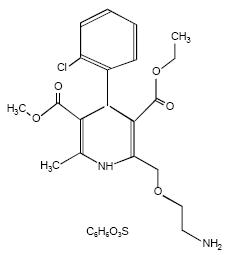
Amlodipine besylate is chemically described as 3-Ethyl-5-methyl (±)-2-[(2-aminoethoxy)methyl]-4-(2-chlorophenyl)-1,4-dihydro-6-methyl-3,5-pyridinedicarboxylate, monobenzenesulphonate. Its molecular formula is C20H25CIN2O5•C6H6O3S, and its structural formula is:
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Amlodipine is a dihydropyridine calcium antagonist (calcium ion antagonist or slow-channel blocker) that inhibits the transmembrane influx of calcium ions into vascular smooth muscle and cardiac muscle. Experimental data suggest that amlodipine binds to both dihydropyridine and nondihydropyridine binding sites. The contractile processes of cardiac muscle and vascular smooth muscle are dependent upon the movement of extracellular calcium ions into these cells through specific ion channels. Amlodipine inhibits calcium ion influx across cell membranes selectively, with a greater effect on vascular smooth muscle cells than on cardiac muscle cells. Negative inotropic effects can be detected in vitro but such effects have not been seen in intact animals at therapeutic doses. Serum calcium concentration is not affected by amlodipine. Within the physiologic pH range, amlodipine is an ionized compound (pKa=8.6), and its kinetic interaction with the calcium channel receptor is characterized by a gradual rate of association and dissociation with the receptor binding site, resulting in a gradual onset of effect.
Amlodipine is a peripheral arterial vasodilator that acts directly on vascular smooth muscle to cause a reduction in peripheral vascular resistance and reduction in blood pressure.
The precise mechanisms by which amlodipine relieves angina have not been fully delineated, but are thought to include the following:
Exertional Angina: In patients with exertional angina, amlodipine besylate reduces the total peripheral resistance (afterload) against which the heart works and reduces the rate pressure product, and thus myocardial oxygen demand, at any given level of exercise.
Vasospastic Angina: Amlodipine besylate has been demonstrated to block constriction and restore blood flow in coronary arteries and arterioles in response to calcium, potassium epinephrine, serotonin, and thromboxane A2 analog in experimental animal models and in human coronary vessels in vitro. This inhibition of coronary spasm is responsible for the effectiveness of amlodipine besylate in vasospastic (Prinzmetal’s or variant) angina.
12.2 Pharmacodynamics
Hemodynamics: Following administration of therapeutic doses to patients with hypertension, amlodipine besylate produces vasodilation resulting in a reduction of supine and standing blood pressures. These decreases in blood pressure are not accompanied by a significant change in heart rate or plasma catecholamine levels with chronic dosing. Although the acute intravenous administration of amlodipine decreases arterial blood pressure and increases heart rate in hemodynamic studies of patients with chronic stable angina, chronic oral administration of amlodipine in clinical trials did not lead to clinically significant changes in heart rate or blood pressures in normotensive patients with angina.
With chronic once daily oral administration, antihypertensive effectiveness is maintained for at least 24 hours. Plasma concentrations correlate with effect in both young and elderly patients. The magnitude of reduction in blood pressure with amlodipine besylate is also correlated with the height of pretreatment elevation; thus, individuals with moderate hypertension (diastolic pressure 105 to 114 mmHg) had about a 50% greater response than patients with mild hypertension (diastolic pressure 90 to 104 mmHg). Normotensive subjects experienced no clinically significant change in blood pressures (+1/–2 mmHg).
In hypertensive patients with normal renal function, therapeutic doses of amlodipine besylate resulted in a decrease in renal vascular resistance and an increase in glomerular filtration rate and effective renal plasma flow without change in filtration fraction or proteinuria.
As with other calcium channel blockers, hemodynamic measurements of cardiac function at rest and during exercise (or pacing) in patients with normal ventricular function treated with amlodipine besylate have generally demonstrated a small increase in cardiac index without significant influence on dP/dt or on left ventricular end diastolic pressure or volume. In hemodynamic studies, amlodipine besylate has not been associated with a negative inotropic effect when administered in the therapeutic dose range to intact animals and man, even when coadministered with beta-blockers to man. Similar findings, however, have been observed in normal or well-compensated patients with heart failure with agents possessing significant negative inotropic effects.
Electrophysiologic Effects: Amlodipine besylate does not change sinoatrial nodal function or atrioventricular conduction in intact animals or man. In patients with chronic stable angina, intravenous administration of 10 mg did not significantly alter A-H and H-V conduction and sinus node recovery time after pacing. Similar results were obtained in patients receiving amlodipine besylate and concomitant beta-blockers. In clinical studies in which amlodipine besylate was administered in combination with beta-blockers to patients with either hypertension or angina, no adverse effects on electrocardiographic parameters were observed. In clinical trials with angina patients alone, amlodipine besylate therapy did not alter electrocardiographic intervals or produce higher degrees of AV blocks.
12.3 Pharmacokinetics and Metabolism
After oral administration of therapeutic doses of amlodipine besylate, absorption produces peak plasma concentrations between 6 and 12 hours. Absolute bioavailability has been estimated to be between 64 and 90%. The bioavailability of amlodipine besylate is not altered by the presence of food.
Amlodipine is extensively (about 90%) converted to inactive metabolites via hepatic metabolism with 10% of the parent compound and 60% of the metabolites excreted in the urine. Ex vivo studies have shown that approximately 93% of the circulating drug is bound to plasma proteins in hypertensive patients. Elimination from the plasma is biphasic with a terminal elimination half-life of about 30 to 50 hours. Steady-state plasma levels of amlodipine are reached after 7 to 8 days of consecutive daily dosing.
The pharmacokinetics of amlodipine are not significantly influenced by renal impairment. Patients with renal failure may therefore receive the usual initial dose.
Elderly patients and patients with hepatic insufficiency have decreased clearance of amlodipine with a resulting increase in AUC of approximately 40 to 60%, and a lower initial dose may be required. A similar increase in AUC was observed in patients with moderate to severe heart failure.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Rats and mice treated with amlodipine maleate in the diet for up to two years, at concentrations calculated to provide daily dosage levels of 0.5, 1.25, and 2.5 amlodipine mg/kg/day, showed no evidence of a carcinogenic effect of the drug. For the mouse, the highest dose was, on a mg/m2 basis, similar to the maximum recommended human dose of 10 mg amlodipine/day.3 For the rat, the highest dose was, on a mg/m2 basis, about twice the maximum recommended human dose.3
Mutagenicity studies conducted with amlodipine maleate revealed no drug related effects at either the gene or chromosome level.
There was no effect on the fertility of rats treated orally with amlodipine maleate (males for 64 days and females for 14 days prior to mating) at doses up to 10 mg amlodipine/kg/day (8 times the maximum recommended human dose3 of 10 mg/day on a mg/m2 basis).
3 Based on patient weight of 50 kg
14 CLINICAL STUDIES
14.1 Effects in Hypertension
Adult Patients
The antihypertensive efficacy of amlodipine besylate has been demonstrated in a total of 15 double-blind, placebo-controlled, randomized studies involving 800 patients on amlodipine besylate and 538 on placebo. Once daily administration produced statistically significant placebo-corrected reductions in supine and standing blood pressures at 24 hours postdose, averaging about 12/6 mmHg in the standing position and 13/7 mmHg in the supine position in patients with mild to moderate hypertension. Maintenance of the blood pressure effect over the 24-hour dosing interval was observed, with little difference in peak and trough effect. Tolerance was not demonstrated in patients studied for up to 1 year. The 3 parallel, fixed dose, dose response studies showed that the reduction in supine and standing blood pressures was dose-related within the recommended dosing range. Effects on diastolic pressure were similar in young and older patients. The effect on systolic pressure was greater in older patients, perhaps because of greater baseline systolic pressure. Effects were similar in black patients and in white patients.
Pediatric Patients
Two hundred sixty-eight hypertensive patients aged 6 to 17 years were randomized first to amlodipine besylate 2.5 or 5 mg once daily for 4 weeks and then randomized again to the same dose or to placebo for another 4 weeks. Patients receiving 2.5 mg or 5 mg at the end of 8 weeks had significantly lower systolic blood pressure than those secondarily randomized to placebo. The magnitude of the treatment effect is difficult to interpret, but it is probably less than 5 mmHg systolic on the 5 mg dose and 3.3 mmHg systolic on the 2.5 mg dose. Adverse events were similar to those seen in adults.
14.2 Effects in Chronic Stable Angina
The effectiveness of 5 to 10 mg/day of amlodipine besylate in exercise-induced angina has been evaluated in 8 placebo-controlled, double-blind clinical trials of up to 6 weeks duration involving 1038 patients (684 amlodipine besylate, 354 placebo) with chronic stable angina. In 5 of the 8 studies, significant increases in exercise time (bicycle or treadmill) were seen with the 10 mg dose. Increases in symptom-limited exercise time averaged 12.8% (63 sec) for amlodipine besylate 10 mg, and averaged 7.9% (38 sec) for amlodipine besylate 5 mg. Amlodipine besylate 10 mg also increased time to 1 mm ST segment deviation in several studies and decreased angina attack rate. The sustained efficacy of amlodipine besylate in angina patients has been demonstrated over long-term dosing. In patients with angina, there were no clinically significant reductions in blood pressures (4/1 mmHg) or changes in heart rate (+0.3 bpm).
14.3 Effects in Vasospastic Angina
In a double-blind, placebo-controlled clinical trial of 4 weeks duration in 50 patients, amlodipine besylate therapy decreased attacks by approximately 4/week compared with a placebo decrease of approximately 1/week (p<0.01). Two of 23 amlodipine besylate and 7 of 27 placebo patients discontinued from the study due to lack of clinical improvement.
14.4 Effects in Documented Coronary Artery Disease
In PREVENT, 825 patients with angiographically documented coronary artery disease were randomized to amlodipine besylate (5 to 10 mg once daily) or placebo and followed for 3 years. Although the study did not show significance on the primary objective of change in coronary luminal diameter as assessed by quantitative coronary angiography, the data suggested a favorable outcome with respect to fewer hospitalizations for angina and revascularization procedures in patients with CAD.
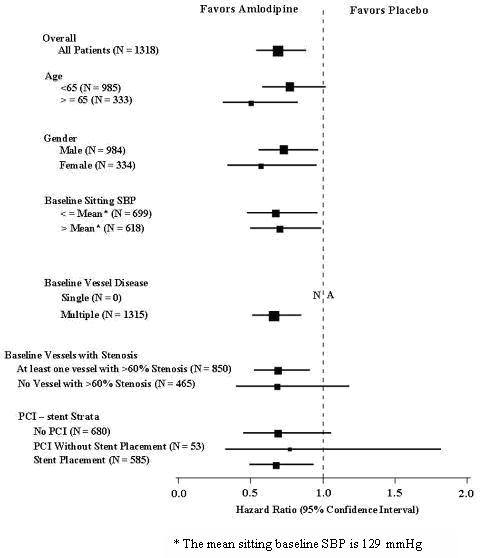
CAMELOT enrolled 1318 patients with CAD recently documented by angiography, without left main coronary disease and without heart failure or an ejection fraction <40%. Patients (76% males, 89% Caucasian, 93% enrolled at U.S. sites, 89% with a history of angina, 52% without PCI, 4% with PCI and no stent, and 44% with a stent) were randomized to double-blind treatment with either amlodipine besylate (5 to 10 mg once daily) or placebo in addition to standard care that included aspirin (89%), statins (83%), beta-blockers (74%), nitroglycerin (50%), anticoagulants (40%), and diuretics (32%), but excluded other calcium channel blockers. The mean duration of follow-up was 19 months. The primary endpoint was the time to first occurrence of one of the following events: hospitalization for angina pectoris, coronary revascularization, myocardial infarction, cardiovascular death, resuscitated cardiac arrest, hospitalization for heart failure, stroke/TIA, or peripheral vascular disease. A total of 110 (16.6%) and 151 (23.1%) first events occurred in the amlodipine besylate and placebo groups, respectively, for a hazard ratio of 0.691 (95% CI: 0.54 to 0.884, p = 0.003). The primary endpoint is summarized in Figure 1 below. The outcome of this study was largely derived from the prevention of hospitalizations for angina and the prevention of revascularization procedures (see Table 1). Effects in various subgroups are shown in Figure 2.
In an angiographic substudy (n=274) conducted within CAMELOT, there was no significant difference between amlodipine and placebo on the change of atheroma volume in the coronary artery as assessed by intravascular ultrasound.
Figure 1 - Kaplan-Meier Analysis of Composite Clinical Outcomes for Amlodipine Besylate versus Placebo
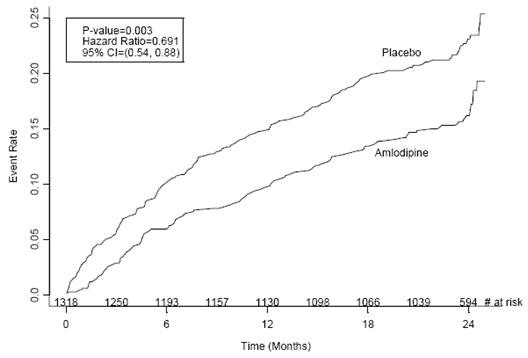
Figure 2 - Effects on Primary Endpoint of Amlodipine Besylate versus Placebo across Sub-Groups
| Clinical Outcomes N (%) | Amlodipine Besylate (N=663) | Placebo (N=655) | Risk Reduction (p-value) |
|---|---|---|---|
| * Total patients with these events |
|||
| Composite CV Endpoint
| 110
(16.6) | 151
(23.1) | 31%
(0.003) |
| Hospitalization for Angina* | 51 (7.7) | 84 (12.8) | 42% (0.002) |
| Coronary Revascularization* | 78 (11.8) | 103 (15.7) | 27% (0.033) |
14.5 Studies in Patients with Heart Failure
Amlodipine besylate has been compared to placebo in four 8 to 12 week studies of patients with NYHA Class II/III heart failure, involving a total of 697 patients. In these studies, there was no evidence of worsened heart failure based on measures of exercise tolerance, NYHA classification, symptoms, or left ventricular ejection fraction. In a long-term (follow-up at least 6 months, mean 13.8 months) placebo-controlled mortality/morbidity study of amlodipine besylate 5 to 10 mg in 1153 patients with NYHA Classes III (n=931) or IV (n=222) heart failure on stable doses of diuretics, digoxin, and ACE inhibitors, amlodipine besylate had no effect on the primary endpoint of the study which was the combined endpoint of all-cause mortality and cardiac morbidity (as defined by life-threatening arrhythmia, acute myocardial infarction, or hospitalization for worsened heart failure), or on NYHA classification, or symptoms of heart failure. Total combined all-cause mortality and cardiac morbidity events were 222/571 (39%) for patients on amlodipine besylate and 246/583 (42%) for patients on placebo; the cardiac morbid events represented about 25% of the endpoints in the study.
Another study (PRAISE-2) randomized patients with NYHA Class III (80%) or IV (20%) heart failure without clinical symptoms or objective evidence of underlying ischemic disease, on stable doses of ACE inhibitors (99%), digitalis (99%), and diuretics (99%), to placebo (n=827) or amlodipine besylate (n=827) and followed them for a mean of 33 months. There was no statistically significant difference between amlodipine besylate and placebo in the primary endpoint of all-cause mortality (95% confidence limits from 8% reduction to 29% increase on amlodipine besylate). With amlodipine besylate there were more reports of pulmonary edema.
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 2.5 mg Tablets
Amlodipine Besylate Tablets USP, 2.5 mg (amlodipine besylate equivalent to 2.5 mg of amlodipine per tablet) are white to off-white, flat, bevel edged round uncoated tablets, debossed with ‘C’ on one side and ‘57’ on the other side, and is supplied as follows:
Bottles of 30 NDC 65862-101-30
Bottles of 90 NDC 65862-101-90
Bottles of 500 NDC 65862-101-05
Bottles of 1000 NDC 65862-101-99
16.2 5 mg Tablets
Amlodipine Besylate Tablets USP, 5 mg (amlodipine besylate equivalent to 5 mg of amlodipine per tablet) are white to off-white, flat, bevel edged barrel shaped uncoated tablets, debossed with ‘C’ on one side and ‘58’ on the other side, and is supplied as follows:
Bottles of 30 NDC 65862-102-30
Bottles of 90 NDC 65862-102-90
Bottles of 500 NDC 65862-102-05
Bottles of 1000 NDC 65862-102-99
16.3 10 mg Tablets
Amlodipine Besylate Tablets USP, 10 mg (amlodipine besylate equivalent to 10 mg of amlodipine per tablet) are white to off-white, flat, bevel edged round uncoated tablets, debossed with ‘C’ on one side and ‘59’ on the other side, and is supplied as follows:
Bottles of 30 NDC 65862-103-30
Bottles of 90 NDC 65862-103-90
Bottles of 500 NDC 65862-103-05
Bottles of 1000 NDC 65862-103-99
16.4 Storage
Store at 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature].
Dispense in tight, light-resistant containers as defined in the USP.
Manufactured for:
Aurobindo Pharma USA, Inc.
2400 Route 130 North
Dayton, NJ 08810
Manufactured by:
Aurobindo Pharma Limited
Hyderabad-500 072, India
Revised: 12/2011
Patient Information
Amlodipine Besylate Tablets, USP
Read this information carefully before you start taking amlodipine besylate tablets and each time you refill your prescription. There may be new information. This information does not replace talking with your doctor. If you have any questions about amlodipine besylate tablets, ask your doctor. Your doctor will know if amlodipine besylate tablets are right for you.
What are amlodipine besylate tablets?
Amlodipine besylate tablets are a type of medicine known as a calcium channel blocker (CCB). They are used to treat high blood pressure (hypertension) and a type of chest pain called angina. They can be used alone or with other medicines to treat these conditions.
High Blood Pressure (hypertension)
High blood pressure comes from blood pushing too hard against your blood vessels. Amlodipine besylate tablets relax your blood vessels, which lets your blood flow more easily and help lower your blood pressure. Drugs that lower blood pressure lower your risk of having a stroke or heart attack.
Angina
Angina is a pain or discomfort that keeps coming back when part of your heart does not get enough blood. Angina feels like a pressing or squeezing pain, usually in your chest under the breastbone. Sometimes you can feel it in your shoulders, arms, neck, jaws, or back. Amlodipine besylate tablets can relieve this pain.
Who should not use amlodipine besylate tablets?
Do not use amlodipine besylate tablets if you are allergic to amlodipine (the active ingredient in amlodipine besylate tablets), or to the inactive ingredients. Your doctor or pharmacist can give you a list of these ingredients.
What should I tell my doctor before taking amlodipine besylate tablets?
Tell your doctor about any prescription and non-prescription medicines you are taking, including natural or herbal remedies. Tell your doctor if you:
- ever had heart disease
- ever had liver problems
- are pregnant, or plan to become pregnant. Your doctor will decide if amlodipine besylate tablets are the best treatment for you.
- are breastfeeding. Do not breastfeed while taking amlodipine besylate tablets. You can stop breastfeeding or take a different medicine.
How should I take amlodipine besylate tablets?
- Take amlodipine besylate tablets once a day, with or without food.
- It may be easier to take your dose if you do it at the same time every day, such as with breakfast or dinner, or at bedtime. Do not take more than one dose of amlodipine besylate tablets at a time.
- If you miss a dose, take it as soon as you remember. Do not take amlodipine besylate tablets if it has been more than 12 hours since you missed your last dose. Wait and take the next dose at your regular time.
- Other medicines: You can use nitroglycerin and amlodipine besylate tablets together. If you take nitroglycerin for angina, don’t stop taking it while you are taking amlodipine besylate tablets.
- While you are taking amlodipine besylate tablets, do not stop taking your other prescription medicines, including any other blood pressure medicines, without talking to your doctor.
- If you took too much amlodipine besylate tablets, call your doctor or Poison Control Center, or go to the nearest hospital emergency room right away.
What should I avoid while taking amlodipine besylate tablets?
- Do not breastfeed. It is not known if amlodipine besylate will pass through your milk.
- Do not start any new prescription or non-prescription medicines or supplements, unless you check with your doctor first.
What are the possible side effects of amlodipine besylate tablets?
Amlodipine besylate tablets may cause the following side effects. Most side effects are mild or moderate:
- headache
- swelling of your legs or ankles
- tiredness, extreme sleepiness
- stomach pain, nausea
- dizziness
- flushing (hot or warm feeling in your face)
- arrhythmia (irregular heartbeat)
- heart palpitations (very fast heartbeat)
It is rare, but when you first start taking amlodipine besylate tablets or increase your dose, you may have a heart attack or your angina may get worse. If that happens, call your doctor right away or go directly to a hospital emergency room.
Tell your doctor if you are concerned about any side effects you experience. These are not all the possible side effects of amlodipine besylate tablets. For a complete list, ask your doctor or pharmacist.
How do I store amlodipine besylate tablets?
Keep amlodipine besylate tablets away from children. Store amlodipine besylate tablets at 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. Keep amlodipine besylate tablets out of the light. Do not store in the bathroom. Keep amlodipine besylate tablets in a dry place.
General advice about amlodipine besylate tablets
Sometimes, doctors will prescribe a medicine for a condition that is not written in the patient information leaflets. Only use amlodipine besylate tablets the way your doctor told you to. Do not give amlodipine besylate tablets to other people, even if they have the same symptoms you have. They may harm them.
You can ask your pharmacist or doctor for information about amlodipine besylate tablets, or you can call 1-866-850-2876.
Manufactured for:
Aurobindo Pharma USA, Inc.
2400 Route 130 North
Dayton, NJ 08810
Manufactured by:
Aurobindo Pharma Limited
Hyderabad-500 072, India
Revised: 12/2011
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 2.5 mg (30 Tablet Bottle)
NDC 65862-101-30
Amlodipine Besylate Tablets, USP
2.5 mg*
Rx only 30 Tablets
AUROBINDO

| AMLODIPINE BESYLATE
amlodipine besylate tablet |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| AMLODIPINE BESYLATE
amlodipine besylate tablet |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| AMLODIPINE BESYLATE
amlodipine besylate tablet |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Labeler - Aurobindo Pharma Limited (650082092) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Aurobindo Pharma Limited | 918917642 | MANUFACTURE, ANALYSIS | |
Revised: 1/2012
Document Id: 2e8e384c-6306-4597-8ef4-3e140899a56a
Set id: b6f298ba-2d7e-4a3c-9edb-8b60aba716d6
Version: 7
Effective Time: 20120119
Aurobindo Pharma Limited

