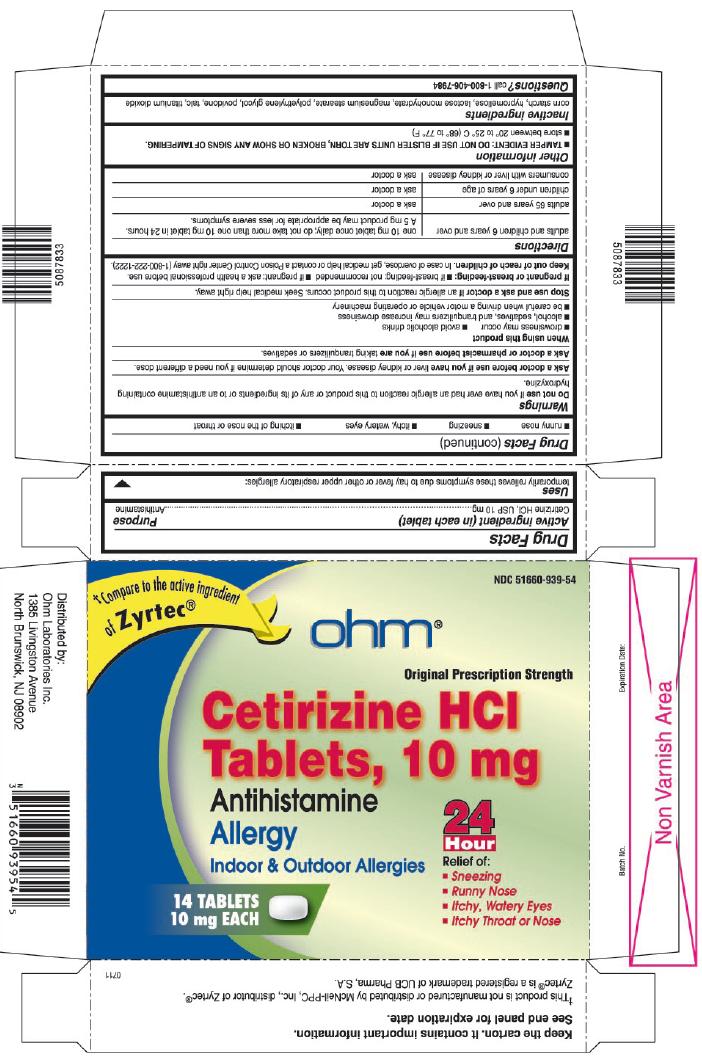
CETIRIZINE HYDROCHLORIDE
-
cetirizine hydrochloride tablet
Ohm Laboratories Inc.
----------
USES
temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- runny nose
- sneezing
- itchy, watery eyes
- itching of the nose or throat
WARNINGS
Do not use
if you have ever had an allergic reaction to this product or any of its ingredients or to an antihistamine containing hydroxyzine.
Ask a doctor before use if you have
liver or kidney disease. Your doctor should determine if you need a different dose.
When using this product
- drowsiness may occur
- avoid alcoholic drinks
- alcohol, sedatives, and tranquilizers may increase drowsiness
- be careful when driving a motor vehicle or operating machinery
Stop use and ask a doctor if
an allergic reaction to this product occurs. Seek medical help right away.
DIRECTIONS
adults and children 6 years and over: one 10 mg tablet once daily; do not take more than one 10 mg tablet in 24 hours. A 5 mg product may be appropriate for less severe symptoms.
adults 65 years and over: ask a doctor
OTHER INFORMATION
- TAMPER EVIDENT: DO NOT USE IF IMPRINTED SEAL IS BROKEN OR MISSING FROM BOTTLE.(for bottle cartons/stand-alone labels only)
- TAMPER EVIDENT: DO NOT USE IF BLISTER UNITS ARE TORN, BROKEN OR SHOW ANY SIGNS OF TAMPERING.(for blister cartons only)
- store between 20° to 25° C (68° to 77° F)
INACTIVE INGREDIENTS
corn starch, hypromellose, lactose monohydrate, magnesium stearate, polyethylene glycol, povidone, talc, titanium dioxide
PRINCIPAL DISPLAY PANEL
†Compare to the active ingredient of Zyrtec®
Original Prescription Strength
- Sneezing
- Runny Nose
- Itchy, Watery Eyes
- Itchy Throat or Nose
†This product is not manufactured or distributed by McNeil-PPC, Inc., distributor of Zyrtec®. Zyrtec® is a registered trademark of UCB Pharma, S.A.
| CETIRIZINE HYDROCHLORIDE
cetirizine hydrochloride tablet |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| ANDA | ANDA077498 | 12/27/2007 | |
| Labeler - Ohm Laboratories Inc. (184769029) |
| Registrant - Ranbaxy Pharmaceuticals Inc. (937890044) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| Ohm Laboratories Inc. | 184769029 | manufacture | |
Revised: 01/2012 Ohm Laboratories Inc.