GENUINE TRIPLE ANTIBIOTIC
-
bacitracin zinc,
neomycin sulfate and
polymyxin b sulfate ointment
Genuine First Aid LLC
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
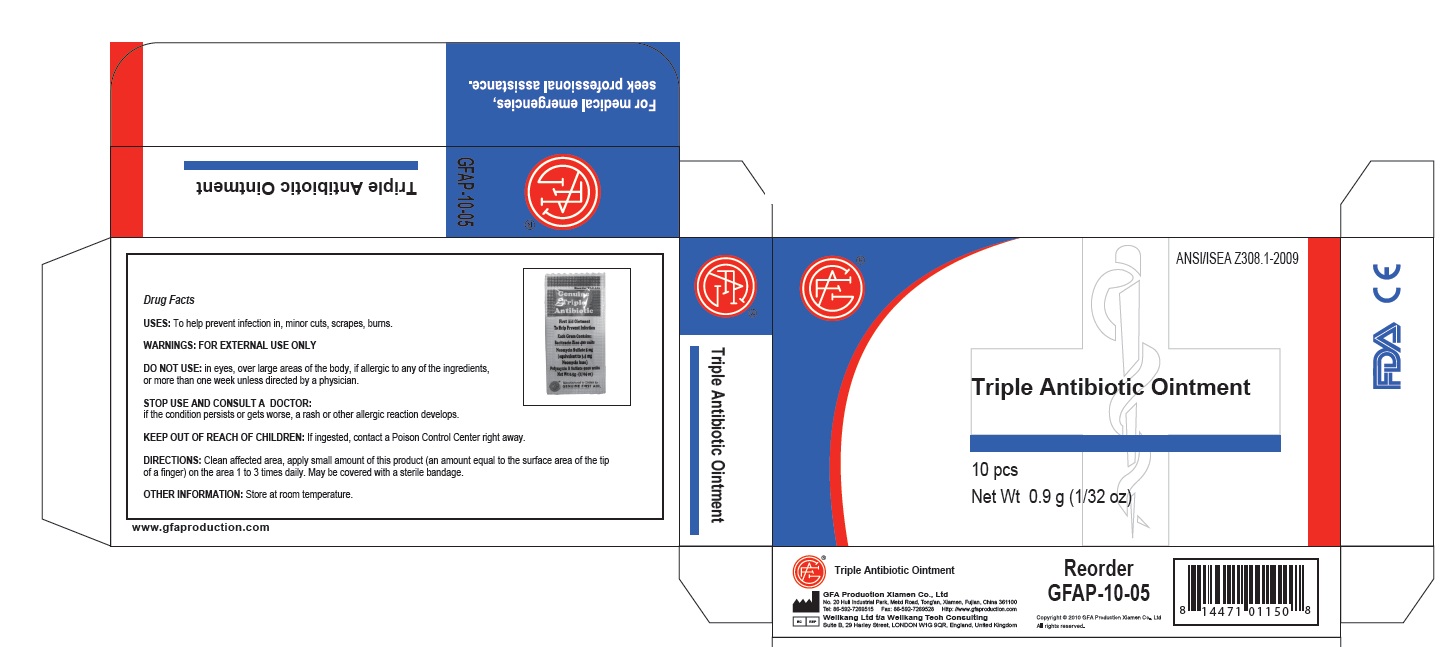
Active Ingredients
Active Ingredient: .........Bacitracin Zinc 400 units
Neomycin Sulfate 5mg ( equivalent to 3.5 mg Neomycin base)
Polymyxin B Sulfate 5000 units
Do not use: in eyes; over large areas of the body;
If allergic to any of the ingredients; for more than one week unless directed by a physician.
Stop use and consult a doctor:
if the condition persists or gets worse; a rash or other allergic reaction develops
Directions
Directions: clean affected area; apply small amount of this product (an amount equal to the surface area of the tip of a finger) on the area 1 to 3 times daily; may be covered with a sterile bandage
Genuine Triple Antibiotic
First Aid Ointment
To Help Prevent Infection
Each Gram Contains:
Bacitracin Zinc 400 units
Neomycin Sulfate 5 mg
(equivalent to 3.5 mg
Neomycin base)
Polymyxin B Sulfate 5000 units
Net Wt. 0.5g ; (1/64 oz)
Manufactured in CHINA for
GENUINE FIRST AID.
Triple Antibiotic Ointment 10pcs
Net wt. 0.9g (1/32oz)
100
Triple Antibiotic
| GENUINE TRIPLE ANTIBIOTIC
bacitracin zinc,neomycin sulfate,polymyxin b sulfate ointment |
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| OTC monograph final | part333B | 02/16/2010 | |
| Labeler - Genuine First Aid LLC (619609857) |
Revised: 11/2011 Genuine First Aid LLC