ELIDEL
-
pimecrolimus cream
Valeant Pharmaceuticals North America LLC
----------
Prescribing Information
See WARNINGS, boxed WARNING concerning long-term safety of topical calcineurin inhibitors.
DESCRIPTION
ELIDEL® (pimecrolimus) Cream 1% contains the compound pimecrolimus, the immunosuppressant 33-epichloro-derivative of the macrolactam ascomycin.
Chemically, pimecrolimus is (1R,9S,12S,13R,14S,17R, 18E,21S,23S,24R,25S,27R)-12-[(1E)-2-{(1R,3R,4S)-4-chloro-3-methoxycyclohexyl}-1-methylvinyl]-17-ethyl-1, 14-dihydroxy-23,25-dimethoxy-13,19,21,27-tetramethyl-11,28-dioxa-4-aza-tricyclo[22.3.1.0 4,9]octacos-18-ene-2,3,10,16-tetraone.
The compound has the empirical formula C 43H68CINO11 and the molecular weight of 810.47. The structural formula is
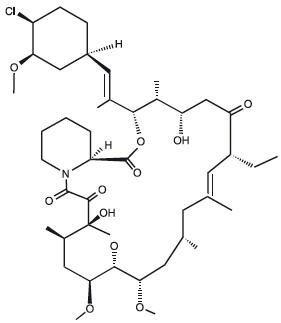
Pimecrolimus is a white to off-white fine crystalline powder. It is soluble in methanol and ethanol and insoluble in water.
Each gram of ELIDEL Cream 1% contains 10 mg of pimecrolimus in a whitish cream base of benzyl alcohol, cetyl alcohol, citric acid, mono- and di-glycerides, oleyl alcohol, propylene glycol, sodium cetostearyl sulphate, sodium hydroxide, stearyl alcohol, triglycerides, and water.
CLINICAL PHARMACOLOGY
Mechanism of Action/Pharmacodynamics
The mechanism of action of pimecrolimus in atopic dermatitis is not known. While the following have been observed, the clinical significance of these observations in atopic dermatitis is not known. It has been demonstrated that pimecrolimus binds with high affinity to macrophilin-12 (FKBP-12) and inhibits the calcium-dependent phosphatase, calcineurin. As a consequence, it inhibits T cell activation by blocking the transcription of early cytokines. In particular, pimecrolimus inhibits at nanomolar concentrations Interleukin-2 and interferon gamma (Th1-type) and Interleukin-4 and Interleukin-10 (Th2-type) cytokine synthesis in human T cells. In addition, pimecrolimus prevents the release of inflammatory cytokines and mediators from mast cells in vitro after stimulation by antigen/IgE.
Pharmacokinetics
Absorption
In adult patients (n=52) being treated for atopic dermatitis [13%-62% Body Surface Area (BSA) involvement] for periods up to a year, a maximum pimecrolimus concentration of 1.4 ng/mL was observed among those subjects with detectable blood levels. In the majority of samples in adult (91%; 1,244/1,362) subjects, blood concentrations of pimecrolimus were below 0.5 ng/mL. Data on blood levels of pimecrolimus measured in pediatric patients are described below in Special Populations, Pediatrics.
Distribution
Laboratory in vitro plasma protein binding studies using equilibrium gel filtration have shown that 99.5% of pimecrolimus in plasma is bound to proteins over the pimecrolimus concentration range of 2-100 ng/mL tested. The major fraction of pimecrolimus in plasma appears to be bound to various lipoproteins. As with other topical calcineurin inhibitors, it is not known whether pimecrolimus is absorbed into cutaneous lymphatic vessels or in regional lymph nodes.
Metabolism
Following the administration of a single oral radiolabeled dose of pimecrolimus numerous circulating O-demethylation metabolites were seen. Studies with human liver microsomes indicate that pimecrolimus is metabolized in vitro by the CYP3A sub-family of metabolizing enzymes. No evidence of skin mediated drug metabolism was identified in vivo using the minipig or in vitro using stripped human skin.
Elimination
Based on the results of the aforementioned radiolabeled study, following a single oral dose of pimecrolimus ~81% of the administered radioactivity was recovered, primarily in the feces (78.4%) as metabolites. Less than 1% of the radioactivity found in the feces was due to unchanged pimecrolimus.
Special Populations
Pediatrics
The systemic exposure to pimecrolimus from ELIDEL® (pimecrolimus) Cream 1% was investigated in 28 pediatric patients with atopic dermatitis (20%-80% BSA involvement) between the ages of 8 months-14 yrs. Following twice daily application for three weeks, blood concentrations of pimecrolimus were <2 ng/mL with 60% (96/161) of the blood samples having blood concentration below the limit of quantification (0.5 ng/mL). However, the children (23 children out of the total 28 children investigated) had at least one detectable blood level as compared to the adults (12 adults out of the total 52 adults investigated) over a 3-week treatment period. Due to the erratic nature of the blood levels observed, no correlation could be made between amount of cream, degree of BSA involvement, and blood concentrations. In general, the blood concentrations measured in adult atopic dermatitis patients were comparable to those seen in the pediatric population.
In a second group of 30 pediatric patients aged 3-23 months with 10%-92% BSA involvement, following twice daily application for three weeks, blood concentrations of pimecrolimus were <2.6 ng/mL with 65% (75/116) of the blood samples having blood concentration below 0.5ng/mL, and 27% (31/116) below the limit of quantification (0.1 ng/mL) for these studies.
Overall, a higher proportion of detectable blood levels was seen in the pediatric patient population as compared to adult population. This increase in the absolute number of positive blood levels may be due to the larger surface area to body mass ratio seen in these younger subjects. In addition, a higher incidence of upper respiratory symptoms/infections was also seen relative to the older age group in the PK studies. At this time, a causal relationship between these findings and ELIDEL use cannot be ruled out.
ELIDEL Cream is not indicated for use in children less than 2 years of age (see INDICATIONS AND USAGE, WARNINGS, boxed WARNING, and PRECAUTIONS, Pediatric Use).
CLINICAL STUDIES
Three randomized, double-blind, vehicle-controlled, multicenter, Phase 3 studies were conducted in 589 pediatric patients ages 3 months-17 years old to evaluate ELIDEL® (pimecrolimus) Cream 1% for the treatment of mild to moderate atopic dermatitis. Two of the three trials support the use of ELIDEL Cream in patients 2 years and older with mild to moderate atopic dermatitis (see PRECAUTIONS, Pediatric Use). Three other trials in 1,619 pediatric and adult patients provided additional data regarding the safety of ELIDEL Cream in the treatment of atopic dermatitis. Two of these other trials were vehicle-controlled with optional sequential use of a medium potency topical corticosteroid in pediatric patients and one trial was an active comparator trial in adult patients with atopic dermatitis (see PRECAUTIONS, Pediatric Use and ADVERSE REACTIONS).
Two identical 6-week, randomized, vehicle-controlled, multi-center, Phase 3 trials were conducted to evaluate ELIDEL Cream for the treatment of mild to moderate atopic dermatitis. A total of 403 pediatric patients 2-17 years old were included in the studies. The male/female ratio was approximately 50% and 29% of the patients were African American. At study entry, 59% of patients had moderate disease and the mean body surface area (BSA) affected was 26%. About 75% of patients had atopic dermatitis affecting the face and/or neck region. In these studies, patients applied either ELIDEL Cream or vehicle cream twice daily to 5% to 96% of their BSA for up to 6 weeks. At endpoint, based on the physician's global evaluation of clinical response, 35% of patients treated with ELIDEL Cream were clear or almost clear of signs of atopic dermatitis compared to only 18% of vehicle-treated patients. More ELIDEL patients (57%) had mild or no pruritus at 6 weeks compared to vehicle patients (34%). The improvement in pruritus occurred in conjunction with the improvement of the patients' atopic dermatitis.
In these two 6-week studies of ELIDEL, the combined efficacy results at endpoint are as follows:
| % Patients | ||
|---|---|---|
| Elidel® (N= 267) | Vehicle (N= 136) | |
| Global Assessment | ||
| Clear | 28 (10%) | 5 (4%) |
| Clear or Almost Clear | 93 (35%) | 25 (18%) |
| Clear to Mild Disease | 180 (67%) | 55 (40%) |
In the two pediatric studies that independently support the use of ELIDEL Cream in mild to moderate atopic dermatitis, a significant treatment effect was seen by day 15. Of the key signs of atopic dermatitis, erythema, infiltration/papulation, lichenification, and excoriations, erythema and infiltration/papulation were reduced at day 8 when compared to vehicle.
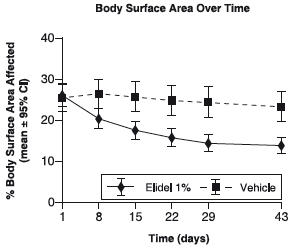
The following graph depicts the time course of improvement in the percent body surface area affected as a result of treatment with ELIDEL Cream in 2-17 year olds.
Figure 1
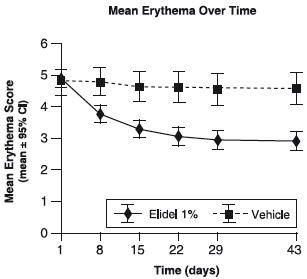
The following graph shows the time course of improvement in erythema as a result of treatment with ELIDEL Cream in 2-17 year olds.
Figure 2
INDICATIONS AND USAGE
ELIDEL® (pimecrolimus) Cream 1% is indicated as second-line therapy for the short-term and non-continuous chronic treatment of mild to moderate atopic dermatitis in non-immunocompromised adults and children 2 years of age and older, who have failed to respond adequately to other topical prescription treatments, or when those treatments are not advisable.
ELIDEL Cream is not indicated for use in children less than 2 years of age (see WARNINGS, boxed WARNING, and PRECAUTIONS, Pediatric Use).
CONTRAINDICATIONS
ELIDEL® (pimecrolimus) Cream 1% is contraindicated in individuals with a history of hypersensitivity to pimecrolimus or any of the components of the cream.
WARNINGS
WARNING
Long-term Safety of Topical Calcineurin Inhibitors Has Not Been Established
Although a causal relationship has not been established, rare cases of malignancy (e.g., skin and lymphoma) have been reported in patients treated with topical calcineurin inhibitors, including ELIDEL Cream.
Therefore:
- Continuous long-term use of topical calcineurin inhibitors, including ELIDEL Cream, in any age group should be avoided, and application limited to areas of involvement with atopic dermatitis.
- ELIDEL Cream is not indicated for use in children less than 2 years of age.
Prolonged systemic use of calcineurin inhibitors for sustained immunosuppression in animal studies and transplant patients following systemic administration has been associated with an increased risk of infections, lymphomas, and skin malignancies. These risks are associated with the intensity and duration of immunosuppression.
Based on this information and the mechanism of action, there is a concern about a potential risk with the use of topical calcineurin inhibitors, including ELIDEL Cream. While a causal relationship has not been established, rare cases of skin malignancy and lymphoma have been reported in patients treated with topical calcineurin inhibitors, including ELIDEL Cream. Therefore:
- ELIDEL Cream should not be used in immunocompromised adults and children.
- If signs and symptoms of atopic dermatitis do not improve within 6 weeks, patients should be re-examined by their healthcare provider and their diagnosis be confirmed (see PRECAUTIONS).
- The safety of ELIDEL Cream has not been established beyond one year of non-continuous use.
(See CLINICAL PHARMACOLOGY, WARNINGS, boxed WARNING, PRECAUTIONS, INDICATIONS AND USAGE, and DOSAGE AND ADMINISTRATION.)
PRECAUTIONS
General
The use of ELIDEL Cream should be avoided on malignant or pre-malignant skin conditions. Malignant or pre-malignant skin conditions, such as cutaneous T-cell lymphoma (CTCL), can present as dermatitis.
ELIDEL Cream should not be used in patients with Netherton's Syndrome or other skin diseases where there is the potential for increased systemic absorption of pimecrolimus . The safety of ELIDEL Cream has not been established in patients with generalized erythroderma.
The use of ELIDEL Cream may cause local symptoms such as skin burning (burning sensation, stinging, soreness) or pruritus. Localized symptoms are most common during the first few days of ELIDEL Cream application and typically improve as the lesions of atopic dermatitis resolve (see ADVERSE REACTIONS).
Bacterial and Viral Skin Infections
Before commencing treatment with ELIDEL Cream, bacterial or viral infections at treatment sites should be resolved. Studies have not evaluated the safety and efficacy of ELIDEL Cream in the treatment of clinically infected atopic dermatitis.
While patients with atopic dermatitis are predisposed to superficial skin infections including eczema herpeticum (Kaposi's varicelliform eruption), treatment with ELIDEL Cream may be independently associated with an increased risk of varicella zoster virus infection (chicken pox or shingles), herpes simplex virus infection, or eczema herpeticum.
In clinical studies, 15/1,544 (1%) cases of skin papilloma (warts) were observed in patients using ELIDEL Cream. The youngest patient was age 2 and the oldest was age 12. In cases where there is worsening of skin papillomas or they do not respond to conventional therapy, discontinuation of ELIDEL Cream should be considered until complete resolution of the warts is achieved.
Patients with Lymphadenopathy
In clinical studies, 14/1,544 (0.9%) cases of lymphadenopathy were reported while using ELIDEL Cream. These cases of lymphadenopathy were usually related to infections and noted to resolve upon appropriate antibiotic therapy. Of these 14 cases, the majority had either a clear etiology or were known to resolve. Patients who receive ELIDEL Cream and who develop lymphadenopathy should have the etiology of their lymphadenopathy investigated. In the absence of a clear etiology for the lymphadenopathy, or in the presence of acute infectious mononucleosis, ELIDEL Cream should be discontinued. Patients who develop lymphadenopathy should be monitored to ensure that the lymphadenopathy resolves.
Information for Patients
(See Medication Guide.)
Patients using ELIDEL Cream should receive the following information and instructions:
What is the most important information a patient should know about ELIDEL Cream?
The safety of using ELIDEL Cream for a long period of time is not known. A very small number of people who have used ELIDEL Cream have had cancer (for example, skin or lymphoma). However, a link with ELIDEL Cream use has not been shown. Because of this concern:
- A patient should not use ELIDEL Cream continuously for a long time.
- ELIDEL Cream should be used only on areas of skin that have eczema.
- ELIDEL Cream is not for use on a child under 2 years old.
How should a patient use ELIDEL Cream?
- A patient should use ELIDEL Cream exactly as prescribed.
- A patient should use ELIDEL Cream only on areas of skin that have eczema.
- A patient should use ELIDEL Cream for short periods, and if needed, treatment may be repeated with breaks in between.
- A patient should stop ELIDEL Cream when the signs and symptoms of eczema, such as itching, rash, and redness go away, or as directed by the physician.
- A patient should follow the physician's advice if symptoms of eczema return after a treatment with ELIDEL Cream.
- A patient should contact the physician if:
- symptoms get worse with ELIDEL Cream
- the patient gets a skin infection
- if burning on the skin is severe or lasts for more than one week
- if eye irritation does not go away
- symptoms do not improve after 6 weeks of treatment
To apply ELIDEL Cream:
- A patient or caregiver should wash their hands before using ELIDEL Cream. When applying ELIDEL Cream after a bath or shower, the skin should be dry.
- A patient or caregiver should apply a thin layer of ELIDEL Cream only to the affected skin areas, twice a day, as directed by the physician.
- A patient or caregiver should use the smallest amount of ELIDEL Cream needed to control the signs and symptoms of eczema.
- Caregivers applying ELIDEL Cream to a patient, or a patient who is not treating the hands should wash their hands with soap and water after applying ELIDEL Cream. This should remove any cream left on the hands.
- A patient should not bathe, shower or swim right after applying ELIDEL Cream. This could wash off the cream.
- A patient can use moisturizers with ELIDEL Cream. They should be sure to check with the physician first about the products that are right for them. Because the skin of patients with eczema can be very dry, it is important they keep up good skin care practices. If a patient uses moisturizers, he or she should apply them after ELIDEL Cream.
What should a patient avoid while using ELIDEL Cream?
- A patient should not use sun lamps, tanning beds, or get treatment with ultraviolet light therapy during treatment with ELIDEL Cream.
- A patient should limit sun exposure during treatment with ELIDEL Cream even when the medicine is not on the skin. If a patient needs to be outdoors after applying ELIDEL Cream, the patient should wear loose fitting clothing that protects the treated area from the sun. The physician should advise the patient about other types of protection from the sun.
- A patient should not cover the skin being treated with bandages, dressings or wraps. A patient can wear normal clothing.
- ELIDEL Cream is for use on the skin only. Do not get ELIDEL Cream in your eyes, nose, mouth, vagina, or rectum (mucous membranes). If you get ELIDEL Cream in any of these areas, burning or irritation can happen. Wipe off any ELIDEL Cream from the affected area and then rinse the area well with cold water. ELIDEL Cream is for external use only.
- A patient should not swallow ELIDEL Cream and should contact the physician if they do.
Drug Interactions
Potential interactions between ELIDEL and other drugs, including immunizations, have not been systematically evaluated. Due to low blood levels of pimecrolimus detected in some patients after topical application, systemic drug interactions are not expected, but cannot be ruled out. The concomitant administration of known CYP3A family of inhibitors in patients with widespread and/or erythrodermic disease should be done with caution. Some examples of such drugs are erythromycin, itraconazole, ketoconazole, fluconazole, calcium channel blockers and cimetidine.
Carcinogenesis, Mutagenesis, Impairment of Fertility
In a 2-year rat dermal carcinogenicity study using ELIDEL Cream, a statistically significant increase in the incidence of follicular cell adenoma of the thyroid was noted in low, mid and high dose male animals compared to vehicle and saline control male animals. Follicular cell adenoma of the thyroid was noted in the dermal rat carcinogenicity study at the lowest dose of 2 mg/kg/day [0.2% pimecrolimus cream; 1.5× the Maximum Recommended Human Dose (MRHD) based on AUC comparisons]. No increase in the incidence of follicular cell adenoma of the thyroid was noted in the oral carcinogenicity study in male rats up to 10 mg/kg/day (66× MRHD based on AUC comparisons). However, oral studies may not reflect continuous exposure or the same metabolic profile as by the dermal route. In a mouse dermal carcinogenicity study using pimecrolimus in an ethanolic solution, no increase in incidence of neoplasms was observed in the skin or other organs up to the highest dose of 4 mg/kg/day (0.32% pimecrolimus in ethanol) 27× MRHD based on AUC comparisons. However, lymphoproliferative changes (including lymphoma) were noted in a 13 week repeat dose dermal toxicity study conducted in mice using pimecrolimus in an ethanolic solution at a dose of 25 mg/kg/day (47× MRHD based on AUC comparisons). No lymphoproliferative changes were noted in this study at a dose of 10 mg/kg/day (17× MRHD based on AUC comparison). However, the latency time to lymphoma formation was shortened to 8 weeks after dermal administration of pimecrolimus dissolved in ethanol at a dose of 100 mg/kg/day (179-217× MRHD based on AUC comparisons).
In a mouse oral (gavage) carcinogenicity study, a statistically significant increase in the incidence of lymphoma was noted in high dose male and female animals compared to vehicle control male and female animals. Lymphomas were noted in the oral mouse carcinogenicity study at a dose of 45 mg/kg/day (258-340× MRHD based on AUC comparisons). No drug-related tumors were noted in the mouse oral carcinogenicity study at a dose of 15 mg/kg/day (60-133× MRHD based on AUC comparisons). In an oral (gavage) rat carcinogenicity study, a statistically significant increase in the incidence of benign thymoma was noted in 10 mg/kg/day pimecrolimus treated male and female animals compared to vehicle control treated male and female animals. In addition, a significant increase in the incidence of benign thymoma was noted in another oral (gavage) rat carcinogenicity study in 5 mg/kg/day pimecrolimus treated male animals compared to vehicle control treated male animals. No drug-related tumors were noted in the rat oral carcinogenicity study at a dose of 1 mg/kg/day male animals (1.1× MRHD based on AUC comparisons) and at a dose of 5 mg/kg/day for female animals (21× MRHD based on AUC comparisons).
In a 52-week dermal photo-carcinogenicity study, the median time to onset of skin tumor formation was decreased in hairless mice following chronic topical dosing with concurrent exposure to UV radiation (40 weeks of treatment followed by 12 weeks of observation) with the ELIDEL Cream vehicle alone. No additional effect on tumor development beyond the vehicle effect was noted with the addition of the active ingredient, pimecrolimus, to the vehicle cream.
A 39-week oral monkey toxicology study was conducted with pimecrolimus doses of 15, 45 and 120 mg/kg/day. A dose dependent increase in expression of immunosuppressive-related lymphoproliferative disorder (IRLD) associated with lymphocryptovirus (a monkey strain of virus related to human Epstein Barr virus) was observed. IRLD in monkeys mirrors what has been noted in human transplant patients after chronic systemic immunosuppressive therapy, post transplantation lymphoproliferative disease (PTLD), after treatment with chronic systemic immunosuppressive therapy. Both IRLD and PTLD can progress to lymphoma, which is dependent on the dose and duration of systemic immunosuppressive therapy. A dose dependent increase in opportunistic infections (a signal of systemic immunosuppression) was also noted in this monkey study. A no observed adverse effect level (NOAEL) for IRLD and opportunistic infections was not established in this study. IRLD occurred at the lowest dose of 15 mg/kg/day for 39 weeks [31× the Maximum Recommended Human Dose (MRHD) of ELIDEL Cream based on AUC comparisons] in this study. A partial recovery from IRLD was noted upon cessation of dosing in this study.
A battery of in vitro genotoxicity tests, including Ames assay, mouse lymphoma L5178Y assay, and chromosome aberration test in V79 Chinese hamster cells and an in vivo mouse micronucleus test revealed no evidence for a mutagenic or clastogenic potential for the drug.
An oral fertility and embryofetal developmental study in rats revealed estrus cycle disturbances, post-implantation loss and reduction in litter size at the 45 mg/kg/day dose (38× MRHD based on AUC comparisons). No effect on fertility in female rats was noted at 10 mg/kg/day (12× MRHD based on AUC comparisons). No effect on fertility in male rats was noted at 45 mg/kg/day (23× MRHD based on AUC comparisons), which was the highest dose tested in this study.
A second oral fertility and embryofetal developmental study in rats revealed reduced testicular and epididymal weights, reduced testicular sperm counts and motile sperm for males and estrus cycle disturbances, decreased corpora lutea, decreased implantations and viable fetuses for females at 45 mg/kg/day dose (123× MRHD for males and 192× MRHD for females based on AUC comparisons). No effect on fertility in female rats was noted at 10 mg/kg/day (5× MRHD based on AUC comparisons). No effect on fertility in male rats was noted at 2 mg/kg/day (0.7× MRHD based on AUC comparisons).
Pregnancy
Teratogenic Effects
Pregnancy Category C
There are no adequate and well-controlled studies of topically administered pimecrolimus in pregnant women. The experience with ELIDEL Cream when used by pregnant women is too limited to permit assessment of the safety of its use during pregnancy.
In dermal embryofetal developmental studies, no maternal or fetal toxicity was observed up to the highest practicable doses tested, 10 mg/kg/day (1% pimecrolimus cream) in rats (0.14× MRHD based on body surface area) and 10 mg/kg/day (1% pimecrolimus cream) in rabbits (0.65× MRHD based on AUC comparisons). The 1% pimecrolimus cream was administered topically for 6 hours/day during the period of organogenesis in rats and rabbits (gestational days 6-21 in rats and gestational days 6-20 in rabbits).
A second dermal embryofetal development study was conducted in rats using pimecrolimus cream applied dermally to pregnant rats (1 g cream/kg body weight of 0.2%, 0.6% and 1.0% pimecrolimus cream) from gestation day 6 to 17 at doses of 2, 6, and 10 mg/kg/day with daily exposure of approximately 22 hours. No maternal, reproductive, or embryo-fetal toxicity attributable to pimecrolimus was noted at 10 mg/kg/day (0.66× MRHD based on AUC comparisons), the highest dose evaluated in this study. No teratogenicity was noted in this study at any dose.
A combined oral fertility and embryofetal developmental study was conducted in rats and an oral embryofetal developmental study was conducted in rabbits. Pimecrolimus was administered during the period of organogenesis (2 weeks prior to mating until gestational day 16 in rats, gestational days 6-18 in rabbits) up to dose levels of 45 mg/kg/day in rats and 20 mg/kg/day in rabbits. In the absence of maternal toxicity, indicators of embryofetal toxicity (post-implantation loss and reduction in litter size) were noted at 45 mg/kg/day (38× MRHD based on AUC comparisons) in the oral fertility and embryofetal developmental study conducted in rats. No malformations in the fetuses were noted at 45 mg/kg/day (38× MRHD based on AUC comparisons) in this study. No maternal toxicity, embryotoxicity or teratogenicity were noted in the oral rabbit embryofetal developmental toxicity study at 20 mg/kg/day (3.9× MRHD based on AUC comparisons), which was the highest dose tested in this study.
A second oral embryofetal development study was conducted in rats. Pimecrolimus was administered during the period of organogenesis (gestational days 6 – 17) at doses of 2, 10 and 45 mg/kg/day. Maternal toxicity, embryolethality and fetotoxicity were noted at 45 mg/kg/day (271× MRHD based on AUC comparisons). A slight increase in skeletal variations that were indicative of delayed skeletal ossification was also noted at this dose. No maternal toxicity, embryolethality or fetotoxicity were noted at 10 mg/kg/day (16× MRHD based on AUC comparisons). No teratogenicity was noted in this study at any dose.
A second oral embryofetal development study was conducted in rabbits. Pimecrolimus was administered during the period of organogenesis (gestational days 7 – 20) at doses of 2, 6 and 20 mg/kg/day. Maternal toxicity, embryotoxicity and fetotoxicity were noted at 20 mg/kg/day (12× MRHD based on AUC comparisons). A slight increase in skeletal variations that were indicative of delayed skeletal ossification was also noted at this dose. No maternal toxicity, embryotoxicity or fetotoxicity were noted at 6 mg/kg/ day (5× MRHD based on AUC comparisons). No teratogenicity was noted in this study at any dose.
An oral peri- and post-natal developmental study was conducted in rats. Pimecrolimus was administered from gestational day 6 through lactational day 21 up to a dose level of 40 mg/kg/day. Only 2 of 22 females delivered live pups at the highest dose of 40 mg/kg/day. Postnatal survival, development of the F1 generation, their subsequent maturation and fertility were not affected at 10 mg/kg/day (12× MRHD based on AUC comparisons), the highest dose evaluated in this study.
Pimecrolimus was transferred across the placenta in oral rat and rabbit embryofetal developmental studies.
There are, however, no adequate and well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, this drug should be used only if clearly needed during pregnancy.
Nursing Mothers
It is not known whether this drug is excreted in human milk. Because of the potential for serious adverse reactions in nursing infants from pimecrolimus, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Pediatric Use
ELIDEL Cream is not indicated for use in children less than 2 years of age.
The long-term safety and effects of ELIDEL Cream on the developing immune system are unknown (see WARNINGS, boxed WARNING, and INDICATIONS AND USAGE).
Three Phase 3 pediatric studies were conducted involving 1,114 patients 2-17 years of age. Two studies were 6-week randomized vehicle-controlled studies with a 20-week open-label phase and one was a vehicle-controlled (up to 1 year) safety study with the option for sequential topical corticosteroid use. Of these patients 542 (49%) were 2-6 years of age. In the short-term studies, 11% of ELIDEL patients did not complete these studies and 1.5% of ELIDEL patients discontinued due to adverse events. In the one-year study, 32% of ELIDEL patients did not complete this study and 3% of ELIDEL patients discontinued due to adverse events. Most discontinuations were due to unsatisfactory therapeutic effect.
The most common local adverse event in the short-term studies of ELIDEL Cream in pediatric patients ages 2-17 was application site burning (10% vs. 13% vehicle); the incidence in the long-term study was 9% ELIDEL vs. 7% vehicle (see ADVERSE REACTIONS). Adverse events that were more frequent (>5%) in patients treated with ELIDEL Cream compared to vehicle were headache (14% vs. 9%) in the short-term trial. Nasopharyngitis (26% vs. 21%), influenza (13% vs. 4%), pharyngitis (8% vs. 3%), viral infection (7% vs. 1%), pyrexia (13% vs. 5%), cough (16% vs. 11%), and headache (25% vs. 16%) were increased over vehicle in the 1-year safety study (see ADVERSE REACTIONS). In 843 patients ages 2-17 years treated with ELIDEL Cream, 9 (0.8%) developed eczema herpeticum (5 on ELIDEL Cream alone and 4 on ELIDEL Cream used in sequence with corticosteroids). In 211 patients on vehicle alone, there were no cases of eczema herpeticum. The majority of adverse events were mild to moderate in severity.
Two Phase 3 studies were conducted involving 436 infants age 3 months-23 months. One 6-week randomized vehicle-controlled study with a 20-week open-label phase and one safety study, up to one year, were conducted. In the 6-week study, 11% of ELIDEL and 48% of vehicle patients did not complete this study; no patient in either group discontinued due to adverse events. Infants on ELIDEL Cream had an increased incidence of some adverse events compared to vehicle. In the 6-week vehicle-controlled study these adverse events included pyrexia (32% vs. 13% vehicle), URI (24% vs. 14%), nasopharyngitis (15% vs. 8%), gastroenteritis (7% vs. 3%), otitis media (4% vs. 0%), and diarrhea (8% vs. 0%). In the open-label phase of the study, for infants who switched to ELIDEL Cream from vehicle, the incidence of the above-cited adverse events approached or equaled the incidence of those patients who remained on ELIDEL Cream. In the 6 month safety data, 16% of ELIDEL and 35% of vehicle patients discontinued early and 1.5% of ELIDEL and 0% of vehicle patients discontinued due to adverse events. Infants on ELIDEL Cream had a greater incidence of some adverse events as compared to vehicle. These included pyrexia (30% vs. 20%), URI (21% vs. 17%), cough (15% vs. 9%), hypersensitivity (8% vs. 2%), teething (27% vs. 22%), vomiting (9% vs. 4%), rhinitis (13% vs. 9%), viral rash (4% vs. 0%), rhinorrhea (4% vs. 0%), and wheezing (4% vs. 0%).
ADVERSE REACTIONS
No phototoxicity and no photoallergenicity were detected in clinical studies with 24 and 33 normal volunteers, respectively. In human dermal safety studies, ELIDEL® (pimecrolimus) Cream 1% did not induce contact sensitization or cumulative irritation.
In a one-year safety study in pediatric patients age 2-17 years old involving sequential use of ELIDEL Cream and a topical corticosteroid, 43% of ELIDEL patients and 68% of vehicle patients used corticosteroids during the study. Corticosteroids were used for more than 7 days by 34% of ELIDEL patients and 54% of vehicle patients. An increased incidence of impetigo, skin infection, superinfection (infected atopic dermatitis), rhinitis, and urticaria were found in the patients that had used ELIDEL Cream and topical corticosteroid sequentially as compared to ELIDEL Cream alone.
In 3 randomized, double-blind vehicle-controlled pediatric studies and one active-controlled adult study, 843 and 328 patients respectively, were treated with ELIDEL Cream. In these clinical trials, 48 (4%) of the 1,171 ELIDEL patients and 13 (3%) of 408 vehicle-treated patients discontinued therapy due to adverse events. Discontinuations for AEs were primarily due to application site reactions, and cutaneous infections. The most common application site reaction was application site burning, which occurred in 8%-26% of patients treated with ELIDEL Cream.
The following table depicts the incidence of adverse events pooled across the 2 identically designed 6-week studies with their open label extensions and the 1-year safety study for pediatric patients ages 2-17. Data from the adult active-controlled study is also included in this table. Adverse events are listed regardless of relationship to study drug.
Two cases of septic arthritis have been reported in infants less than one year of age in clinical trials conducted with ELIDEL Cream (n = 2,443). Causality has not been established.
POST-MARKETING EVENTS
The following adverse reactions have been reported in patients using ELIDEL Cream. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
OVERDOSAGE
There has been no experience of overdose with ELIDEL® (pimecrolimus) Cream 1%. If oral ingestion occurs, medical advice should be sought.
DOSAGE AND ADMINISTRATION
- The patient or care giver should apply a thin layer of ELIDEL (pimecrolimus) Cream 1% to the affected skin twice daily. The patient or caregiver should stop using when signs and symptoms (e.g., itch, rash and redness) resolve and should be instructed on what actions to take if symptoms recur.
- If signs and symptoms persist beyond 6 weeks, patients should be re-examined by their health care provider to confirm the diagnosis of atopic dermatitis.
- Continuous long-term use of ELIDEL Cream should be avoided, and application should be limited to areas of involvement with atopic dermatitis.
The safety of ELIDEL Cream under occlusion, which may promote systemic exposure, has not been evaluated. ELIDEL Cream should not be used with occlusive dressings.
HOW SUPPLIED
ELIDEL® (pimecrolimus) Cream 1% is available in tubes of 30 grams, 60 grams, and 100 grams.
| 30 gram tube | NDC 0187-5100-01 |
| 60 gram tube | NDC 0187-5101-02 |
| 100 gram tube | NDC 0187-5102-03 |
| Pediatric Patients* Vehicle-Controlled | Pediatric Patients*
Open-Label | Pediatric Patients* Vehicle-Controlled | Adult Active Comparator | |||
|---|---|---|---|---|---|---|
| (6 weeks) | (20 weeks) | (1 year) | (1 year) | |||
| Elidel® Cream | Vehicle | Elidel® Cream | Elidel® Cream | Vehicle | Elidel® Cream | |
| (N=267) N (%) | (N=136) N (%) | (N=335) N (%) | (N=272) N (%) | (N=75) N (%) | (N=328) N (%) |
|
|
||||||
| At least 1 AE | 182 (68.2%) | 97 (71.3%) | 240 (72.0%) | 230 (84.6%) | 56 (74.7%) | 256 (78.0%) |
| Infections and Infestations | ||||||
| Upper Respiratory Tract Infection NOS | 38 (14.2%) | 18 (13.2%) | 65 (19.4%) | 13 (4.8%) | 6 (8.0%) | 14 (4.3%) |
| Nasopharyngitis | 27 (10.1%) | 10 (7.4%) | 32 (19.6%) | 72 (26.5%) | 16 (21.3%) | 25 (7.6%) |
| Skin Infection NOS | 8 (3.0%) | 9 (5.1%) | 18 (5.4%) | 6 (2.2%) | 3 (4.0%) | 21 (6.4%) |
| Influenza | 8 (3.0%) | 1 (0.7%) | 22 (6.6%) | 36 (13.2%) | 3 (4.0%) | 32 (9.8%) |
| Ear Infection NOS | 6 (2.2%) | 2 (1.5%) | 19 (5.7%) | 9 (3.3%) | 1 (1.3%) | 2 (0.6%) |
| Otitis Media | 6 (2.2%) | 1 (0.7%) | 10 (3.0%) | 8 (2.9%) | 4 (5.3%) | 2 (0.6%) |
| Impetigo | 5 (1.9%) | 3 (2.2%) | 12 (3.6%) | 11 (4.0%) | 4 (5.3%) | 8 (2.4%) |
| Bacterial Infection | 4 (1.5%) | 3 (2.2%) | 4 (1.2%) | 3 (1.1%) | 0 | 6 (1.8%) |
| Folliculitis | 3 (1.1%) | 1 (0.7%) | 3 (0.9%) | 6 (2.2%) | 3 (4.0%) | 20 (6.1%) |
| Sinusitis | 3 (1.1%) | 1 (0.7%) | 11 (3.3%) | 6 (2.2%) | 1 (1.3%) | 2 (0.6%) |
| Pneumonia NOS | 3 (1.1%) | 1 (0.7%) | 5 (1.5%) | 0 | 1 (1.3%) | 1 (0.3%) |
| Pharyngitis NOS | 2 (0.7%) | 2 (1.5%) | 3 (0.9%) | 22 (8.1%) | 2 (2.7%) | 3 (0.9%) |
| Pharyngitis Streptococcal | 2 (0.7%) | 2 (1.5%) | 10 (3.0%) | 0 | <1% | 0 |
| Molluscum Contagiosum | 2 (0.7%) | 0 | 4 (1.2%) | 5 (1.8%) | 0 | 0 |
| Staphylococcal Infection | 1 (0.4%) | 5 (3.7%) | 7 (2.1%) | 0 | <1% | 3 (0.9%) |
| Bronchitis NOS | 1 (0.4%) | 3 (2.2%) | 4 (1.2%) | 29 (10.7%) | 6 (8.0%) | 8 (2.4%) |
| Herpes Simplex | 1 (0.4%) | 0 | 4 (1.2%) | 9 (3.3%) | 2 (2.7%) | 13 (4.0%) |
| Tonsillitis NOS | 1 (0.4%) | 0 | 3 (0.9%) | 17 (6.3%) | 0 | 2 (0.6%) |
| Viral Infection NOS | 2 (0.7%) | 1 (0.7%) | 1 (0.3%) | 18 (6.6%) | 1 (1.3%) | 0 |
| Gastroenteritis NOS | 0 | 3 (2.2%) | 2 (0.6%) | 20 (7.4%) | 2 (2.7%) | 6 (1.8%) |
| Chickenpox | 2 (0.7%) | 0 | 3 (0.9%) | 8 (2.9%) | 3 (4.0%) | 1 (0.3%) |
| Skin Papilloma | 1 (0.4%) | 0 | 2 (0.6%) | 9 (3.3%) | <1% | 0 |
| Tonsillitis Acute NOS | 0 | 0 | 0 | 7 (2.6%) | 0 | 0 |
| Upper Respiratory Tract Infection Viral NOS | 1 (0.4%) | 0 | 3 (0.9%) | 4 (1.5%) | 0 | 1 (0.3%) |
| Herpes Simplex Dermatitis | 0 | 0 | 1 (0.3%) | 4 (1.5%) | 0 | 2 (0.6%) |
| Bronchitis Acute NOS | 0 | 0 | 0 | 4 (1.5%) | 0 | 0 |
| Eye Infection NOS | 0 | 0 | 0 | 3 (1.1%) | <1% | 1 (0.3%) |
| General Disorders and Administration Site Conditions | ||||||
| Application Site Burning | 28 (10.4%) | 17 (12.5%) | 5 (1.5%) | 23 (8.5%) | 5 (6.7%) | 85 (25.9%) |
| Pyrexia | 20 (7.5%) | 12 (8.8%) | 41 (12.2%) | 34 (12.5%) | 4 (5.3%) | 4 (1.2%) |
| Application Site Reaction NOS | 8 (3.0%) | 7 (5.1%) | 7 (2.1%) | 9 (3.3%) | 2 (2.7%) | 48 (14.6%) |
| Application Site Irritation | 8 (3.0%) | 8 (5.9%) | 3 (0.9%) | 1 (0.4%) | 3 (4.0%) | 21 (6.4%) |
| Influenza Like Illness | 1 (0.4%) | 0 | 2 (0.6%) | 5 (1.8%) | 2 (2.7%) | 6 (1.8%) |
| Application Site Erythema | 1 (0.4%) | 0 | 0 | 6 (2.2%) | 0 | 7 (2.1%) |
| Application Site Pruritus | 3 (1.1%) | 2 (1.5%) | 2 (0.6%) | 5 (1.8%) | 0 | 18 (5.5%) |
| Respiratory, Thoracic and Mediastinal Disorders | ||||||
| Cough | 31 (11.6%) | 11 (8.1%) | 31 (9.3%) | 43 (15.8%) | 8 (10.7%) | 8 (2.4%) |
| Nasal Congestion | 7 (2.6%) | 2 (1.5%) | 6 (1.8%) | 4 (1.5%) | 1 (1.3%) | 2 (0.6%) |
| Rhinorrhea | 5 (1.9%) | 1 (0.7%) | 3 (0.9%) | 1 (0.4%) | 1 (1.3%) | 0 |
| Asthma Aggravated | 4 (1.5%) | 3 (2.2%) | 13 (3.9%) | 3 (1.1%) | 1 (1.3%) | 0 |
| Sinus Congestion | 3 (1.1%) | 1 (0.7%) | 2 (0.6%) | <1% | <1% | 3 (0.9%) |
| Rhinitis | 1 (0.4%) | 0 | 5 (1.5%) | 12 (4.4%) | 5 (6.7%) | 7 (2.1%) |
| Wheezing | 1 (0.4%) | 1 (0.7%) | 4 (1.2%) | 2 (0.7%) | <1% | 0 |
| Asthma NOS | 2 (0.7%) | 1 (0.7%) | 11 (3.3%) | 10 (3.7%) | 2 (2.7%) | 8 (2.4%) |
| Epistaxis | 0 | 1 (0.7%) | 0 | 9 (3.3%) | 1 (1.3%) | 1 (0.3%) |
| Dyspnea NOS | 0 | 0 | 0 | 5 (1.8%) | 1 (1.3%) | 2 (0.6%) |
| Gastrointestinal Disorders | ||||||
| Abdominal Pain Upper | 11 (4.1%) | 6 (4.4%) | 10 (3.0%) | 15 (5.5%) | 5 (6.7%) | 1 (0.3%) |
| Sore Throat | 9 (3.4%) | 5 (3.7%) | 15 (5.4%) | 22 (8.1%) | 4 (5.3%) | 12 (3.7%) |
| Vomiting NOS | 8 (3.0%) | 6 (4.4%) | 14 (4.2%) | 18 (6.6%) | 6 (8.0%) | 2 (0.6%) |
| Diarrhea NOS | 3 (1.1%) | 1 (0.7%) | 2 (0.6%) | 21 (7.7%) | 4 (5.3%) | 7 (2.1%) |
| Nausea | 1 (0.4%) | 3 (2.2%) | 4 (1.2%) | 11 (4.0%) | 5 (6.7%) | 6 (1.8%) |
| Abdominal Pain NOS | 1 (0.4%) | 1 (0.7%) | 5 (1.5%) | 12 (4.4%) | 3 (4.0%) | 1 (0.3%) |
| Toothache | 1 (0.4%) | 1 (0.7%) | 2 (0.6%) | 7 (2.6%) | 1 (1.3%) | 2 (0.6%) |
| Constipation | 1 (0.4%) | 0 | 2 (0.6%) | 10 (3.7%) | <1% | 0 |
| Loose Stools | 0 | 1 (0.7%) | 4 (1.2%) | <1% | <1% | 0 |
| Reproductive System and Breast Disorders | ||||||
| Dysmenorrhea | 3 (1.1%) | 0 | 5 (1.5%) | 3 (1.1%) | 1 (1.3%) | 4 (1.2%) |
| Eye Disorders | ||||||
| Conjunctivitis NEC | 2 (0.7%) | 1 (0.7%) | 7 (2.1%) | 6 (2.2%) | 3 (4.0%) | 10 (3.0%) |
| Skin & Subcutaneous Tissue Disorders | ||||||
| Urticaria | 3 (1.1%) | 0 | 1 (0.3%) | 1 (0.4%) | <1% | 3 (0.9%) |
| Acne NOS | 0 | 1 (0.7%) | 1 (0.3%) | 4 (1.5%) | <1% | 6 (1.8%) |
| Immune System Disorders | ||||||
| Hypersensitivity NOS | 11 (4.1%) | 6 (4.4%) | 16 (4.8%) | 14 (5.1%) | 1 (1.3%) | 11 (3.4%) |
| Injury and Poisoning | ||||||
| Accident NOS | 3 (1.1%) | 1 (0.7%) | 1 (0.3%) | <1% | 1 (1.3%) | 0 |
| Laceration | 2 (0.7%) | 1 (0.7%) | 5 (1.5%) | <1% | <1% | 0 |
| Musculoskeletal, Connective Tissue and Bone Disorders | ||||||
| Back Pain | 1 (0.4%) | 2 (1.5%) | 1 (0.3%) | <1% | 0 | 6 (1.8%) |
| Arthralgias | 0 | 0 | 1 (0.3%) | 3 (1.1%) | 1 (1.3%) | 5 (1.5%) |
| Ear and Labyrinth Disorders | ||||||
| Earache | 2 (0.7%) | 1 (0.7%) | 0 | 8 (2.9%) | 2 (2.7%) | 0 |
| Nervous System Disorders | ||||||
| Headache | 37 (13.9%) | 12 (8.8%) | 38 (11.3%) | 69 (25.4%) | 12 (16.0%) | 23 (7.0%) |
Manufactured by:
Novartis Pharma Produktions GmbH
Wehr, Germany
Distributed by:
Valeant Pharmaceuticals North America, LLC
Bridgewater, NJ 08807
2079799 US WEH
6US300
Medication Guide
ELIDEL® [EL´-ee-del]
(pimecrolimus) Cream 1%
Important Note: ELIDEL Cream is for use on the skin only (topical). Do not get ELIDEL Cream in your eyes, nose, mouth, vagina, or rectum.
Read the Medication Guide that comes with ELIDEL Cream before you or a family member start using it and each time you refill your prescription. There may be new information. This Medication Guide does not take the place of talking with your doctor about your medical condition or treatment. If you have any questions about ELIDEL Cream, ask your doctor or pharmacist for more information.
What is the most important information I should know about ELIDEL Cream?
It is not known if ELIDEL Cream is safe to use for a long period of time. A very small number of people who have used ELIDEL Cream have gotten cancer (for example, skin cancer or lymphoma). But a link that ELIDEL Cream use caused these cancers has not been shown. Because of this concern:
- Do not use ELIDEL Cream continuously for a long time.
- Use ELIDEL Cream only on areas of your skin that have eczema.
- Do not use ELIDEL Cream on a child under 2 years old.
What is ELIDEL Cream?
ELIDEL Cream is a prescription medicine used on the skin (topical) to treat eczema (atopic dermatitis). ELIDEL Cream is in a class of medicines called topical calcineurin inhibitors. ELIDEL Cream is for adults and children age 2 years and older who do not have a weakened immune system. ELIDEL Cream is used on the skin for short periods, and if needed, treatment may be repeated with breaks in between. ELIDEL Cream is for use after other prescription medicines have not worked for you or if your doctor recommends that other prescription medicines should not be used. It is not known if ELIDEL Cream is safe and effective in people who have a weakened immune system.
ELIDEL Cream is not for use in children under 2 years of age.
Who should not use ELIDEL Cream?
Do not use ELIDEL Cream:
- if you are allergic to ELIDEL Cream or anything in it. See the end of this Medication Guide for a complete list of ingredients in ELIDEL Cream.
What should I tell my doctor before starting ELIDEL Cream?
Before you start using ELIDEL Cream, tell your doctor about all of your medical conditions, including if you:
- have a skin disease called Netherton's syndrome (a rare inherited condition)
- have any infection on your skin including chicken pox or herpes
- have been told you have a weakened immune system
- are pregnant, breastfeeding, or planning to become pregnant.
Tell your doctor about all the medicines you take including prescription and nonprescription medicines, vitamins, and herbal supplements. Tell your doctor about all the skin medicines and products you use.
Know the medicines you take. Keep a list of them with you to show your doctor and pharmacist each time you get a new medicine.
How should I use ELIDEL Cream?
- Use ELIDEL Cream exactly as prescribed.
- Use ELIDEL Cream only on areas of your skin that have eczema.
- Use ELIDEL Cream for short periods, and if needed, treatment may be repeated with breaks in between.
- Stop ELIDEL Cream when the signs and symptoms of eczema, such as itching, rash, and redness go away, or as directed by your doctor.
- Follow your doctor's advice if symptoms of eczema return after a treatment with ELIDEL Cream.
- Call your doctor if:
- your symptoms get worse with ELIDEL Cream
- you get an infection on your skin
- your symptoms do not improve after 6 weeks of treatment. Sometimes other skin diseases can look like eczema.
To apply ELIDEL Cream:
Read and carefully follow the directions below.
- Wash your hands before using ELIDEL Cream. If you apply ELIDEL Cream after a bath or shower, make sure your skin is dry.
- Apply a thin layer of ELIDEL Cream only to the affected skin areas, two times each day, as directed by your doctor.
- Use the smallest amount of ELIDEL Cream needed to control the signs and symptoms of eczema.
- If you apply ELIDEL Cream to another person, or if you have eczema and are not treating your hands, it is important for you to wash your hands with soap and water after applying ELIDEL Cream. This should remove any cream left on your hands.
- Do not bathe, shower or swim right after applying ELIDEL Cream. This could wash off the cream.
- You can use moisturizers with ELIDEL Cream. Make sure you check with your doctor first about the products that are right for you. People with eczema can have very dry skin, so it is important to keep up good skin care practices. If you use moisturizers, apply them after ELIDEL Cream.
What should I avoid while using ELIDEL Cream?
- You should not use sun lamps, tanning beds, or get treatment with ultraviolet light therapy during treatment with ELIDEL Cream.
- Limit your time in the sun during treatment with ELIDEL Cream even when the medicine is not on your skin. If you need to be outdoors after applying ELIDEL Cream, wear loose fitting clothing that protects the treated area from the sun. Ask your doctor what other types of protection from the sun you should use. It is not known how ELIDEL Cream may affect your skin with exposure to ultraviolet light.
- Do not cover the skin being treated with bandages, dressings or wraps. You can wear normal clothing.
- ELIDEL Cream is for use on the skin only. Do not get ELIDEL Cream in your eyes, nose, mouth, vagina, or rectum (mucous membranes). If you get ELIDEL Cream in any of these areas, burning or irritation can happen. Wipe off any ELIDEL Cream from the affected area and then rinse the area well with cold water.
- Do not swallow ELIDEL Cream. If you do, call your doctor.
- Avoid using ELIDEL Cream on skin areas that have cancers or pre-cancers.
What are the possible side effects of ELIDEL Cream?
ELIDEL Cream may cause serious side effects. A very small number of people who have used ELIDEL Cream have gotten cancer (for example, skin cancer or lymphoma). But, a link that ELIDEL Cream caused these cancers has not been shown.
The most common side effect at the skin application site is burning or a feeling of warmth. These side effects are usually mild or moderate, happen during the first few days of treatment, and usually clear up in a few days. Call your doctor if the burning feeling is severe or lasts for more than 1 week.
Other side effects include headache, common cold or stuffy nose, sore throat, influenza, fever, viral infection, and cough. Some people may get viral skin infections (like cold sores, chicken pox, shingles, or warts) or swollen lymph nodes (glands). Tell your doctor if you have a skin infection or if you have any side effect (for example, swollen glands) that bothers you or that does not go away.
These are not all the possible side effects with ELIDEL Cream. Ask your doctor or pharmacist for more information.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store ELIDEL Cream?
- Store ELIDEL Cream at room temperature between 59° to 86°F (15° to 30°C).
- Keep ELIDEL Cream and all medicines out of the reach of children.
General advice about ELIDEL Cream
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Use ELIDEL Cream only for the condition for which it was prescribed. Do not give ELIDEL Cream to other people even if they have the same symptoms you have, as it may not be right for them.
This Medication Guide summarizes the most important information about ELIDEL Cream. If you would like more information, talk with your doctor.
Your doctor or pharmacist can give you information about ELIDEL Cream that is written for health care professionals. For more information, you can also visit the Internet site at www.Elidel.com or call the ELIDEL Cream help line at 1-800-321-4576.
What are the ingredients in ELIDEL Cream?
Active ingredient: pimecrolimus
Inactive ingredients: benzyl alcohol, cetyl alcohol, citric acid, mono- and di-glycerides, oleyl alcohol, propylene glycol, sodium cetostearyl sulphate, sodium hydroxide, stearyl alcohol, triglycerides, and water
Manufactured by:
Novartis Pharma Produktions GmbH
Wehr, Germany
Distributed by:
Valeant Pharmaceuticals North America, LLC
Bridgewater, NJ 08807
This Medication Guide has been approved by the U.S. Food and Drug Administration
REV. 06/2011
© Novartis
2079799 US WEH
T2010-70/T2010-71
6US300
PRINCIPAL DISPLAY PANEL - 100 g Carton
NDC 0187-5102-03
ELIDEL®
(pimecrolimus) cream 1%
100 g
Rx only
FOR TOPICAL USE ONLY.
NOT FOR OPHTHALMIC USE.
If Elidel Cream gets in your eyes,
rinse your eyes with cold water.
ATTENTION PHARMACIST:
Each patient is required to receive
the enclosed Medication Guide.
VALEANT®
Pharmaceuticals North America

| ELIDEL
pimecrolimus cream |
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| NDA | NDA021302 | 12/02/2001 | |
| Labeler - Valeant Pharmaceuticals North America LLC (042230623) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| Novartis Pharma Produktions GmbH | 333288046 | MANUFACTURE | |
Revised: 10/2011 Valeant Pharmaceuticals North America LLC