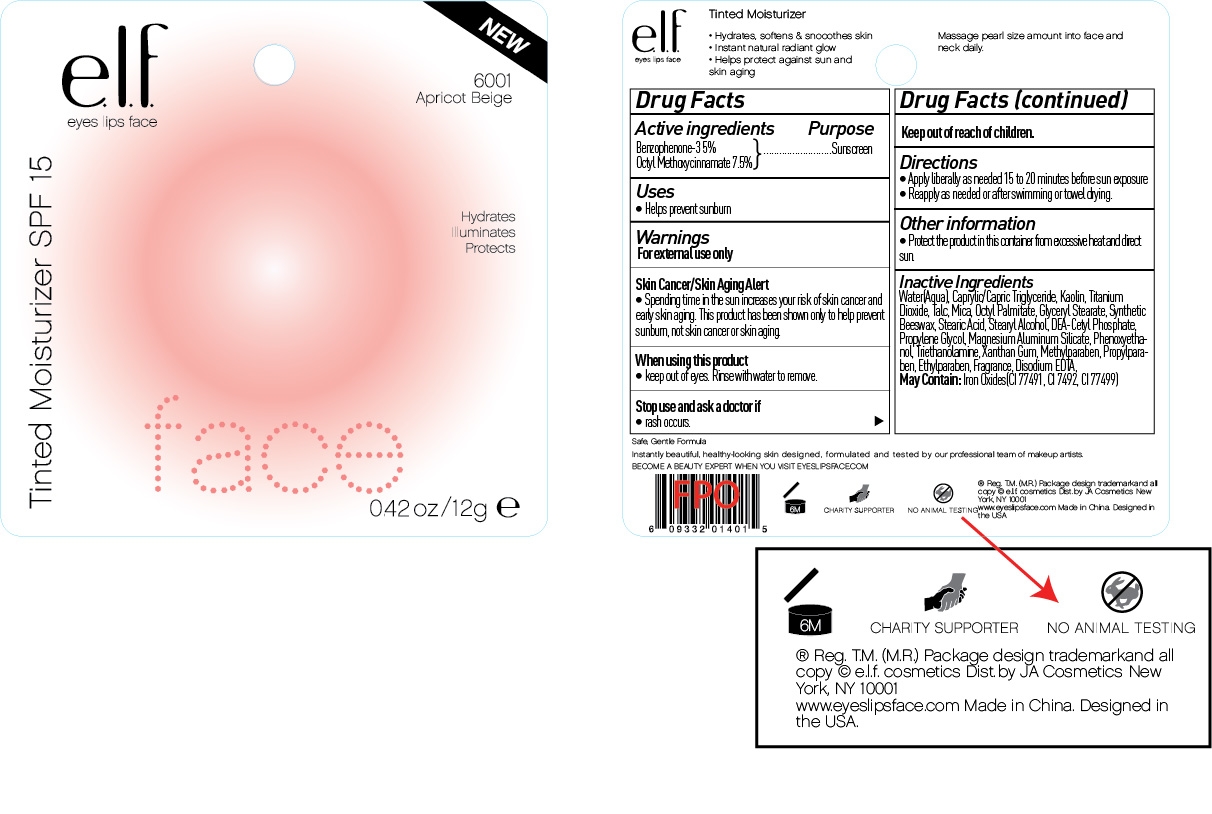
ELF TINTED MOISTURIZER SPF 15
-
oxybenzone and
octinoxate cream
J. A. Cosmetics U.S. INC
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Warning:
For external use only
Skin Cancer/Skin Aging Alert:
Spending time in the sun increases your risk of skin cancer and early skin aging. This product has been shown to only help prevent sunburn, not skin cancer or skin aging.
Directions:
Apply liberally as needed 15-20 minutes before sun exposure
Reapply as needed or after swimming or towel drying
Inactive Ingredient:
Water, Caprylic/Capric Triglyceride, Kaolin, Titanium Dioxide, Talc, Mica, Octyl Palmitate, Glyceryl Stearate, Synthetic Beeswax, Stearic Acid, Stearyl Alcohol, DEA-Cetyl Phosphate, Propylene Glycol, Magnesium Aluminum Silicate, Phenoxyehtanol, Triethanolamine, Xanthan Gum, Fragrance, Methylparaben, Propylparaben, Ethylparaben, Disodium EDTA.
May contain:
Iron Oxides (CI 77491, CI 77492, CI 77499)
| ELF TINTED MOISTURIZER SPF 15
oxybenzone cream |
||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| OTC monograph not final | part352 | 10/09/2011 | |
| Labeler - J. A. Cosmetics U.S. INC (186705047) |
| Registrant - J. A. Cosmetics U.S. INC (186705047) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| Shanghai Justking Enterprise Co., Ltd | 421267459 | manufacture | |
Revised: 10/2011 J. A. Cosmetics U.S. INC