LA PRAIRIE CELLULAR RADIANCE EMULSION
-
avobenzone,
octinoxate and
octisalate cream
La Prairie Group AG
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
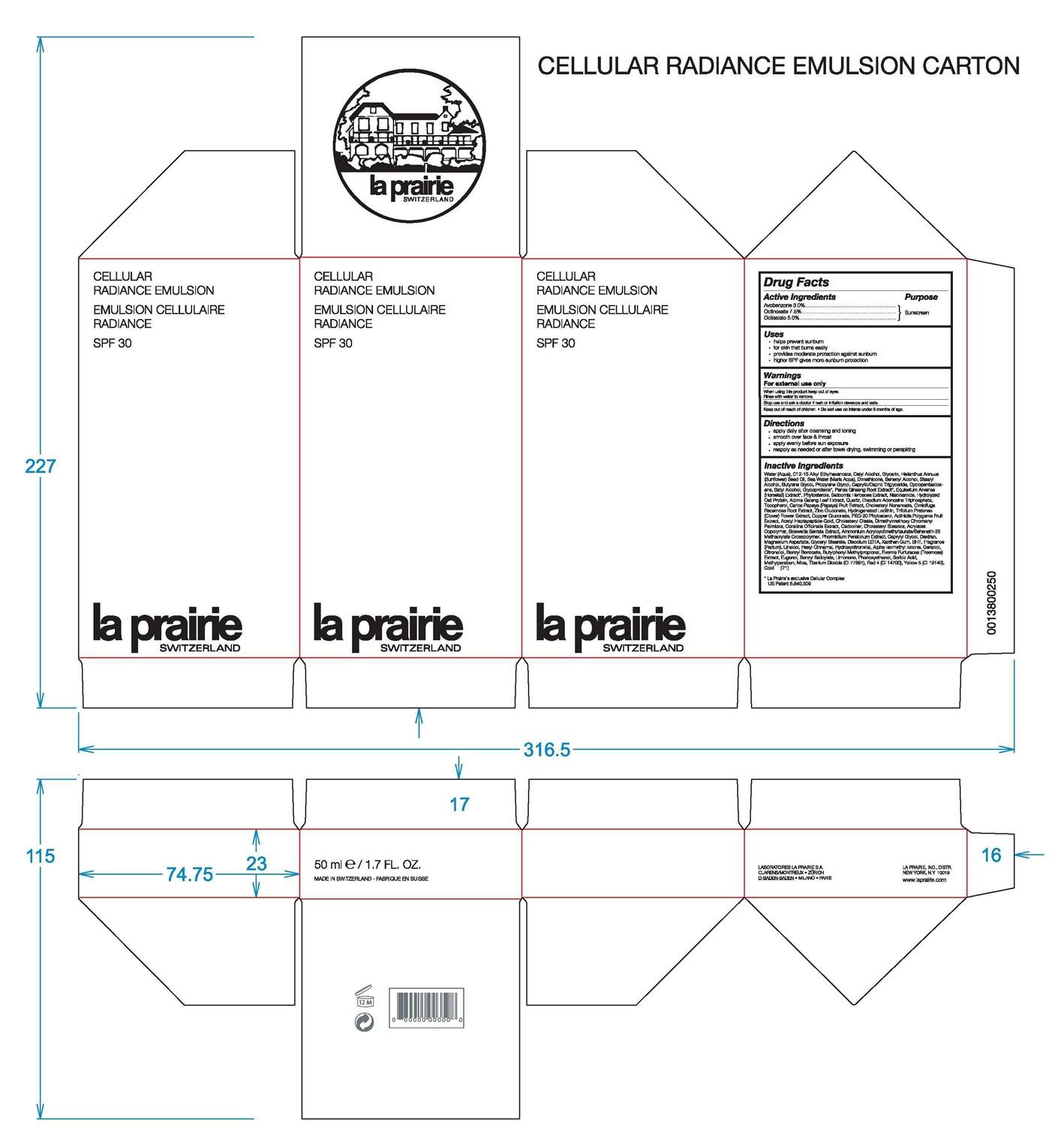
Active Ingredients Purpose
Avobenzone 3.0% Sunscreen
Octinoxate 7.5% Sunscreen
Octisalate 5.0% Sunscreen
Uses
-helps prevent sunburn
-for skin that burns easily
-provides moderate protection against sunburn
-higher SPF gives more sunburn protection
-for external use only
-when using this product keep out of eyes
-rinse with water to remove
-do not use on infants under 6 months of age
Directions
Apply daily after cleansing and toning
Smooth over face and throat
Apply evenly before sun exposure
Reapply as needed or after towel drying, swimming, or perspiring.
Inactive Ingredients: Water (Aqua), C12-15 Alkyl Ethylhexanoate, Cetyl Alcohol, Glycerin, Helianthus Annuus (Sunflower) Seed Oil, Sea Water (Maris Aqua), Dimethicone, Behenyl Alcohol, Stearyl Alcohol, Butylene Glycol, Propylene Glycol, Caprylic/Capric Triglyceride, Cyclopentasiloxane, Batyl Alcohol, Glycoproteins, Panax Ginseng Root Extract, Equisetum Arvense (Horsetail) Extract, Phytosterols,
Silicornia Herbacea Extract, Niacinamide, Hydrolyzed Oat Protein, Alpinia Galang Leaf Extract, Quartz,
Disodium Adenosine Triphosphate, Tocopherol, Carica Papaya (Papaya) Fruit Extract, Cholesteryl Nonanoate,
Cimicifuga Racemosa Root Extract, Copper Gluconate, PEG-20 Phytosterol, Actinidia Polygama Fruit Extract,
Acetyl Heptapeptide-Gold, Cholesteryl Oleate, Dimethylmethoxy Chromanyl Palmitate, Corallina Officinalis Extract,
Carbomer, Cholsteryl Stearate, Acrylates Copolymer, Boswellia Serrata Extract, Ammonium Acryloyldimethltaurate/Beheneth-25 Methacrylate Crosspolymer, Phormidium Persicinum Extract, Caprylyl Glycol, Dextran, Magnesium Asparate Glyceryl Stearate, Disodium EDTA, Xanthan Gum, BHT, Fragrance (Parfum), Linalool, Hexyl Cinnamal, Hydroxycitronellal, Alpha-Isomethyl Ionone, Geraniol, Citronellol, Benzyl Benzoate, Butylphenyl Methylpropional, Evernia Furfuracea (Treemoss) Extract, Eugenol, Benzyl Salicylate, Limonene, Phenoxyethanol, Sorbia Acid, Methylparaben, Mica, Titanium Dioxide (CI 77891), Red (CI 14700), Yellow 5 (CI 19140), Gold

| LA PRAIRIE
CELLULAR RADIANCE EMULSION
avobenzone, octinoxate, octisalate cream |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| OTC monograph not final | part352 | 01/01/2009 | |
| Labeler - La Prairie Group AG (482051638) |
| Registrant - Temmentec Ag (480586411) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| Temmentec Ag | 480586411 | manufacture | |
Revised: 06/2011 La Prairie Group AG