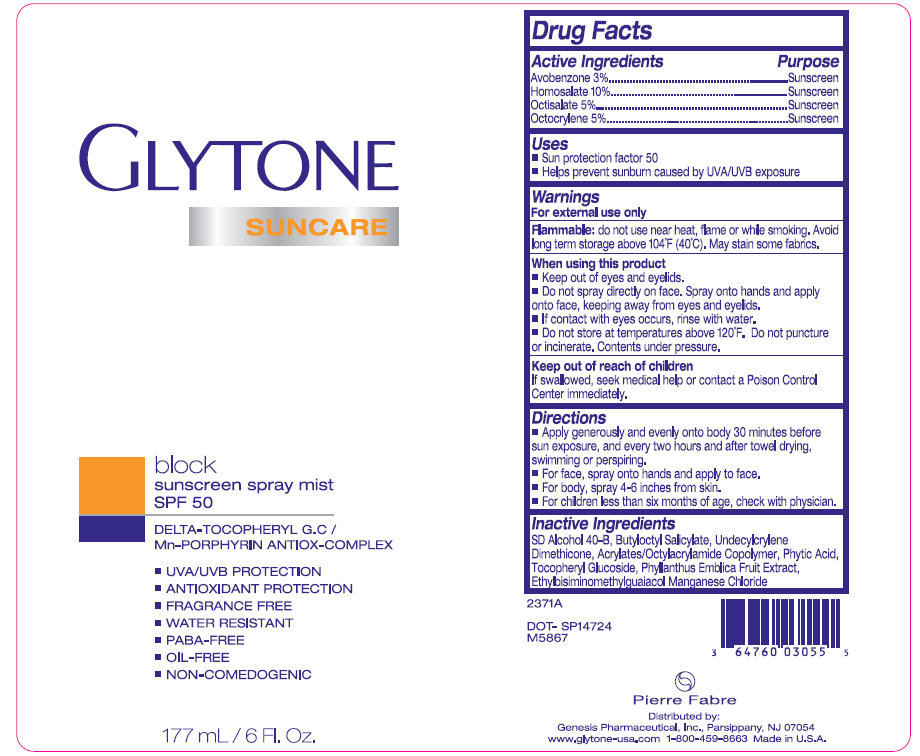
GLYTONE SUNCARE SPF 50
-
avobenzone,
homosalate,
octisalate and
octocrylene spray
Pierre Fabre Dermo Cosmetique
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Active Ingredients Purpose
Avobenzone 3%.............................................Sunscreen
Homosalate 10%............................................Sunscreen
Octisalate 5%.................................................Sunscreen
Octocrylene 5%..............................................Sunscreen
Keep out of reach of children. If swallowed, seek medical help or contact a Poison Control Center immediately.
Directions - Apply generously and evenly onto body 30 minutes before sun exposure, and every two hours and after towel drying, swimming or perspiring. - For face, spray onto hands and apply to face. - For body, spray 4-6 inches from skin. - For children less than six months of age, check with physician.
Inactive Ingredients
SD Alcohol 40-B, Butyloctyl Salicylate, Undecylcrylene Dimethicone, Acrylates/Octylacrylamide Copolymer, Phytic Acid, Tocopheryl Glucoside, Phyllanthus Emblica Fruit Extract, Ethylbisiminomethylguaiacol Manganese Chloride.
2371A DOT-SP14724 M5867 3 64760 03055 5 Pierre Fabre Distributed by: Genesis Pharmaceutical, Inc., Parsippany, NJ 07054 www.glytone-usa.com 1-800-459-8663 Made in U.S.A.
GluTONE SUNCARE block sunscreen spray mist SPF 50 DELTA-TOCOPHERYL G.C / Mn-PORPHYRIN ANTIOX-COMPLEX - UVA/UVB PROTECTION - ANTIOXIDANT PROTECTION - FRAGRANCE FREE - WATER RESISTANT - PABA-FREE - OIL-FREE - NON-COMEDOGENIC 177 mL / 6 Fi. Oz.
| GLYTONE SUNCARE SPF 50
avobenzone, homosalate, octisalate, octocrylene spray |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| OTC monograph final | part352 | 06/09/2011 | |
| Labeler - Pierre Fabre Dermo Cosmetique (378028104) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| Formulated Solutions, LLC | 143266687 | manufacture | |
Revised: 06/2011 Pierre Fabre Dermo Cosmetique