EQUALINE ALLERGY
-
diphenhydramine hydrochloride capsule
Supervalu Inc
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
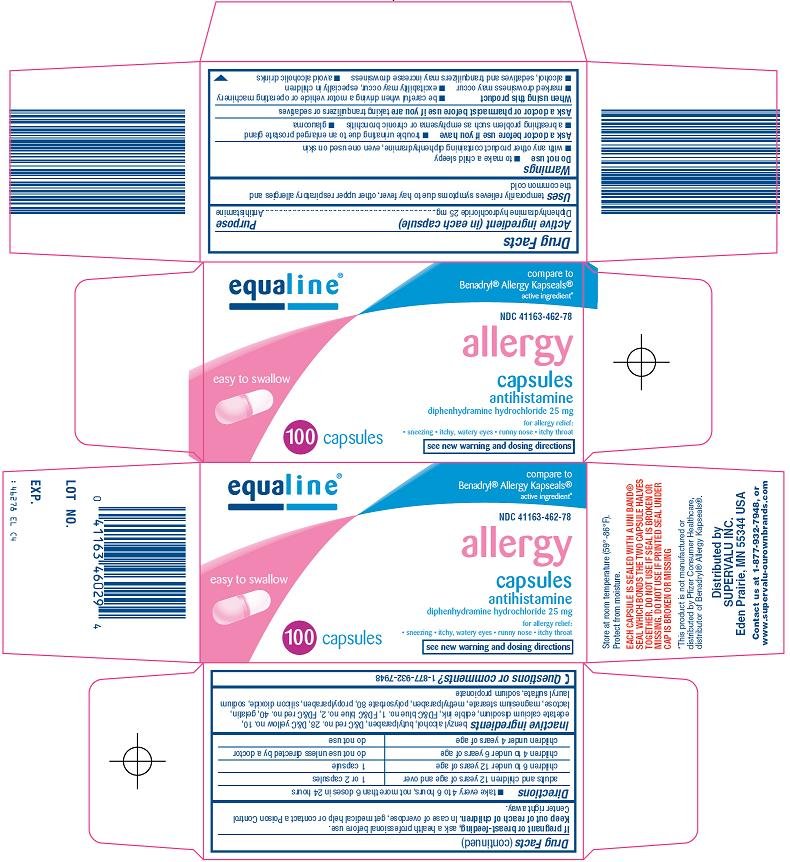
Uses (Blister Carton)
- temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- runny nose
- sneezing
- itchy, watery eyes
- itchy nose or throat
- temporarily relieves these symptoms of the common cold:
- runny nose
- sneezing
Uses (Bottle Carton)
temporarily relieves symptoms due to hay fever, other upper respiratory allergies and the common cold
Warnings
Do not use
- with any other product containing diphenhydramine, even one used on skin
- to make a child sleepy
Ask a doctor before use if you have
- trouble urinating due to an enlarged prostate gland
- glaucoma
- a breathing problem such as emphysema or chronic bronchitis
Directions
- take every 4 to 6 hours, not more than 6 doses in 24 hours
| adults and children 12 years of age and over | 1 or 2 capsules |
| children 6 to under 12 years of age | 1 capsule |
| children 4 to under 6 years of age | do not use unless directed by a doctor |
| children under 4 years of age | do not use |
Inactive ingredients
benzyl alcohol, butylparaben, D&C red no. 28, D&C yellow no. 10, edetate calcium disodium, edible ink, FD&C blue no. 1, FD&C blue no. 2, FD&C red no. 40, gelatin, lactose, magnesium stearate, methylparaben, polysorbate 80, propylparaben, silicon dioxide, sodium lauryl sulfate, sodium propionate
Principal Display Panel
Compare to Benadryl® Allergy Kapseals® active ingredient
Allergy
Capsules
Antihistamine
Diphenhydramine Hydrochloride 25 mg
For Allergy Relief:
Sneezing
Itchy, Watery Eyes
Runny Nose
Itchy Throat
See New Warning and Dosing Directions
Easy to Swallow
Allergy Capsules Carton
| EQUALINE ALLERGY
diphenhydramine hydrochloride capsule |
||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| OTC monograph final | part341 | 08/17/2004 | |
| Labeler - Supervalu Inc (006961411) |
Revised: 11/2009 Supervalu Inc