CREST SENSITIVITY TREATMENT AND PROTECTION
-
stannous fluoride paste, dentifrice
Procter & Gamble Manufacturing Company
----------
CrestSENSITIVITY
TREATMENT & PROTECTION
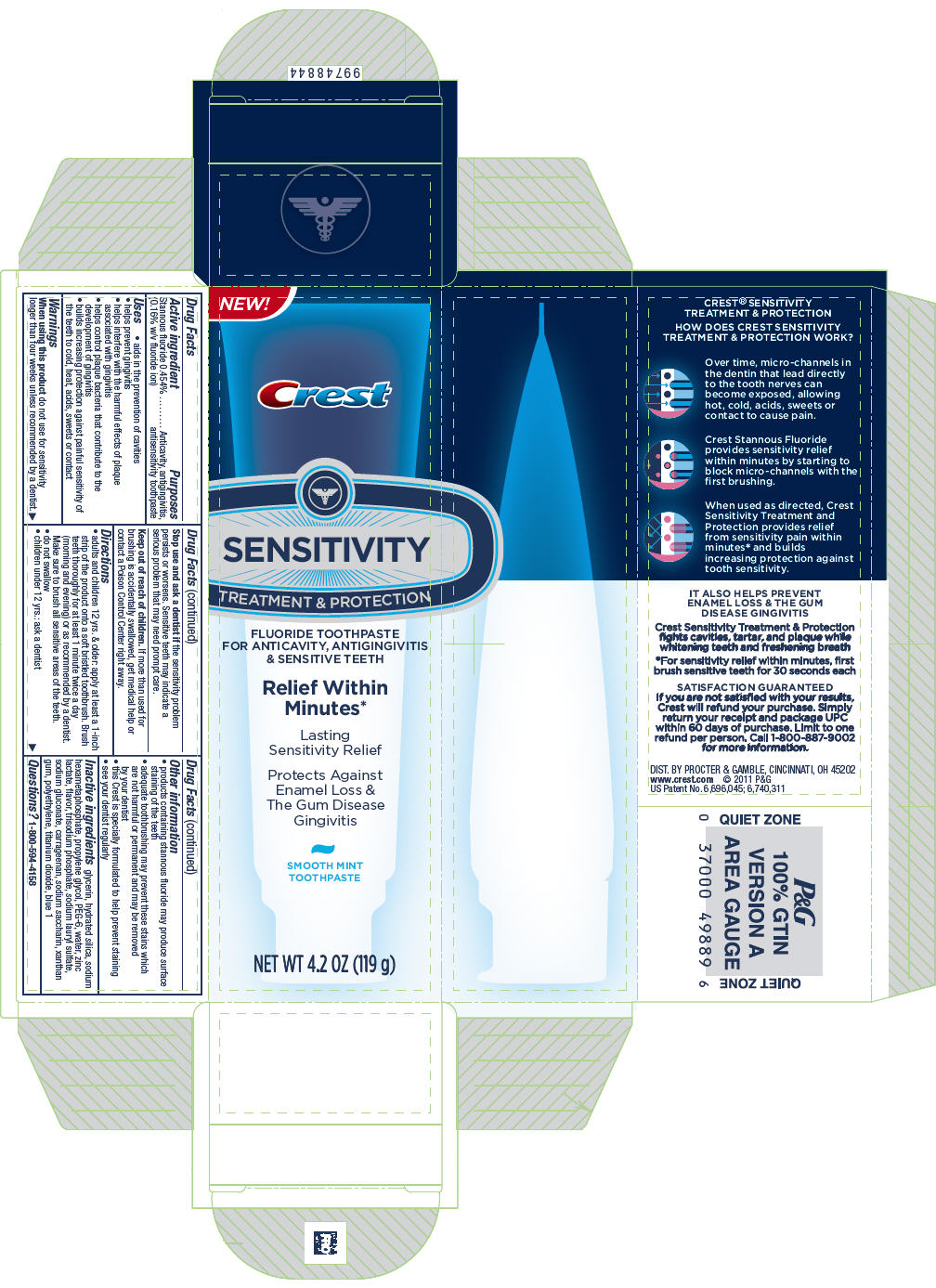
Drug Facts
Active ingredient
Stannous fluoride 0.454% (0.16% w/v fluoride ion)
Purposes
Anticavity, antigingivitis, antisensitivity toothpaste
Uses
- aids in the prevention of cavities
- helps prevent gingivitis
- helps interfere with the harmful effects of plaque associated with gingivitis
- helps control plaque bacteria that contribute to the development of gingivitis
- builds increasing protection against painful sensitivity of the teeth to cold, heat, acids, sweets or contact
Warnings
When using this product do not use for sensitivity longer than four weeks unless recommended by a dentist.
Stop use and ask a dentist if the sensitivity problem persists or worsens. Sensitive teeth may indicate a serious problem that may need prompt care.
Keep out of reach of children. If more than used for brushing is accidentally swallowed, get medical help or contact a Poison Control Center right away.
Directions
- adults and children 12 yrs. & older: apply at least a 1-inch strip of the product onto a soft bristled toothbrush. Brush teeth thoroughly for at least 1 minute twice a day (morning and evening) or as recommended by a dentist. Make sure to brush all sensitive areas of the teeth.
- do not swallow
- children under 12 yrs.: ask a dentist
Other information
- products containing stannous fluoride may produce surface staining of the teeth
- adequate toothbrushing may prevent these stains which are not harmful or permanent and may be removed by your dentist
- this Crest is specially formulated to help prevent staining
- see your dentist regularly
Inactive ingredients
glycerin, hydrated silica, sodium hexametaphosphate, propylene glycol, PEG-6, water, zinc lactate, flavor, trisodium phosphate, sodium lauryl sulfate, sodium gluconate, carrageenan, sodium saccharin, xanthan gum, polyethylene, titanium dioxide, blue 1
Questions?
1-800-594-4158
PRINCIPAL DISPLAY PANEL - 119 g Carton
NEW!
Crest
SENSITIVITY
TREATMENT & PROTECTION
FLUORIDE TOOTHPASTE
FOR ANTICAVITY, ANTIGINGIVITIS
& SENSITIVE TEETH
Relief Within
Minutes*
Lasting
Sensitivity Relief
Protects Against
Enamel Loss &
The Gum Disease
Gingivitis
SMOOTH MINT
TOOTHPASTE
NET WT 4.2 OZ (119 g)
| CREST SENSITIVITY
TREATMENT AND PROTECTION
stannous fluoride paste, dentifrice |
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| OTC monograph final | part355 | 03/01/2011 | |
| Labeler - Procter & Gamble Manufacturing Company (004238200) |