humatrope (Somatropin)
[ELI LILLY AND COMPANY]
DESCRIPTION
Humatrope® (Somatropin, rDNA Origin, for Injection) is a polypeptide hormone of recombinant DNA origin. Humatrope has 191 amino acid residues and a molecular weight of about 22,125 daltons. The amino acid sequence of the product is identical to that of human growth hormone of pituitary origin. Humatrope is synthesized in a strain of Escherichia coli that has been modified by the addition of the gene for human growth hormone.
Humatrope is a sterile, white, lyophilized powder intended for subcutaneous or intramuscular administration after reconstitution. Humatrope is a highly purified preparation. Phosphoric acid and/or sodium hydroxide may have been added to adjust the pH. Reconstituted solutions have a pH of approximately 7.5. This product is oxygen sensitive.
VIAL — Each vial of Humatrope contains 5 mg somatropin (15 IU or 225 nanomoles); 25 mg mannitol; 5 mg glycine; and 1.13 mg dibasic sodium phosphate. Each vial is supplied in a combination package with an accompanying 5–mL vial of diluting solution. The diluent contains Water for Injection with 0.3% Metacresol as a preservative and 1.7% glycerin.
CARTRIDGE — The cartridges of somatropin contain either 6 mg (18 IU), 12 mg (36 IU), or 24 mg (72 IU) of somatropin. The 6, 12, and 24 mg cartridges contain respectively: mannitol 18, 36, and 72 mg; glycine 6, 12, and 24 mg; dibasic sodium phosphate 1.36, 2.72, and 5.43 mg. Each cartridge is supplied in a combination package with an accompanying syringe containing approximately 3 mL of diluting solution. The diluent contains Water for Injection; 0.3% Metacresol as a preservative; and 1.7%, 0.29%, and 0.29% glycerin in the 6, 12, and 24 mg cartridges, respectively.
CLINICAL PHARMACOLOGY
General
Linear Growth — Humatrope stimulates linear growth in pediatric patients who lack adequate normal endogenous growth hormone. In vitro, preclinical, and clinical testing have demonstrated that Humatrope is therapeutically equivalent to human growth hormone of pituitary origin and achieves equivalent pharmacokinetic profiles in normal adults. Treatment of growth hormone–deficient pediatric patients and patients with Turner syndrome with Humatrope produces increased growth rate and IGF–I (Insulin–like Growth Factor–I/Somatomedin–C) concentrations similar to those seen after therapy with human growth hormone of pituitary origin.
In addition, the following actions have been demonstrated for Humatrope and/or human growth hormone of pituitary origin.
A. Tissue Growth — 1. Skeletal Growth: Humatrope stimulates skeletal growth in pediatric patients with growth hormone deficiency. The measurable increase in body length after administration of either Humatrope or human growth hormone of pituitary origin results from an effect on the growth plates of long bones. Concentrations of IGF–I, which may play a role in skeletal growth, are low in the serum of growth hormone–deficient pediatric patients but increase during treatment with Humatrope. Elevations in mean serum alkaline phosphatase concentrations are also seen. 2. Cell Growth: It has been shown that there are fewer skeletal muscle cells in short–statured pediatric patients who lack endogenous growth hormone as compared with normal pediatric populations. Treatment with human growth hormone of pituitary origin results in an increase in both the number and size of muscle cells.
B. Protein Metabolism — Linear growth is facilitated in part by increased cellular protein synthesis. Nitrogen retention, as demonstrated by decreased urinary nitrogen excretion and serum urea nitrogen, follows the initiation of therapy with human growth hormone of pituitary origin. Treatment with Humatrope results in a similar decrease in serum urea nitrogen.
C. Carbohydrate Metabolism — Pediatric patients with hypopituitarism sometimes experience fasting hypoglycemia that is improved by treatment with Humatrope. Large doses of human growth hormone may impair glucose tolerance. Untreated patients with Turner syndrome have an increased incidence of glucose intolerance. Administration of human growth hormone to normal adults or patients with Turner syndrome resulted in increases in mean serum fasting and postprandial insulin levels although mean values remained in the normal range. In addition, mean fasting and postprandial glucose and hemoglobin A1c levels remained in the normal range.
D. Lipid Metabolism — In growth hormone–deficient patients, administration of human growth hormone of pituitary origin has resulted in lipid mobilization, reduction in body fat stores, and increased plasma fatty acids.
E. Mineral Metabolism — Retention of sodium, potassium, and phosphorus is induced by human growth hormone of pituitary origin. Serum concentrations of inorganic phosphate increased in patients with growth hormone deficiency after therapy with Humatrope or human growth hormone of pituitary origin. Serum calcium is not significantly altered in patients treated with either human growth hormone of pituitary origin or Humatrope.
Pharmacokinetics
Absorption — Humatrope has been studied following intramuscular, subcutaneous, and intravenous administration in adult volunteers. The absolute bioavailability of somatropin is 75% and 63% after subcutaneous and intramuscular administration, respectively.
Distribution — The volume of distribution of somatropin after intravenous injection is about 0.07 L/kg.
Metabolism — Extensive metabolism studies have not been conducted. The metabolic fate of somatropin involves classical protein catabolism in both the liver and kidneys. In renal cells, at least a portion of the breakdown products of growth hormone is returned to the systemic circulation. In normal volunteers, mean clearance is 0.14 L/hr/kg. The mean half–life of intravenous somatropin is 0.36 hours, whereas subcutaneously and intramuscularly administered somatropin have mean half–lives of 3.8 and 4.9 hours, respectively. The longer half–life observed after subcutaneous or intramuscular administration is due to slow absorption from the injection site.
Excretion — Urinary excretion of intact Humatrope has not been measured. Small amounts of somatropin have been detected in the urine of pediatric patients following replacement therapy.
Special Populations
Geriatric — The pharmacokinetics of Humatrope has not been studied in patients greater than 65 years of age.
Pediatric — The pharmacokinetics of Humatrope in pediatric patients is similar to adults.
Gender — No studies have been performed with Humatrope. The available literature indicates that the pharmacokinetics of growth hormone is similar in both men and women.
Race — No data are available.
Renal, Hepatic insufficiency — No studies have been performed with Humatrope.
|
Cmax
|
t1/2
|
AUC0-∞
|
Cls |
Vβ |
|
|
0.02 mg (0.05 IU†)/kg | |||||
|
iv | |||||
|
MEAN |
415 |
0.363 |
156 |
0.135 |
0.0703 |
|
SD |
75 |
0.053 |
33 |
0.029 |
0.0173 |
|
0.1 mg (0.27 IU†)/kg | |||||
|
im | |||||
|
MEAN |
53.2 |
4.93 |
495 |
0.215 |
1.55 |
|
SD |
25.9 |
2.66 |
106 |
0.047 |
0.91 |
|
0.1 mg (0.27 IU†)/kg | |||||
|
sc | |||||
|
MEAN |
63.3 |
3.81 |
585 |
0.179 |
0.957 |
|
SD |
18.2 |
1.40 |
90 |
0.028 |
0.301 |
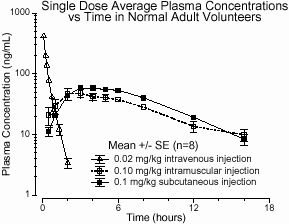
Figure 1
CLINICAL TRIALS
Effects of Humatrope Treatment in Adults with Growth Hormone Deficiency
Two multicenter trials in adult–onset growth hormone deficiency (n=98) and two studies in childhood–onset growth hormone deficiency (n=67) were designed to assess the effects of replacement therapy with Humatrope. The primary efficacy measures were body composition (lean body mass and fat mass), lipid parameters, and the Nottingham Health Profile. The Nottingham Health Profile is a general health–related quality of life questionnaire. These four studies each included a 6–month randomized, blinded, placebo–controlled phase followed by 12 months of open–label therapy for all patients. The Humatrope dosages for all studies were identical: 1 month of therapy at 0.00625 mg/kg/day followed by the proposed maintenance dose of 0.0125 mg/kg/day. Adult–onset patients and childhood–onset patients differed by diagnosis (organic vs. idiopathic pituitary disease), body size (normal vs. small for mean height and weight), and age (mean=44 vs. 29 years). Lean body mass was determined by bioelectrical impedance analysis (BIA), validated with potassium 40. Body fat was assessed by BIA and sum of skinfold thickness. Lipid subfractions were analyzed by standard assay methods in a central laboratory.
Humatrope–treated adult–onset patients, as compared to placebo, experienced an increase in lean body mass (2.59 vs. –0.22 kg, p<0.001) and a decrease in body fat (–3.27 vs. 0.56 kg, p<0.001). Similar changes were seen in childhood–onset growth hormone–deficient patients. These significant changes in lean body mass persisted throughout the 18–month period as compared to baseline for both groups, and for fat mass in the childhood–onset group. Total cholesterol decreased short–term (first 3 months) although the changes did not persist. However, the low HDL cholesterol levels observed at baseline (mean=30.1 mg/mL and 33.9 mg/mL in adult–onset and childhood–onset patients) normalized by the end of 18 months of therapy (a change of 13.7 and 11.1 mg/dL for the adult–onset and childhood–onset groups, p<0.001). Adult–onset patients reported significant improvements as compared to placebo in the following two of six possible health–related domains: physical mobility and social isolation (Table 2). Patients with childhood–onset disease failed to demonstrate improvements in Nottingham Health Profile outcomes.
Two additional studies on the effect of Humatrope on exercise capacity were also conducted. Improved physical function was documented by increased exercise capacity (VO2 max, p<0.005) and work performance (Watts, p<0.01) (J Clin Endocrinol Metab 1995; 80:552–557).
Two studies evaluating the effect of Humatrope on bone mineralization were subsequently conducted. In a 2–year, randomized, double–blind, placebo–controlled trial, 67 patients with previously untreated adult–onset growth hormone (GH) deficiency received placebo or Humatrope treatment titrated to maintain serum IGF–I within the age–adjusted normal range. In men, but not women, lumbar spine bone mineral density (BMD) increased with Humatrope treatment compared to placebo with a treatment difference of approximately 4% (p=0.001). There was no significant change in hip BMD with Humatrope treatment in men or women, when compared to placebo. In a 2–year, open–label, randomized trial, 149 patients with childhood–onset GH deficiency, who had completed pediatric GH therapy, had attained final height (height velocity <1 cm/yr) and were confirmed to be GH–deficient as young adults (commonly referred to as transition patients), received Humatrope 12.5 µg/kg/day, Humatrope 25 µg/kg/day, or were followed with no therapy. Patients who were randomized to treatment with Humatrope at 12.5 µg/kg/day achieved a 2.9% greater increase from baseline than control in total body bone mineral content (BMC) (8.1 ± 9.0% vs. 5.2 ± 8.2%, p=0.02), whereas patients treated with Humatrope at 25 µg/kg/day had no significant change in BMC. These results include data from patients who received less than 2 years of treatment. A greater treatment effect was observed for patients who completed 2 years of treatment. Increases in lumbar spine BMD and BMC were also statistically significant compared to control with the 12.5 µg/kg/day dose but not the 25 µg/kg/day dose. Hip BMD and BMC did not change significantly compared to control with either dose. The effect of GH treatment on BMC and BMD in transition patients at doses lower than 12.5 µg/kg/day was not studied. The effect of Humatrope on the occurrence of osteoporotic fractures has not been studied.
|
Outcome |
Placebo |
Humatrope Therapy |
|
|
Energy level |
-11.4 |
-15.5 |
NS |
|
Physical mobility |
-3.1 |
-10.5 |
p<0.01 |
|
Social isolation |
0.5 |
-4.7 |
p<0.01 |
|
Emotional reactions |
-4.5 |
-5.4 |
NS |
|
Sleep |
-6.4 |
-3.7 |
NS |
|
Pain |
-2.8 |
-2.9 |
NS |
Effects of Growth Hormone Treatment in Patients with Turner Syndrome
One long–term, randomized, open–label multicenter concurrently controlled study, two long–term, open–label multicenter, historically controlled studies and one long–term, randomized, dose–response study were conducted to evaluate the efficacy of growth hormone for the treatment of patients with short stature due to Turner syndrome.
In the randomized study, GDCT, comparing growth hormone–treated patients to a concurrent control group who received no growth hormone, the growth hormone–treated patients who received a dose of 0.3 mg/kg/wk given 6 times per week from a mean age of 11.7 years for a mean duration of 4.7 years attained a mean near final height of 146.0 ± 6.2 cm (n=27, mean ± SD) as compared to the control group who attained a near final height of 142.1 ± 4.8 cm (n=19). By analysis of covariance1, the effect of growth hormone therapy was a mean height increase of 5.4 cm (p=0.001).
__________________
In two of the studies (85–023 and 85–044), the effect of long–term growth hormone treatment (0.375 mg/kg/wk given either 3 times per week or daily) on adult height was determined by comparing adult heights in the treated patients with those of age–matched historical controls with Turner syndrome who never received any growth–promoting therapy. The greatest improvement in adult height was observed in patients who received early growth hormone treatment and estrogen after age 14 years. In Study 85–023, this resulted in a mean adult height gain of 7.4 cm (mean duration of GH therapy of 7.6 years) vs. matched historical controls by analysis of covariance.
In Study 85–044, patients treated with early growth hormone therapy were randomized to receive estrogen replacement therapy (conjugated estrogens, 0.3 mg escalating to 0.625 mg daily) at either age 12 or 15 years. Compared with matched historical controls, early GH therapy (mean duration of GH therapy 5.6 years) combined with estrogen replacement at age 12 years resulted in an adult height gain of 5.9 cm (n=26), whereas patients who initiated estrogen at age 15 years (mean duration of GH therapy 6.1 years) had a mean adult height gain of 8.3 cm (n=29). Patients who initiated GH therapy after age 11 (mean age 12.7 years; mean duration of GH therapy 3.8 years) had a mean adult height gain of 5.0 cm (n=51).
In a randomized blinded dose–response study, GDCI, patients were treated from a mean age of 11.1 years for a mean duration of 5.3 years with a weekly dose of either 0.27 mg/kg or 0.36 mg/kg administered 3 or 6 times weekly. The mean near final height of patients receiving growth hormone was 148.7 ± 6.5 cm (n=31). When compared to historical control data, the mean gain in adult height was approximately 5 cm.
In some studies, Turner syndrome patients (n=181) treated to final adult height achieved statistically significant average height gains ranging from 5.0 to 8.3 cm.
|
Study/ |
Study |
N at Adult |
GH |
Estrogen |
GH |
Adult Height |
|
|
|||||||
|
GDCT |
RCT |
27 |
11.7 |
13 |
4.7 |
5.4 |
|
|
85-023 |
MHT |
17 |
9.1 |
15.2 |
7.6 |
7.4 |
|
|
85-044: |
A‡ |
MHT |
29 |
9.4 |
15 |
6.1 |
8.3 |
|
B‡ |
26 |
9.6 |
12.3 |
5.6 |
5.9 |
||
|
C‡ |
51 |
12.7 |
13.7 |
3.8 |
5 |
||
|
GDCI |
RDT |
31 |
11.1 |
8-13.5 |
5.3 |
~5§ |
|
- 1
- Analysis of covariance includes adjustments for baseline height relative to age and for mid–parental height.
Effect of Humatrope Treatment in Pediatric Patients with Idiopathic Short Stature
Two randomized, multicenter trials, 1 placebo–controlled and 1 dose–response, were conducted in pediatric patients with idiopathic short stature, also called non–growth hormone–deficient short stature. The diagnosis of idiopathic short stature was made after excluding other known causes of short stature, as well as growth hormone deficiency. Limited safety and efficacy data are available below the age of 7 years. No specific studies have been conducted in pediatric patients with familial short stature or who were born small for gestational age (SGA).
The placebo–controlled study enrolled 71 pediatric patients (55 males, 16 females) 9 to 15 years old (mean age 12.38 ± 1.51 years), with short stature, 68 of whom received study drug. Patients were predominately Tanner I (45.1%) and Tanner II (46.5%) at baseline.
In this double–blind trial, patients received subcutaneous injections of either Humatrope 0.222 mg/kg/wk or placebo. Study drug was given in divided doses 3 times per week until height velocity decreased to ≤1.5 cm/year (“final height”). Thirty–three subjects (22 Humatrope, 11 placebo) had final height measurements after a mean treatment duration of 4.4 years (range 0.11–9.08 years).
The Humatrope group achieved a mean final height Standard Deviation Score (SDS) of –1.8 (Table 4). Placebo–treated patients had a mean final height SDS of –2.3 (mean treatment difference = 0.51, p=0.017). Height gain across the duration of the study and final height SDS minus baseline predicted height SDS were also significantly greater in Humatrope–treated patients than in placebo–treated patients (Table 4 and 5). In addition, the number of patients who achieved a final height above the 5th percentile of the general population for age and sex was significantly greater in the Humatrope group than the placebo group (41% vs. 0%, p<0.05), as was the number of patients who gained at least 1 SDS unit in height across the duration of the study (50% vs. 0%, p<0.05).
|
Humatrope |
Placebo |
Treatment Effect |
p–value |
|
|
||||
|
Baseline height SDS |
-2.7 (0.6) |
-2.75 (0.6) |
0.77 |
|
|
BPH SDS |
-2.1 (0.7) |
-2.3 (0.8) |
0.53 |
|
|
Final height SDS‡ |
-1.8 (0.8) |
-2.3 (0.6) |
0.51 (0.10, 0.92) |
0.017 |
|
FH SDS - baseline height SDS |
0.9 (0.7) |
0.4 (0.2) |
0.51 (0.04, 0.97) |
0.034 |
|
FH SDS - BPH SDS |
0.3 (0.6) |
-0.1 (0.6) |
0.46 (0.02, 0.89) |
0.043 |
The dose–response study included 239 pediatric patients (158 males, 81 females), 5 to 15 years old, (mean age 9.8 ± 2.3 years). Mean baseline characteristics included: a height SDS of –3.21 (±0.70), a predicted adult height SDS of –2.63 (±1.08), and a height velocity SDS of –1.09 (±1.15). All but 3 patients were Tanner I. Patients were randomized to one of three Humatrope treatment groups: 0.24 mg/kg/wk; 0.24 mg/kg/wk for 1 year, followed by 0.37 mg/kg/wk; and 0.37 mg/kg/wk.
The primary hypothesis of this study was that treatment with Humatrope would increase height velocity during the first 2 years of therapy in a dose–dependent manner. Additionally, after completing the initial 2–year dose–response phase of the study, 50 patients were followed to final height.
Patients receiving 0.37 mg/kg/wk had a significantly greater increase in mean height velocity after 2 years of treatment than patients receiving 0.24 mg/kg/wk (4.04 vs. 3.27 cm/year, p=0.003). The mean difference between final height and baseline predicted height was 7.2 cm for patients receiving 0.37 mg/kg/wk and 5.4 cm for patients receiving 0.24 mg/kg/wk (Table 5). While no patient had height above the 5th percentile in any dose group at baseline, 82% of the patients receiving 0.37 mg/kg/wk and 47% of the patients receiving 0.24 mg/kg/wk achieved a final height above the 5th percentile of the general population height standards (p=NS).
|
Placebo-controlled
Trial |
Dose Response
Trial |
||||
|
Placebo |
Humatrope |
Humatrope |
Humatrope |
Humatrope |
|
|
|||||
|
FH– Baseline PH |
|
|
|
|
|
Effect of Humatrope Treatment in Patients with SHOX Deficiency
SHOX deficiency may result either from a deletion of one copy of the short stature homeobox-containing gene (SHOX) or from a mutation within or outside one copy of the SHOX gene that impairs the production or function of SHOX protein.
A randomized, controlled, two-year, three-arm, open-label study was conducted to evaluate the efficacy of Humatrope treatment of short stature in pediatric patients with SHOX deficiency who were not GH deficient. 52 patients (24 male, 28 female) with SHOX deficiency, 3.0 to 12.3 years of age, were randomized to either a Humatrope-treated arm (27 patients; mean age 7.3 ± 2.1 years) or an untreated control arm (25 patients; mean age 7.5 ± 2.7 years). To determine the comparability of treatment effect between patients with SHOX deficiency and patients with Turner syndrome, the third study arm enrolled 26 patients with Turner syndrome, 4.5 to 11.8 years of age (mean age 7.5 ± 1.9 years), to Humatrope treatment. All patients were prepubertal at study entry. Patients in the Humatrope-treated group(s) received daily subcutaneous injections of 0.05 mg/kg of Humatrope. Patients in the untreated group received no injections.
Patients with SHOX deficiency who received Humatrope had significantly greater first-year height velocity than untreated patients (8.7 cm/year vs. 5.2 cm/year, p<0.001, primary efficacy analysis) and similar first-year height velocity to Humatrope-treated patients with Turner syndrome (8.7 cm/year vs. 8.9 cm/year, CI: (-1.3, 0.7). In addition, patients who received Humatrope had significantly greater second year height velocity, and first and second year height gain than untreated patients (Table 6).
|
SHOX Deficiency |
Turner Syndrome |
|||
|
Untreated |
Humatrope |
Treatment Difference*
|
Humatrope |
|
|
Height Velocity (cm/yr) | ||||
|
1st Year | ||||
|
Mean (SD) |
5.2 (1.1) |
8.7 (1.6)† |
+3.5 (2.8, 4.2) |
8.9 (2.0) |
|
2nd Year | ||||
|
Mean (SD) |
5.4 (1.2) |
7.3 (1.1)† |
+2.0 (1.3, 2.6) |
7.0 (1.1) |
|
Height change (cm) | ||||
|
Baseline to 1st Year | ||||
|
Mean (SD) |
+5.4 (1.2) |
+9.1 (1.5)† |
+3.7 (2.9, 4.5) |
+8.9 (1.9) |
|
Baseline to 2nd Year | ||||
|
Mean (SD) |
+10.5 (1.9) |
+16.4 (2.0)† |
+5.8 (4.6, 7.1) |
+15.7 (2.7) |
|
Height SDS change | ||||
|
Baseline to 1st Year | ||||
|
Mean (SD) |
+0.1 (0.5) |
+0.7 (0.5)† |
+0.5 (0.3, 0.8) |
+0.8 (0.5) |
|
Baseline to 2nd Year | ||||
|
Mean (SD) |
+0.2 (0.5) |
+1.2 (0.7)† |
+1.0 (0.7, 1.3) |
+1.2 (0.7) |
|
Patients with height SDS> –2.0 at 2 years |
1 (4%) |
11 (41%)‡ |
8 (31%) |
|
INDICATIONS AND USAGE
Pediatric Patients — Humatrope is indicated for the treatment of pediatric patients who have growth failure due to an inadequate secretion of normal endogenous growth hormone.
Humatrope is indicated for the treatment of short stature associated with Turner syndrome in patients whose epiphyses are not closed.
Humatrope is indicated for the treatment of idiopathic short stature, also called non–growth hormone–deficient short stature, defined by height SDS ≤–2.25, and associated with growth rates unlikely to permit attainment of adult height in the normal range, in pediatric patients whose epiphyses are not closed and for whom diagnostic evaluation excludes other causes associated with short stature that should be observed or treated by other means.
Humatrope is indicated for the treatment of short stature or growth failure in children with SHOX (short stature homeobox-containing gene) deficiency whose epiphyses are not closed.
Adult Patients — Humatrope [somatropin (rDNA origin) for injection] is indicated for replacement of endogenous growth hormone in adults with growth hormone deficiency who meet either of the following two criteria:
1. Adult Onset: Patients who have growth hormone deficiency, either alone or associated with multiple hormone deficiencies (hypopituitarism), as a result of pituitary disease, hypothalamic disease, surgery, radiation therapy, or trauma; or
2. Childhood Onset: Patients who were growth hormone deficient during childhood as a result of congenital, genetic, acquired, or idiopathic causes.
In general, confirmation of the diagnosis of adult growth hormone deficiency in both groups usually requires an appropriate growth hormone stimulation test. However, confirmatory growth hormone stimulation testing may not be required in patients with congenital/genetic growth hormone deficiency or multiple pituitary hormone deficiencies due to organic disease.
CONTRAINDICATIONS
Patients with a known sensitivity to either Metacresol or glycerin should not receive Humatrope reconstituted with the supplied Diluent for Humatrope.
Somatropin should not be used for growth promotion in pediatric patients with closed epiphyses.
Somatropin is contraindicated in patients with proliferative or preproliferative diabetic retinopathy.
In general, somatropin is contraindicated in the presence of active malignancy. Any preexisting malignancy should be inactive and its treatment complete prior to instituting therapy with somatropin. Somatropin should be discontinued if there is evidence of recurrent activity. Since growth hormone deficiency may be an early sign of the presence of a pituitary tumor (or , rarely, other brain tumors), the presence of such tumors should be ruled out prior to initiation of treatment. Somatropin should not be used in patients with any evidence of progression or recurrence of an underlying intracranial tumor.
Somatropin should not be used to treat patients who have acute critical illness due to complications following open heart surgery, abdominal surgery or multiple accidental trauma, or those with acute respiratory failure. Two placebo–controlled clinical trials in non–growth hormone–deficient adult patients (n=522) with these conditions in intensive care units revealed a significant increase in mortality (41.9% vs. 19.3%) among somatropin–treated patients (doses 5.3 - 8 mg/day) compared to those receiving placebo (see WARNINGS).
Somatropin is contraindicated in patients with Prader–Willi syndrome who are severely obese or have severe respiratory impairment (see WARNINGS). Unless patients with Prader–Willi syndrome also have a diagnosis of growth hormone deficiency, Humatrope is not indicated for the treatment of pediatric patients who have growth failure due to genetically confirmed Prader–Willi syndrome.
WARNINGS
If sensitivity to the diluent should occur, the vials may be reconstituted with Bacteriostatic Water for Injection, USP or, Sterile Water for Injection, USP. When Humatrope is used with Bacteriostatic Water (Benzyl Alcohol preserved), the solution should be kept refrigerated at 2° to 8°C (36° to 46°F) and used within 14 days. Benzyl alcohol as a preservative in Bacteriostatic Water for Injection, USP has been associated with toxicity in newborns. When administering Humatrope to newborns, use the Humatrope diluent provided or if the patient is sensitive to the diluent, use Sterile Water for Injection, USP. When Humatrope is reconstituted with Sterile Water for Injection, USP in this manner, use only one dose per Humatrope vial and discard the unused portion. If the solution is not used immediately, it must be refrigerated [2° to 8°C (36° to 46°F)] and used within 24 hours.
Cartridges should be reconstituted only with the supplied diluent. Cartridges should not be reconstituted with the Diluent for Humatrope provided with Humatrope Vials, or with any other solution. Cartridges should not be used if the patient is allergic to Metacresol or glycerin.
See CONTRAINDICATIONS for information on increased mortality in patients with acute critical illness due to complications following open heart surgery, abdominal surgery, or multiple accidental trauma, or those with acute respiratory failure. The safety of continuing somatropin treatment in patients receiving replacement doses for approved indications who concurrently develop these illnesses has not been established. Therefore, the potential benefit of treatment continuation with somatropin in patients having acute critical illnesses should be weighed against the potential risk.
There have been reports of fatalities after initiating therapy with somatropin in pediatric patients with Prader–Willi syndrome who had one or more of the following risk factors: severe obesity, history of upper airway obstruction or sleep apnea, or unidentified respiratory infection. Male patients with one or more of these factors may be at greater risk than females. Patients with Prader–Willi syndrome should be evaluated for signs of upper airway obstruction and sleep apnea before initiation of treatment with somatropin. If, during treatment with somatropin, patients show signs of upper airway obstruction (including onset of or increased snoring) and/or new onset sleep apnea, treatment should be interrupted. All patients with Prader–Willi syndrome treated with somatropin should also have effective weight control and be monitored for signs of respiratory infection, which should be diagnosed as early as possible and treated aggressively (see CONTRAINDICATIONS). Unless patients with Prader–Willi syndrome also have a diagnosis of growth hormone deficiency, Humatrope is not indicated for the treatment of pediatric patients who have growth failure due to genetically confirmed Prader–Willi syndrome.
PRECAUTIONS
General — Therapy with Humatrope should be directed by physicians who are experienced in the diagnosis and management of pediatric patients with growth hormone deficiency, Turner syndrome, idiopathic short stature, SHOX deficiency, or adult patients with either childhood–onset or adult–onset growth hormone deficiency.
Treatment with somatropin may decrease insulin sensitivity, particularly at higher doses in susceptible patients. As a result, previously undiagnosed impaired glucose tolerance and overt diabetes mellitus may be unmasked during somatropin treatment. Therefore, glucose levels should be monitored periodically in all patients treated with somatropin, especially in those with risk factors for diabetes mellitus, such as obesity (including obese patients with Prader–Willi syndrome), Turner syndrome, or a family history of diabetes mellitus. Patients with preexisting type 1 or type 2 diabetes mellitus or impaired glucose tolerance should be monitored closely during somatropin therapy. The doses of antihyperglycemic drugs (i.e., insulin or oral agents) may require adjustment when somatropin therapy is instituted in these patients.
Patients with preexisting tumors or growth hormone deficiency secondary to an intracranial lesion should be examined routinely for progression or recurrence of the underlying disease process. In pediatric patients, clinical literature has revealed no relationship between somatropin replacement therapy and central nervous system (CNS) tumor recurrence or new extracranial tumors. However, in childhood cancer survivors, an increased risk of a second neoplasm has been reported in patients treated with somatropin after their first neoplasm. Intracranial tumors, in particular meningiomas, in patients treated with radiation to the head for their first neoplasm, were most common of these second neoplasms. In adults, it is unknown whether there is any relationship between somatropin replacement therapy and CNS tumor recurrence.
Intracranial hypertension (IH) with papilledema, visual changes, headache, nausea, and/or vomiting has been reported in a small number of patients treated with somatropin products. Symptoms usually occurred within the first eight (8) weeks after the initiation of somatropin therapy. In all reported cases, IH-associated signs and symptoms rapidly resolved after cessation of therapy or a reduction of the somatropin dose. Funduscopic examination should be performed routinely before initiating treatment with somatropin to exclude preexisting papilledema, and periodically during the course of somatropin therapy. If papilledema is observed by funduscopy during somatropin treatment, treatment should be stopped. If somatropin-induced IH is diagnosed, treatment with somatropin can be restarted at a lower dose after IH-associated signs and symptoms have resolved. Patients with Turner syndrome, chronic renal insufficiency, and Prader–Willi syndrome may be at increased risk for the development of IH.
In patients with hypopituitarism (multiple hormone deficiencies), standard hormonal replacement therapy should be monitored closely when somatropin therapy is administered.
Undiagnosed/untreated hypothyroidism may prevent an optimal response to somatropin, in particular, the growth response in children. Patients with Turner syndrome have an inherently increased risk of developing autoimmune thyroid disease and primary hypothyroidism. In patients with growth hormone deficiency, central (secondary) hypothyroidism may first become evident or worsen during somatropin treatment. Therefore, patients treated with somatropin should have periodic thyroid function tests and thyroid hormone replacement therapy should be initiated or appropriately adjusted when indicated.
Patients should be monitored carefully for any malignant transformation of skin lesions.
When somatropin is administered subcutaneously at the same site over a long period of time, tissue atrophy may result. This can be avoided by rotating the injection site.
As with any protein, local or systemic allergic reactions may occur. Parents/Patients should be informed that such reactions are possible and that prompt medical attention should be sought if allergic reactions occur.
Pediatric Patients (see PRECAUTIONS, General) — Slipped capital femoral epiphysis may occur more frequently in patients with endocrine disorders (including pediatric growth hormone deficiency and Turner syndrome) or in patients undergoing rapid growth. Any pediatric patient with the onset of a limp or complaints of hip or knee pain during somatropin therapy should be carefully evaluated.
Progression of scoliosis can occur in patients who experience rapid growth. Because somatropin increases growth rate, patients with a history of scoliosis who are treated with somatropin should be monitored for progression of scoliosis. However, somatropin has not been shown to increase the occurrence of scoliosis. Skeletal abnormalities including scoliosis are commonly seen in untreated Turner syndrome patients. Scoliosis is also commonly seen in untreated patients with Prader–Willi syndrome. Physicians should be alert to these abnormalities, which may manifest during somatropin therapy.
Patients with Turner syndrome should be evaluated carefully for otitis media and other ear disorders since these patients have an increased risk of ear and hearing disorders (see ADVERSE REACTIONS). Somatropin treatment may increase the occurrence of otitis media in patients with Turner syndrome. In addition, patients with Turner syndrome should be monitored closely for cardiovascular disorders (e.g., stroke, aortic aneurysm/dissection, hypertension) as these patients are also at risk for these conditions.
Adult Patients (see PRECAUTIONS, General)— Patients with epiphyseal closure who were treated with somatropin replacement therapy in childhood should be reevaluated according to the criteria in INDICATIONS AND USAGE before continuation of somatropin therapy at the reduced dose level recommended for growth hormone deficient adults. Fluid retention during somatropin replacement therapy in adults may occur. Clinical manifestations of fluid retention are usually transient and dose dependant (see ADVERSE REACTIONS).
Experience with prolonged somatropin treatment in adults is limited.
Information for Patients — Patients being treated with Humatrope (and/or their parents) should be informed about the potential benefits and risks associated with Humatrope treatment, including a review of the contents of the Patient Information Insert. This information is intended to better educate patients (and caregivers); it is not a disclosure of all possible adverse or intended effects.
Patients and caregivers who will administer Humatrope should receive appropriate training and instruction on the proper use of Humatrope from the physician or other suitably qualified health care professional. A puncture-resistant container for the disposal of used needles and syringes should be strongly recommended. Patients and/or parents should be thoroughly instructed in the importance of proper disposal, and cautioned against any reuse of needles and syringes. This information is intended to aid in the safe and effective administration of the medication (see Patient Information Insert).
Laboratory Tests — Serum levels of inorganic phosphorus, alkaline phosphatase, parathyroid hormone (PTH) and IGF-I may increase during somatropin therapy.
Drug Interactions — Somatropin inhibits 11β-hydroxysteroid dehydrogenase type 1 (11βHSD-1) in adipose/hepatic tissue and may significantly impact the metabolism of cortisol and cortisone. As a consequence, in patients treated with somatropin, previously undiagnosed central (secondary) hypoadrenalism may be unmasked requiring glucocorticoid replacement therapy. In addition, patients treated with glucocorticoid replacement therapy for previously diagnosed hypoadrenalism may require an increase in their maintenance or stress doses; this may be especially true for patients treated with cortisone acetate and prednisone since conversion of these drugs to their biologically active metabolites is dependent on the activity of the 11βHSD-1 enzyme.
Excessive glucocorticoid therapy may attenuate the growth promoting effects of somatropin in children. Therefore, glucocorticoid replacement therapy should be carefully adjusted in children with concomitant GH and glucocorticoid deficiency to avoid both hypoadrenalism and an inhibitory effect on growth.
Limited published data indicate that somatropin treatment increases cytochrome P450 (CP450) mediated antipyrine clearance in man. These data suggest that somatropin administration may alter the clearance of compounds known to be metabolized by CP450 liver enzymes (e.g., corticosteroids, sex steroids, anticonvulsants, cyclosporin). Careful monitoring is advisable when somatropin is administered in combination with other drugs known to be metabolized by CP450 liver enzymes. However, formal drug interaction studies have not been conducted.
In adult women on oral estrogen replacement, a larger dose of somatropin may be required to achieve the defined treatment goal (see DOSAGE AND ADMINISTRATION).
In patients with diabetes mellitus requiring drug therapy, the dose of insulin and/or oral agent may require adjustment when somatropin therapy is initiated (see PRECAUTIONS, General).
Carcinogenesis, Mutagenesis, Impairment of Fertility — Long–term animal studies for carcinogenicity and impairment of fertility with this human growth hormone (Humatrope) have not been performed. There has been no evidence to date of Humatrope–induced mutagenicity.
Pregnancy — Pregnancy Category C — Animal reproduction studies have not been conducted with Humatrope. It is not known whether Humatrope can cause fetal harm when administered to a pregnant woman or can affect reproductive capacity. Humatrope should be given to a pregnant woman only if clearly needed.
Nursing Mothers — There have been no studies conducted with Humatrope in nursing mothers. It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when Humatrope is administered to a nursing woman.
Geriatric Use — The safety and effectiveness of Humatrope in patients aged 65 and over has not been evaluated in clinical studies. Elderly patients may be more sensitive to the action of somatropin, and therefore may be more prone to develop adverse reactions. A lower starting dose and smaller dose increments should be considered for older patients (see DOSAGE AND ADMINISTRATION).
ADVERSE REACTIONS
Growth Hormone-Deficient Pediatric Patients
As with all protein pharmaceuticals, a small percentage of patients may develop antibodies to the protein. During the first 6 months of Humatrope therapy in 314 naive patients, only 1.6% developed specific antibodies to Humatrope (binding capacity ≥0.02 mg/L). None had antibody concentrations which exceeded 2 mg/L. Throughout 8 years of this same study, two patients (0.6%) had binding capacity >2 mg/L. Neither patient demonstrated a decrease in growth velocity at or near the time of increased antibody production. It has been reported that growth attenuation from pituitary–derived growth hormone may occur when antibody concentrations are >1.5 mg/L.
In addition to an evaluation of compliance with the treatment program and of thyroid status, testing for antibodies to human growth hormone should be carried out in any patient who fails to respond to therapy.
In studies with growth hormone–deficient pediatric patients, injection site pain was reported infrequently. A mild and transient edema, which appeared in 2.5% of patients, was observed early during the course of treatment.
Leukemia has been reported in a small number of pediatric patients who have been treated with growth hormone, including growth hormone of pituitary origin as well as of recombinant DNA origin (somatrem and somatropin). The relationship, if any, between leukemia and growth hormone therapy is uncertain.
Patients with Turner Syndrome
In a randomized, concurrent controlled trial, there was a statistically significant increase in the occurrence of otitis media (43% vs. 26%), ear disorders (18% vs. 5%) and surgical procedures (45% vs. 27%) in patients receiving Humatrope compared with untreated control patients (Table 7). Other adverse events of special interest to Turner syndrome patients were not significantly different between treatment groups (Table 7). A similar increase in otitis media was observed in an 18–month placebo–controlled trial.
|
Treatment Group | |||
|
Adverse Event |
Untreated* |
Humatrope† |
Significance‡ |
|
Total Number of Patients |
62 |
74 | |
|
Surgical procedure |
17 (27.4%) |
33 (44.6%) |
p≤0.05 |
|
Otitis media |
16 (25.8%) |
32 (43.2%) |
p≤0.05 |
|
Ear disorders |
3 (4.8%) |
13 (17.6%) |
p≤0.05 |
|
Bone disorder |
7 (11.3%) |
6 (8.1%) |
NS |
|
Edema | |||
|
Conjunctival |
1 (1.6%) |
0 |
NS |
|
Non-specific |
1 (1.6%) |
2 (2.7%) |
NS |
|
Facial |
0 |
1 (1.4%) |
NS |
|
Peripheral |
1 (1.6%) |
5 (6.8%) |
NS |
|
Hyperglycemia |
0 |
0 |
NS |
|
Hypothyroidism |
5 (8.1%) |
10 (13.5%) |
NS |
|
Increased nevi§ |
2 (3.2%) |
8 (10.8%) |
NS |
|
Lymphedema |
0 |
0 |
NS |
Patients with Idiopathic Short Stature
In the placebo–controlled study, the adverse events associated with Humatrope therapy were similar to those observed in other pediatric populations treated with Humatrope (Table 8). Mean serum glucose level did not change during Humatrope treatment. Mean fasting serum insulin levels increased 10% in the Humatrope treatment group at the end of treatment relative to baseline values but remained within the normal reference range. For the same duration of treatment the mean fasting serum insulin levels decreased by 2% in the placebo group. The incidence of above–range values for glucose, insulin, and HbA1c were similar in the growth hormone and placebo–treated groups. No patient developed diabetes mellitus. Consistent with the known mechanism of growth hormone action, Humatrope–treated patients had greater mean increases, relative to baseline, in serum insulin–like growth factor–I (IGF–I) than placebo–treated patients at each study observation. However, there was no significant difference between the Humatrope and placebo treatment groups in the proportion of patients who had at least one serum IGF–I concentration more than 2.0 SD above the age– and gender–appropriate mean (Humatrope: 9 of 35 patients [26%]; placebo: 7 of 28 patients [25%]).
|
Treatment Group |
||
|
Adverse Event |
Humatrope |
Placebo |
|
Total Number of Patients |
37 |
31 |
|
Scoliosis |
7 (18.9%) |
4 (12.9%) |
|
Otitis media |
6 (16.2%) |
2 (6.5%) |
|
Hyperlipidemia |
3 (8.1%) |
1 (3.2%) |
|
Gynecomastia |
2 (5.4%) |
1 (3.2%) |
|
Hypothyroidism |
0 |
2 (6.5%) |
|
Aching joints |
0 |
1 (3.2%) |
|
Hip pain |
1 (2.7%) |
0 |
|
Arthralgia |
4 (10.8%) |
1 (3.2%) |
|
Arthrosis |
4 (10.8%) |
2 (6.5%) |
|
Myalgia |
9 (24.3%) |
4 (12.9%) |
|
Hypertension |
1 (2.7%) |
0 |
The adverse events observed in the dose–response study (239 patients treated for 2 years) did not indicate a pattern suggestive of a growth hormone dose effect. Among Humatrope dose groups, mean fasting blood glucose, mean glycosylated hemoglobin, and the incidence of elevated fasting blood glucose concentrations were similar. One patient developed abnormalities of carbohydrate metabolism (glucose intolerance and high serum HbA1c) on treatment.
Patients with SHOX Deficiency
“Clinically significant” adverse events (adverse events previously observed in association with growth hormone treatment in general) were assessed prospectively during the 2-year randomized, open-label study; those observed are presented in Table 9. In both treatment groups, the mean fasting plasma glucose concentration at the end of the first year was similar to the baseline value and remained in the normal range. No patient developed diabetes mellitus or had an above normal value for fasting plasma glucose at the end of one-year of treatment. During the 2 year study period, the proportion of patients who had at least one IGF-I concentration greater than 2.0 SD above the age- and gender-appropriate mean was 10 of 27 [37.0%] for the Humatrope-treated group vs. 0 of 24 patients [0.0%] for the untreated group. The proportion of patients who had at least one IGFBP-3 concentration greater than 2.0 SD above the age and gender appropriate mean was 16 of 27 [59.3%] for the Humatrope treated group vs. 7 of 24 [29.2%] for the untreated group.
|
Adverse Event |
Treatment Group |
|
|
Untreated |
Humatrope |
|
|
Total Number of Patients |
25 |
27 |
|
Patients with at least one event |
2 |
5 |
|
Arthralgia |
2 (8.0%) |
3 (11.1%) |
|
Gynecomastia‡ |
0 (0.0%) |
1 (8.3%) |
|
Excessive number of cutaneous nevi |
0 (0.0%) |
2 (7.4%) |
|
Scoliosis |
0 (0.0%) |
1 (3.7%) |
Adult Patients — In clinical studies in which high doses of Humatrope were administered to healthy adult volunteers, the following events occurred infrequently: headache, localized muscle pain, weakness, mild hyperglycemia, and glucosuria.
In the first 6 months of controlled blinded trials during which patients received either Humatrope or placebo, adult–onset growth hormone–deficient adults who received Humatrope experienced a statistically significant increase in edema (Humatrope 17.3% vs. placebo 4.4%, p=0.043) and peripheral edema (11.5% vs. 0%, respectively, p=0.017). In patients with adult–onset growth hormone deficiency, edema, muscle pain, joint pain, and joint disorder were reported early in therapy and tended to be transient or responsive to dosage titration.
Two of 113 adult–onset patients developed carpal tunnel syndrome after beginning maintenance therapy without a low dose (0.00625 mg/kg/day) lead–in phase. Symptoms abated in these patients after dosage reduction.
All treatment–emergent adverse events with ≥5% overall incidence during 12 or 18 months of replacement therapy with Humatrope are shown in Table 10 (adult–onset patients) and in Table 11 (childhood–onset patients).
Adult patients treated with Humatrope who had been diagnosed with growth hormone deficiency in childhood reported side effects less frequently than those with adult–onset growth hormone deficiency.
|
18 Months
Exposure |
|
|||
|
Adverse Event |
n |
% |
n |
% |
|
Edema† |
7 |
15.2 |
11 |
21.2 |
|
Arthralgia |
7 |
15.2 |
9 |
17.3 |
|
Paresthesia |
6 |
13.0 |
9 |
17.3 |
|
Myalgia |
6 |
13.0 |
7 |
13.5 |
|
Pain |
6 |
13.0 |
7 |
13.5 |
|
Rhinitis |
5 |
10.9 |
7 |
13.5 |
|
Peripheral edema‡ |
8 |
17.4 |
6 |
11.5 |
|
Back pain |
5 |
10.9 |
5 |
9.6 |
|
Headache |
5 |
10.9 |
4 |
7.7 |
|
Hypertension |
2 |
4.3 |
4 |
7.7 |
|
Acne |
0 |
0 |
3 |
5.8 |
|
Joint disorder |
1 |
2.2 |
3 |
5.8 |
|
Surgical procedure |
1 |
2.2 |
3 |
5.8 |
|
Flu syndrome |
3 |
6.5 |
2 |
3.9 |
|
18 Months
Exposure |
|
|||
|
Adverse Event |
n |
% |
n |
% |
|
||||
|
Flu syndrome |
8 |
22.9 |
5 |
15.6 |
|
AST increased† |
2 |
5.7 |
4 |
12.5 |
|
Headache |
4 |
11.4 |
3 |
9.4 |
|
Asthenia |
1 |
2.9 |
2 |
6.3 |
|
Cough increased |
0 |
0 |
2 |
6.3 |
|
Edema |
3 |
8.6 |
2 |
6.3 |
|
Hypesthesia |
0 |
0 |
2 |
6.3 |
|
Myalgia |
2 |
5.7 |
2 |
6.3 |
|
Pain |
3 |
8.6 |
2 |
6.3 |
|
Rhinitis |
2 |
5.7 |
2 |
6.3 |
|
ALT increased |
2 |
5.7 |
2 |
6.3 |
|
Respiratory disorder |
2 |
5.7 |
1 |
3.1 |
|
Gastritis |
2 |
5.7 |
0 |
0 |
|
Pharyngitis |
5 |
14.3 |
1 |
3.1 |
Other adverse drug events that have been reported in growth hormone–treated patients include the following:
-
Metabolic: Infrequent, mild and transient peripheral or generalized edema.
-
Musculoskeletal: Rare carpal tunnel syndrome.
-
Skin: Rare increased growth of pre–existing nevi. Patients should be monitored carefully for malignant transformation.
-
Endocrine: Rare gynecomastia. Rare pancreatitis.
OVERDOSAGE
Acute overdosage could lead initially to hypoglycemia and subsequently to hyperglycemia. Long–term overdosage could result in signs and symptoms of gigantism/acromegaly consistent with the known effects of excess human growth hormone. (See recommended and maximal dosage instructions given below.)
DOSAGE AND ADMINISTRATION
Pediatric Patients
The Humatrope dosage and administration schedule should be individualized for each patient. Therapy should not be continued if epiphyseal fusion has occurred. Response to growth hormone therapy tends to decrease with time. However, failure to increase growth rate, particularly during the first year of therapy, should prompt close assessment of compliance and evaluation of other causes of growth failure such as hypothyroidism, under–nutrition and advanced bone age.
Growth hormone–deficient pediatric patients — The recommended weekly dosage is 0.18 mg/kg (0.54 IU/kg) of body weight. The maximal replacement weekly dosage is 0.3 mg/kg (0.90 IU/kg) of body weight. It should be divided into equal doses given either on 3 alternate days, 6 times per week or daily. The subcutaneous route of administration is preferable; intramuscular injection is also acceptable. The dosage and administration schedule for Humatrope should be individualized for each patient.
Turner Syndrome — A weekly dosage of up to 0.375 mg/kg (1.125 IU/kg) of body weight administered by subcutaneous injection is recommended. It should be divided into equal doses given either daily or on 3 alternate days.
Patients with idiopathic short stature — A weekly dosage of up to 0.37 mg/kg of body weight administered by subcutaneous injection is recommended. It should be divided into equal doses given 6 to 7 times per week.
Patients with SHOX deficiency — A weekly dosage of 0.35 mg/kg of body weight is recommended. It should be divided into equal doses given by daily subcutaneous injection.
Adult Patients
Adult Growth Hormone Deficiency (GHD) — Based on the weight-based dosing utilized in the original pivotal studies described herein, the recommended dosage at the start of therapy is not more than 0.006 mg/kg given as a daily subcutaneous injection. The dose may be increased according to individual patient requirements to a maximum of 0.0125 mg/kg daily in patients. Clinical response, side effects, and determination of age- and gender-adjusted serum IGF-I levels may be used as guidance in dose titration.
Alternatively, taking into account recent literature, a starting dose of approximately 0.2 mg/day (range, 0.15-0.30 mg/day) may be used without consideration of body weight. This dose can be increased gradually every 1-2 months by increments of approximately 0.1-0.2 mg/day, according to individual patient requirements based on the clinical response and serum IGF-I concentrations. During therapy, the dose should be decreased if required by the occurrence of adverse events and/or serum IGF–I levels above the age- and gender-specific normal range. Maintenance dosages vary considerably from person to person.
A lower starting dose and smaller dose increments should be considered for older patients, who are more prone to the adverse effects of somatropin than younger individuals. In addition, obese individuals are more likely to manifest adverse effects when treated with a weight-based regimen. In order to reach the defined treatment goal, estrogen-replete women may need higher doses than men. Oral estrogen administration may increase the dose requirements in women.
Reconstitution
Vial — Each 5–mg vial of Humatrope should be reconstituted with 1.5 to 5 mL of Diluent for Humatrope. The diluent should be injected into the vial of Humatrope by aiming the stream of liquid against the glass wall. Following reconstitution, the vial should be swirled with a GENTLE rotary motion until the contents are completely dissolved. DO NOT SHAKE. The resulting solution should be inspected for clarity. It should be clear. If the solution is cloudy or contains particulate matter, the contents MUST NOT be injected.
Before and after injection, the septum of the vial should be wiped with rubbing alcohol or an alcoholic antiseptic solution to prevent contamination of the contents by repeated needle insertions. Sterile disposable syringes and needles should be used for administration of Humatrope. The volume of the syringe should be small enough so that the prescribed dose can be withdrawn from the vial with reasonable accuracy.
Cartridge — Each cartridge of Humatrope should only be reconstituted using the diluent syringe that accompanies the cartridge and should not be reconstituted with the Diluent for Humatrope provided with Humatrope Vials. (See WARNINGS section.) See Information for the Patient for comprehensive directions on Humatrope cartridge reconstitution.
The reconstituted solution should be inspected for clarity. It should be clear. If the solution is cloudy or contains particulate matter, the contents MUST NOT be injected.
The somatropin concentrations for the reconstituted Humatrope cartridges are as follows: 2.08 mg/mL for the 6 mg cartridge; 4.17 mg/mL for the 12 mg cartridge; and 8.33 mg/mL for the 24 mg cartridge.
This cartridge has been designed for use only with the Humatrope injection device. A sterile disposable needle should be used for each injection of Humatrope.
STABILITY AND STORAGE
Vials
Before Reconstitution — Vials of Humatrope and Diluent for Humatrope are stable when refrigerated [2° to 8°C (36° to 46°F)]. Avoid freezing Diluent for Humatrope. Expiration dates are stated on the labels.
After Reconstitution — Vials of Humatrope are stable for up to 14 days when reconstituted with Diluent for Humatrope or Bacteriostatic Water for Injection, USP and stored in a refrigerator at 2° to 8°C (36° to 46°F). Avoid freezing the reconstituted vial of Humatrope.
After Reconstitution with Sterile Water, USP — Use only one dose per Humatrope vialand discard the unused portion. If the solution is not used immediately, it must be refrigerated [2° to 8°C (36° to 46°F)] and used within 24 hours.
Cartridges
Before Reconstitution — Cartridges of Humatrope and Diluent for Humatrope are stable when refrigerated [2° to 8°C (36° to 46°F)]. Avoid freezing Diluent for Humatrope. Expiration dates are stated on the labels.
After Reconstitution — Cartridges of Humatrope are stable for up to 28 days when reconstituted with Diluent for Humatrope and stored in a refrigerator at 2° to 8°C (36° to 46°F). Store the Humatrope injection device without the needle attached. Avoid freezing the reconstituted cartridge of Humatrope.
HOW SUPPLIED
Vials
|
5 mg (No. 7335) — (6s) NDC 0002–7335–16, and 5–mL vials of Diluent for Humatrope (No. 7336) |
Cartridges
|
Cartridge Kit (MS8147) NDC 0002–8147–01 |
|
6 mg cartridge (VL7554), and prefilled syringe of Diluent for Humatrope (VL7618) |
|
Cartridge Kit (MS8148) NDC 0002–8148–01 |
|
12 mg cartridge (VL7555), and prefilled syringe of Diluent for Humatrope (VL7619) |
|
Cartridge Kit (MS8149) NDC 0002–8149–01 |
|
24 mg cartridge (VL7556), and prefilled syringe of Diluent for Humatrope (VL7619) |
Literature revised November 1, 2006
Manufactured by Lilly France
F-67640
Fegersheim, France
for Eli Lilly and Company
Indianapolis, IN 46285, USA
www.humatrope.com
Supplement Patient Material Section
INFORMATION
AND PATIENT INSTRUCTIONS
HUMATROPE®
Somatropin (rDNA
origin) for Injection
CARTRIDGES
HUMATROPE CARTRIDGES ARE ONLY TO BE USED WITH HUMATROPEN® OR HUMATROPEN® 3 INJECTION DEVICES.
Important Things to Know
It is important to learn the names of the parts of the Humatrope Cartridge Kit and how these parts work before injecting yourself or your child. Make sure you have been properly trained by your nurse, pharmacist or doctor before you mix the drug (add the diluent liquid to the dry Humatrope powder) or inject it. Wash your hands and be careful to follow the instructions given to you by your nurse, pharmacist or doctor. After mixing, throw away the diluent syringe in a puncture–resistant container such as the type your nurse, pharmacist or doctor has told you to use.
Storage
Humatrope must be kept refrigerated (36° to 46°F [2° to 8°C]) before and after it is mixed. Do not freeze. Once Humatrope has been mixed and is in liquid form, it must be used within 28 days. Throw away any mixed Humatrope left over after 28 days. Before giving an injection, check the date on the cartridge. Do not use the cartridge if it has expired.
WARNING
HUMATROPE CARTRIDGES SHOULD NOT BE USED IF THE PATIENT IS ALLERGIC TO METACRESOL OR GLYCERIN.
Contents
-
one cartridge with 6, 12, or 24 mg of dried Humatrope
-
one prefilled syringe with diluent (the liquid used to mix the dried Humatrope)
NOTE: There are three kinds of Humatrope cartridges that have different amounts of Humatrope (6, 12, or 24 mg). Make sure that you have the cartridge that your doctor prescribed.
Mixing the Humatrope in the Cartridge
Use only the prefilled diluent syringe to mix the Humatrope in the cartridge. DO NOT use the diluent that comes in the Humatrope vial box, or any other liquid.
Reconstitution Instructions
|
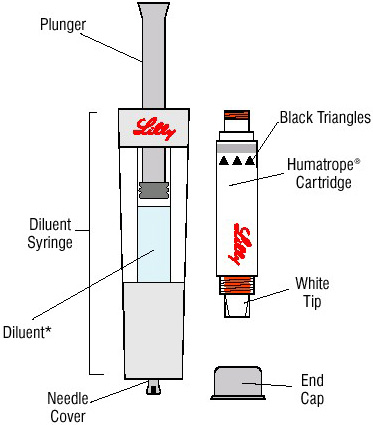
Parts |
 |
|
Use only this kit to prepare the Humatrope cartridge. |
|
*Note: The liquid is colorless. |
|
Preparing Your New Cartridge |
||
 |
 |
 |
|
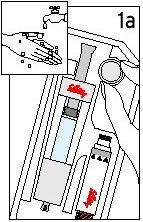
Remove ALL contents from the tray. Note: This product is designed for left or right handed use so you may use whichever hand is most comfortable for you. |
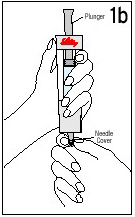
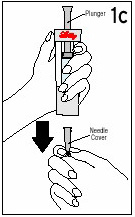
Grasp the gray Needle Cover, at the bottom of the Diluent Syringe. |
Remove the Needle Cover and discard. DO NOT depress the Plunger yet. It is okay if a drop of fluid is lost. It is not necessary to release air from the Diluent Syringe. |
 |
 |
 |
|
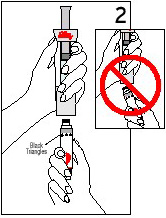
Hold the cartridge, with the Black Triangles toward the Diluent Syringe. Align the cartridge and Diluent Syringe in a straight line. DO NOT insert the cartridge at an angle. |
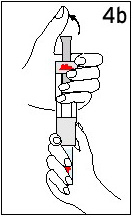
PUSH the cartridge STRAIGHT in until it stops AND the Black Triangles ARE COVERED. You may hear or feel a click. DO NOT twist the cartridge. |
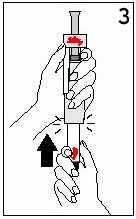
Hold the Diluent Syringe and the cartridge together with TWO HANDS. Push and release the Plunger 2 or 3 times until the Diluent is in the cartridge. |
 |
 |
 |
|
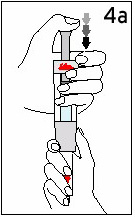
Remove your thumb from the Plunger and check that the Diluent Syringe is empty [it is normal for small drops of Diluent to remain in the Diluent Syringe]. |
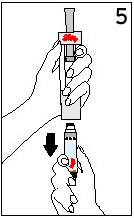
With your thumb OFF the Plunger, pull the cartridge away from the Diluent Syringe. |
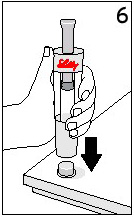
Place the End Cap on a hard, flat surface. Push the Diluent Syringe onto the End Cap and immediately discard the Diluent Syringe as instructed by your healthcare professional. |
 |
 |
|
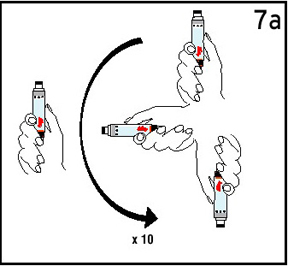
Mix the cartridge by gently inverting 10 times and let it sit for 3 minutes, DO NOT SHAKE. |
Inspect the solution. The Humatrope solution should be clear. If the solution is clear, your cartridge is now prepared and ready to be attached to your pen injection device (see the User Manual for your pen injection device). If the solution is cloudy or contains particles, gently invert the cartridge 10 additional times. Let the cartridge sit for 5 more minutes. If the solution remains cloudy or contains particles, DO NOT USE THE CARTRIDGE. Contact your healthcare professional or Lilly. If you have questions about preparing your Humatrope cartridge, you should contact your Humatrope provider or your healthcare professional. |
Injections can be given in the following areas:
-
Abdomen (above, below, or either side of the navel)
-
Front of the upper thighs
-
Upper, outer buttocks
-
Back of the arms above the elbow and below the shoulder
Discuss use of the pen injection device, the right places to inject, and site rotation with your nurse or doctor.
Literature revised August 1, 2005
Manufactured by Lilly France S.A.S.
F-67640
Fegersheim, France
for Eli Lilly and Company
Indianapolis,
IN 46285, USA
HUMATROPE®
Somatropin (rDNA
origin) for Injection
INFORMATION FOR THE PATIENT
Do not mix (reconstitute) the drug or inject it until you have been thoroughly trained in the proper techniques by your doctor. Use sterile techniques as instructed by your doctor. Destroy and discard syringes and/or needles after each use.
Humatrope should be kept refrigerated (36° to 46°F [2°to 8°C]) before and after reconstitution. Do not freeze. Reconstituted Humatrope should be used within 14 days.
Reconstituting the Vial of Humatrope
Reconstitute Humatrope only with Diluent for Humatrope. Do not use other solutions for reconstitution unless instructed to do so by your doctor. Your doctor will also tell you what size syringe and needle to use and how much diluent to add to the vial of Humatrope.
Always start by washing your hands.
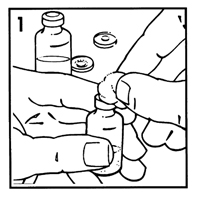
-
Remove and discard plastic caps from tops of vials of diluent and Humatrope. Wipe tops of both vials with an alcohol swab (Figure 1). Remove needle cover and save. Pull back on syringe plunger to draw up an amount of air equal to the amount of diluent your doctor has prescribed. Insert needle in stopper of diluent vial, and inject air into vial.
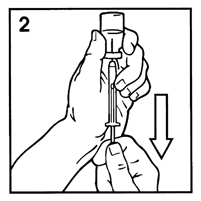
-
Hold vial upside down and, making sure needle tip remains in solution, withdraw the amount of diluent your doctor has prescribed (Figure 2). After making sure that no air bubbles are in the syringe, turn vial upright and, holding barrel, remove syringe.
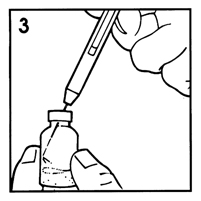
-
Insert same needle into vial of Humatrope and gently aim needle tip toward wall of vial. Slowly inject the diluent by aiming the stream of liquid against the wall of vial (Figure 3). Do not aim it at the white powder at the bottom of the vial. To equalize the pressure, withdraw a volume of air equal to the amount of diluent added before removing the syringe from the vial. If the needle can be removed from the barrel of the syringe, remove, destroy, and discard the needle. If the needle and syringe are made as 1 unit, destroy and discard the entire unit.
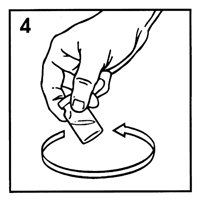
-
Swirl the vial with a gentle rotary motion until contents are completely dissolved (Figure 4). Do not shake.
Preparing the Injection
-
Do not use reconstituted Humatrope if it is cloudy or contains particles.
-
If the needle can be removed from the type of syringe you are using, a new needle should be placed on the syringe before the injection. If the syringe and needle are made as 1 unit, another unit should be used for the injection.
-
Before and after injection, the rubber stopper of the vial should be wiped with rubbing alcohol or an alcoholic antiseptic solution to prevent contamination of the contents by repeated needle insertions.
-
Remove the needle cover and draw an amount of air into the syringe equal to your dose of Humatrope.
-
Insert needle into vial of reconstituted Humatrope and inject the air into the vial. Turn the vial upside down, and, making sure needle tip is in solution, withdraw your correct dose (see Figure 2). Make sure that no air bubbles are in the syringe.
-
Remove syringe and replace needle cover. Write date of reconstitution on vial label, and discard unused diluent.
-
Return unused portion of reconstituted Humatrope to refrigerator and use within 14 days.
-
Destroy needle or the needle and syringe after use.
Injecting Humatrope
-
Gently tap injection site several times with fingers.
-
Wipe the area thoroughly with an alcohol swab. Use a circular motion and work outward from the inside of the circle.
-
Subcutaneous Injection: With the thumb and forefinger, stabilize the skin by spreading or pinching up a large area of skin.
-
Holding the syringe at a 90-degree angle to injection site, quickly insert the needle all the way into the skin.
-
Slowly inject the solution.
-
Remove the needle quickly, and apply pressure over the injection site with a dry gauze pad or cotton ball. Rub for several seconds.
-
Destroy needle or needle and syringe after use.
-
-
Intramuscular Injection: With the thumb and first 2 fingers, press the skin down firmly against a large muscle mass, such as the thigh.
-
Holding the syringe at a 90-degree angle to injection site, quickly insert the needle all the way into the skin.
-
When the needle is in place, slowly pull back on the plunger. If blood enters the syringe, remove needle, discard syringe and drug, and prepare another injection.
-
If no blood enters the syringe, slowly inject the solution.
-
Remove the needle quickly, and apply pressure over the injection site with a dry gauze pad or cotton ball. Rub for several seconds.
-
Destroy needle or needle and syringe after use.
-
If you have any questions, consult your doctor.
Text revised July 6, 1990
Literature revised March 1,
2002
ELI LILLY AND COMPANY, INDIANAPOLIS, IN 46285, USA
PRINTED IN USA
| HUMATROPE (Somatropin) | |||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| HUMATROPE (Somatropin) | |||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| HUMATROPE (Somatropin) | |||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| HUMATROPE (Somatropin) | |||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
Revised: 02/2007ELI LILLY AND COMPANY