CHILDRENS ACETAMINOPHEN
-
acetaminophen suspension
ParaPRO LLC
----------
NDC 52246-023-04
Children's Acetaminophen
oral suspension
80 mg per 1/2 teaspoon (160 mg per 5 mL)
fever reducer/pain releiver
4 FL OZ (118 mL)
Purpose
Pain reliever/fever reducer
Warnings
Liver warning: This product contains acetaminophen. Severe liver damage may occur if your child takes:
- more than 5 doses in 24 hours, which is the maximum daily amount
- with other drugs containing acetaminophen
Do not use with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, as a doctor or pharmacist.
Ask a doctor before use if you child has liver disease.
Ask a doctor or pharmacist before use if your child is taking the blood thinning drug warfarin.
Stop use and ask a doctor if
- pain gets worse or lasts more than 5 days
- fever gets worse or lasts more than 3 days
- new symptoms occur
- redness or swelling is present
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away (1-800-222-1222). Quick medical attention is critical even if you do not notice any signs or symptoms.
Directions
- do not give more than directed (see Liver warning)
- shake well before using
- find right dose on chart. If possible, use weight to dose; otherwise, use age.
- use only enclosed dosing cup designed for use with this product. Do not use any other dosing device
-
- if needed, repeat dose every 4 hours while symptoms last
- do not give more than 5 times in 24 hours
- do not give more than 5 days unless directed by a doctor
- this product does not contain directions or complete warnings for adult use
Under 24 Under 2 years ask a doctor
24-35 2-3 years 1 tsp or 5 mL
36-47 4-5 years 1 1/2 tsp or 7.5 mL
48-59 6-8 years 2 tsp or 10 mL
60-71 9-10 years 2 1/2 tsp or 12.5 mL
72-95 11 years 3 tsp or 15 mL
Attention: use only enclosed dosing cup specifically designed for use with this product. Do not use any other dosing device.
Other information:
- each teaspoon contains: sodium 3 mg
- store at 20o - 25oC (68o - 77oF)
DO NOT USE IF PRINTED SAFETY SEAL IS BROKEN OR MISSING.
Inactive ingredients: anhydrous citric acid, butylparaben, caboxymethylcellulose sodium, carrageenan, D and C red no. 33, FD and C blue no.1, flavor, glycerin, high fructose corn syrup, hodroxyethyl cellulose, microcrystalline cellulose, propylene glycol, purified water, sodium benzoate, sorbitol solution
Active ingredient (in each 5 mL = 1 teaspoon)
Acetaminophen 160 mg
Bottle Label
bl.jpg

cl.jpg
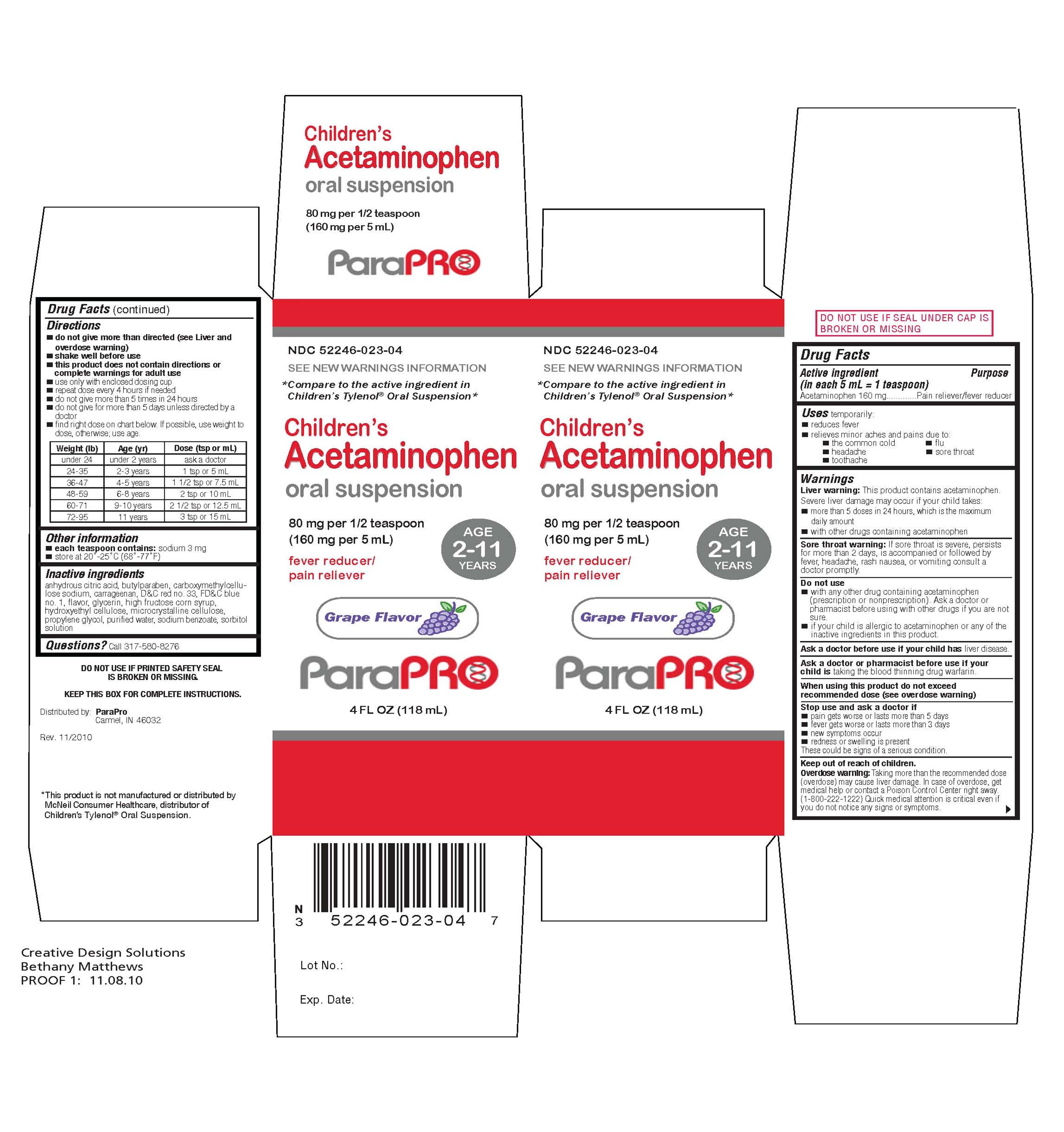
Uses temporarily:
- reduces fever
- relieve minor aches and pain due to:
-
- the common cold
- flu
- headache
- sore throat
- toothache
| CHILDRENS ACETAMINOPHEN
acetaminophen suspension |
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| OTC monograph not final | part343 | 01/03/2011 | 05/16/2011 |
| Labeler - ParaPRO LLC (186639006) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| ParaPRO LLC | 186639006 | manufacture | |